
THERMODYNAMICS (LL)-W/ACCESS >CUSTOM<
9th Edition
ISBN: 9781266657610
Author: CENGEL
Publisher: MCG CUSTOM
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 15.7, Problem 116RP
Consider the combustion of a mixture of an alcohol, CnHmOx, and a hydrocarbon fuel, CwHz, with excess theoretical air and incomplete combustion according to the chemical reaction as follows:
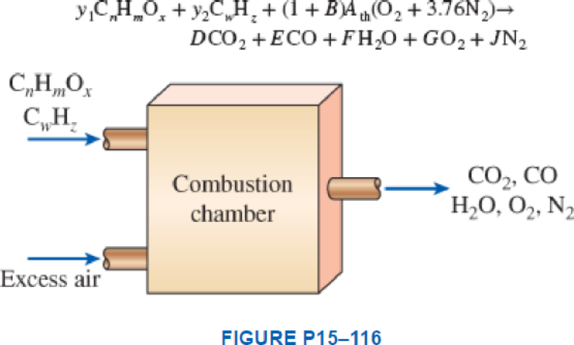
where y1 and y2 are the mole fractions of the fuel mixture, Ath is the theoretical O2 required for this fuel, and B is the excess amount of air in decimal form. If a is the fraction of carbon in the fuel converted to carbon dioxide and b is the remaining fraction converted to carbon monoxide, determine the coefficients Ath, D, E, F, G, and J for a fixed B amount of excess air. Write the coefficients D, E, F, G, and J as functions of y1, y2, n, m, x, w, z, a, b, B, and Ath in the simplest correct forms.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The shaft shown in the figure below is subjected to axial loads as illustrated. The diameters of segments AB, BC, and CD are 20mm, 25mm, and 15mm, respectively. If the modulus of elasticity of the material is 610 MPa. Determine the change of A to D length
Determine the final pressure and temperature.
The final pressure is kPa.
The final temperature is ºC.
Air enters the 1-m2 inlet of an aircraft engine at 100 kPa and 20°C with a velocity of 184 m/s. Determine the volume flow rate, in m3/s, at the engine’s inlet and the mass flow rate, in kg/s, at the engine’s exit. The gas constant of air is R = 0.287 kPa·m3/kg·K