
THERMODYNAMICS (LL)-W/ACCESS >CUSTOM<
9th Edition
ISBN: 9781266657610
Author: CENGEL
Publisher: MCG CUSTOM
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 15.7, Problem 89P
Reconsider Prob. 15–88. The automobile engine is to be converted to natural gas (methane, CH4) fuel. Assuming that all factors remain the same, what is the maximum work that can be produced by the modified engine, in kJ/kg fuel?
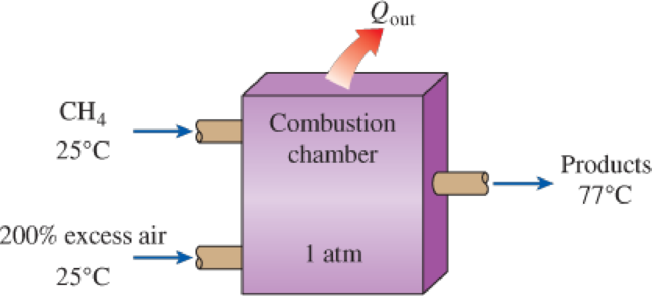
FIGURE P15–89
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Multiple Choice
Circle the best answer to each statement.
1. Which geometry attribute deviation(s) can be limited
with a profile of a surface tolerance?
A. Location
B. Orientation
C.
Form
D.
All of the above
2. A true profile may be defined with:
A.
Basic radii
B. Basic angles
C. Formulas
D. All of the above
3. Which modifier may be applied to the profile tolerance
value?
A
B
C.
D. All of the above
4. The default tolerance zone for a profile tolerance is:
A. Non-uniform
B. Unilateral
C. Bilateral equal distribution
D. Bilateral-unequal distribution
5. An advantage of using a profile tolerance in place of a
coordinate tolerance is:
A. A bonus tolerance is permitted.
B. A datum feature sequence may be specified
C. A profile tolerance always controls size
D. All of the above
6. The shape of the tolerance zone for a profile tolerance is:
A. Two parallel planes
B. The same as the true profile of the toleranced
surface
C. Equal bilateral
D. Cylindrical when the diameter symbol is speci-
fied…
One thousand kg/h of a (50-50 wt%) acetone-in-water solution is to be extracted at 25C in a continuous,
countercurrent system with pure 1,1,2-trichloroethane to obtain a raffinate containing 10 wt% acetone. Using the
following equilibrium data, determine with an equilateral-triangle diagram:
a- the minimum flow rate of solvent;
b- the number of stages required for a solvent rate equal to 1.5 times minimum, and composition of each
streamleaving each stage.
c- Repeat the calculation of (a) and (b) if the solvent used has purity 93wt% (4wr% acetone, 3wt% water
impurities)
acetone water
1,1,2-trichloroethane
Raffinate. Weight
Extract. Weight
0.6
0.13
0.27
Fraction Acetone
Fraction Acetone
0.5
0.04
0.46
0.44
0.56
0.4
0.03
0.57
0.29
0.40
0.3
0.02
0.68
0.12
0.18
0.2
0.015
0.785
0.0
0.0
0.1
0.01
0.89
0.55
0.35
0.1
0.5
0.43
0.07
0.4
0.57
0.03
0.3
0.68
0.02
0.2
0.79
0.01
0.1
0.895
0.005
2500 kg/hr of (20-80) nicotine water solution is to be extracted with benzene containing 0.5% nicotine in
the 1st and 2ed stages while the 3rd stage is free of nicotine. Cross- current operation is used with different amounts
of solvent for each stages 2000kg/hr in the 1st stage, 2300 kg/hr in the 2nd stage, 2600 kg/hr in the 3rd,
determine: -
a- The final raffinate concentration and % extraction.
b-
b- The minimum amount of solvent required for counter-current operation if the minimum concentration
will be reduced to 5% in the outlet raffinate.
Equilibrium data
Wt % Nicotine in water
Wt % Nicotine in benzene
0
4
16
25
0
4
21
30
Chapter 15 Solutions
THERMODYNAMICS (LL)-W/ACCESS >CUSTOM<
Ch. 15.7 - What are the approximate chemical compositions of...Ch. 15.7 - How does the presence of N2 in air affect the...Ch. 15.7 - Prob. 3PCh. 15.7 - Prob. 4PCh. 15.7 - Is the airfuel ratio expressed on a mole basis...Ch. 15.7 - How does the presence of moisture in air affect...Ch. 15.7 - Prob. 7PCh. 15.7 - Prob. 8PCh. 15.7 - Prob. 9PCh. 15.7 - Are complete combustion and theoretical combustion...
Ch. 15.7 - What does 100 percent theoretical air represent?Ch. 15.7 - Consider a fuel that is burned with (a) 130...Ch. 15.7 - What are the causes of incomplete combustion?Ch. 15.7 - Which is more likely to be found in the products...Ch. 15.7 - Methane (CH4) is burned with the stoichiometric...Ch. 15.7 - Prob. 16PCh. 15.7 - n-Butane fuel (C4H10) is burned with the...Ch. 15.7 - Prob. 18PCh. 15.7 - Propane (C3H8) is burned with 75 percent excess...Ch. 15.7 - Propane fuel (C3H8) is burned with 30 percent...Ch. 15.7 - In a combustion chamber, ethane (C2H6) is burned...Ch. 15.7 - Prob. 22PCh. 15.7 - Prob. 23PCh. 15.7 - Ethane (C2H6) is burned with 20 percent excess air...Ch. 15.7 - Octane (C8H18) is burned with 250 percent...Ch. 15.7 - Prob. 26PCh. 15.7 - A fuel mixture of 60 percent by mass methane (CH4)...Ch. 15.7 - Prob. 28PCh. 15.7 - A certain natural gas has the following volumetric...Ch. 15.7 - Prob. 30PCh. 15.7 - A gaseous fuel with a volumetric analysis of 45...Ch. 15.7 - Prob. 33PCh. 15.7 - The fuel mixer in a natural gas burner mixes...Ch. 15.7 - Prob. 35PCh. 15.7 - Prob. 36PCh. 15.7 - Determine the fuelair ratio when coal from...Ch. 15.7 - Prob. 38PCh. 15.7 - Prob. 39PCh. 15.7 - Prob. 40PCh. 15.7 - Prob. 41PCh. 15.7 - When are the enthalpy of formation and the...Ch. 15.7 - Prob. 43PCh. 15.7 - Prob. 44PCh. 15.7 - Prob. 45PCh. 15.7 - Prob. 46PCh. 15.7 - Prob. 48PCh. 15.7 - Repeat Prob. 1546 for liquid octane (C8H18).Ch. 15.7 - Ethane (C2H6) is burned at atmospheric pressure...Ch. 15.7 - Reconsider Prob. 1550. What minimum pressure of...Ch. 15.7 - Calculate the HHV and LHV of gaseous n-octane fuel...Ch. 15.7 - Prob. 53PCh. 15.7 - Consider a complete combustion process during...Ch. 15.7 - Prob. 56PCh. 15.7 - Prob. 57PCh. 15.7 - Prob. 58PCh. 15.7 - Propane fuel (C3H8) is burned with an airfuel...Ch. 15.7 - Prob. 60PCh. 15.7 - Prob. 61PCh. 15.7 - Prob. 62PCh. 15.7 - Octane gas (C8H18) at 25C is burned steadily with...Ch. 15.7 - Liquid ethyl alcohol [C2H5OH(l)] at 25C is burned...Ch. 15.7 - Prob. 66PCh. 15.7 - A gaseous fuel mixture that is 40 percent propane...Ch. 15.7 - A constant-volume tank contains a mixture of 120 g...Ch. 15.7 - Prob. 70PCh. 15.7 - Prob. 71PCh. 15.7 - Prob. 72PCh. 15.7 - A fuel is completely burned first with the...Ch. 15.7 - Prob. 74PCh. 15.7 - Prob. 75PCh. 15.7 - What is the adiabatic flame temperature of methane...Ch. 15.7 - Octane gas (C8H18) at 25C is burned steadily with...Ch. 15.7 - Acetylene gas (C2H2) at 25C is burned during a...Ch. 15.7 - Ethyl alcohol [C2H5OH(g)] is burned with 200...Ch. 15.7 - Prob. 81PCh. 15.7 - Prob. 82PCh. 15.7 - Reconsider Prob. 1582. The combustion products are...Ch. 15.7 - Express the increase of entropy principle for...Ch. 15.7 - Prob. 85PCh. 15.7 - What does the Gibbs function of formation gf of a...Ch. 15.7 - Liquid octane (C8H18) enters a steady-flow...Ch. 15.7 - Prob. 88PCh. 15.7 - Reconsider Prob. 1588. The automobile engine is to...Ch. 15.7 - Benzene gas (C6H6) at 1 atm and 77F is burned...Ch. 15.7 - Prob. 91PCh. 15.7 - n-Octane [C8H18(l)] is burned in the...Ch. 15.7 - A steady-flow combustion chamber is supplied with...Ch. 15.7 - Prob. 94RPCh. 15.7 - Prob. 95RPCh. 15.7 - Prob. 96RPCh. 15.7 - Prob. 97RPCh. 15.7 - Prob. 98RPCh. 15.7 - Prob. 99RPCh. 15.7 - n-Butane (C4H10) is burned with the stoichiometric...Ch. 15.7 - A gaseous fuel mixture of 60 percent propane...Ch. 15.7 - Calculate the higher and lower heating values of...Ch. 15.7 - Prob. 103RPCh. 15.7 - Methane gas (CH4) at 25C is burned steadily with...Ch. 15.7 - A 6-m3 rigid tank initially contains a mixture of...Ch. 15.7 - Propane gas (C3H8) enters a steady-flow combustion...Ch. 15.7 - Determine the highest possible temperature that...Ch. 15.7 - Liquid propane [C3H8(l)] enters a combustion...Ch. 15.7 - Prob. 109RPCh. 15.7 - Prob. 110RPCh. 15.7 - Prob. 111RPCh. 15.7 - A steam boiler heats liquid water at 200C to...Ch. 15.7 - Repeat Prob. 15112 using a coal from Utah that has...Ch. 15.7 - Liquid octane (C8H18) enters a steady-flow...Ch. 15.7 - Prob. 115RPCh. 15.7 - Consider the combustion of a mixture of an...Ch. 15.7 - Prob. 117RPCh. 15.7 - A fuel is burned steadily in a combustion chamber....Ch. 15.7 - A fuel is burned with 70 percent theoretical air....Ch. 15.7 - Prob. 126FEPCh. 15.7 - One kmol of methane (CH4) is burned with an...Ch. 15.7 - The higher heating value of a hydrocarbon fuel...Ch. 15.7 - Acetylene gas (C2H2) is burned completely during a...Ch. 15.7 - An equimolar mixture of carbon dioxide and water...Ch. 15.7 - A fuel is burned during a steady-flow combustion...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Similar questions
- Quiz/An eccentrically loaded bracket is welded to the support as shown in Figure below. The load is static. The weld size for weld w1 is h1=6mm, for w2 h2 5mm, and for w3 is h3 -5.5 mm. Determine the safety factor (S.f) for the welds. F=22 kN. Use an AWS Electrode type (E90xx). 140 101.15 REDMI NOTE 8 PRO AI QUAD CAMERA Farrow_forward(read image)arrow_forwardProblem 3.30 A piston-cylinder device contains 0.85 kg of refrigerant- 134a at -10°C. The piston that is free to move has a mass of 12 kg and a diameter of 25 cm. The local atmospheric pressure is 100 kPa. Now, heat is transferred to refrigerant-134a until the temperature is 15°C. Determine (a) the final pressure, (b) the change in the volume of the refrigerant, and (c) the change in the enthalpy of the refrigerant-134a. please show Al work step by steparrow_forward
- Part 1 The storage tank contains lubricating oil of specific gravity 0.86 In one inclined side of the tank, there is a 0.48 m diameter circular inspection door, mounted on a horizontal shaft along the centre line of the gate. The oil level in the tank rests 8.8 m above the mounted shaft. (Please refer table 01 for relevant SG, D and h values). Describe the hydrostatic force and centre of pressure with the aid of a free body diagram of the inspection door. Calculate the magnitude of the hydrostatic force and locate the centre of pressure. 45° Estimate the moment that would have to be applied to the shaft to open the gate. Stop B If the oil level raised by 2 m from the current level, calculate the new moment required to open the gate. Figure 01arrow_forwardFrom thermodynamics please fill in the table show all work step by steparrow_forwardThe 150-lb skater passes point A with a speed of 6 ft/s. (Figure 1) Determine his speed when he reaches point B. Neglect friction. Determine the normal force exerted on him by the track at this point. 25 ft B = 4x A 20 ft xarrow_forward
- A virtual experiment is designed to determine the effect of friction on the timing and speed of packages being delivered to a conveyor belt and the normal force applied to the tube. A package is held and then let go at the edge of a circular shaped tube of radius R = 5m. The particle at the bottom will transfer to the conveyor belt, as shown below. Run the simulations for μ = 0, 0.1, 0.2, 0.3, 0.4, 0.5, 0.6 and determine the time and speed at which the package is delivered to the conveyor belt. In addition, determine the maximum normal force and its location along the path as measured by angle 0. Submit in hardcopy form: (0) Free Body Diagram, equations underneath, derivations (a) Your MATLAB mfile (b) A table listing the values in 5 columns: μ, T (time of transfer), V (speed of transfer), 0 (angle of max N), Nmax (max N) (c) Based on your results, explain in one sentence what you think will happen to the package if the friction is increased even further, e.g. μ = 0.8. NOTE: The ODE is…arrow_forwardPatm = 1 bar Piston m = 50 kg 5 g of Air T₁ = 600 K P₁ = 3 bar Stops A 9.75 x 10-3 m² FIGURE P3.88arrow_forwardAssume a Space Launch System (Figure 1(a)) that is approximated as a cantilever undamped single degree of freedom (SDOF) system with a mass at its free end (Figure 1(b)). The cantilever is assumed to be massless. Assume a wind load that is approximated with a concentrated harmonic forcing function p(t) = posin(ωt) acting on the mass. The known properties of the SDOF and the applied forcing function are given below. • Mass of SDOF: m =120 kip/g • Acceleration of gravity: g = 386 in/sec2 • Bending sectional stiffness of SDOF: EI = 1015 lbf×in2 • Height of SDOF: h = 2000 inches • Amplitude of forcing function: po = 6 kip • Forcing frequency: f = 8 Harrow_forward
- Assume a Space Launch System (Figure 1(a)) that is approximated as a cantilever undamped single degree of freedom (SDOF) system with a mass at its free end (Figure 1(b)). The cantilever is assumed to be massless. Assume a wind load that is approximated with a concentrated harmonic forcing function p(t) = posin(ωt) acting on the mass. The known properties of the SDOF and the applied forcing function are given below. • Mass of SDOF: m =120 kip/g • Acceleration of gravity: g = 386 in/sec2 • Bending sectional stiffness of SDOF: EI = 1015 lbf×in2 • Height of SDOF: h = 2000 inches • Amplitude of forcing function: po = 6 kip • Forcing frequency: f = 8 Hz Figure 1: Single-degree-of-freedom system in Problem 1. Please compute the following considering the steady-state response of the SDOF system. Do not consider the transient response unless it is explicitly stated in the question. (a) The natural circular frequency and the natural period of the SDOF. (10 points) (b) The maximum displacement of…arrow_forwardAssume a Space Launch System (Figure 1(a)) that is approximated as a cantilever undamped single degree of freedom (SDOF) system with a mass at its free end (Figure 1(b)). The cantilever is assumed to be massless. Assume a wind load that is approximated with a concentrated harmonic forcing function p(t) = posin(ωt) acting on the mass. The known properties of the SDOF and the applied forcing function are given below. • Mass of SDOF: m =120 kip/g • Acceleration of gravity: g = 386 in/sec2 • Bending sectional stiffness of SDOF: EI = 1015 lbf×in2 • Height of SDOF: h = 2000 inches • Amplitude of forcing function: po = 6 kip • Forcing frequency: f = 8 Hz Figure 1: Single-degree-of-freedom system in Problem 1. Please compute the following considering the steady-state response of the SDOF system. Do not consider the transient response unless it is explicitly stated in the question. (a) The natural circular frequency and the natural period of the SDOF. (10 points) (b) The maximum displacement of…arrow_forwardPlease solve 13 * √(2675.16)² + (63.72 + 2255,03)² = 175x106 can you explain the process for getting d seperate thank youarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY

Elements Of Electromagnetics
Mechanical Engineering
ISBN:9780190698614
Author:Sadiku, Matthew N. O.
Publisher:Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:9780134319650
Author:Russell C. Hibbeler
Publisher:PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:9781259822674
Author:Yunus A. Cengel Dr., Michael A. Boles
Publisher:McGraw-Hill Education

Control Systems Engineering
Mechanical Engineering
ISBN:9781118170519
Author:Norman S. Nise
Publisher:WILEY

Mechanics of Materials (MindTap Course List)
Mechanical Engineering
ISBN:9781337093347
Author:Barry J. Goodno, James M. Gere
Publisher:Cengage Learning

Engineering Mechanics: Statics
Mechanical Engineering
ISBN:9781118807330
Author:James L. Meriam, L. G. Kraige, J. N. Bolton
Publisher:WILEY
Extent of Reaction; Author: LearnChemE;https://www.youtube.com/watch?v=__stMf3OLP4;License: Standard Youtube License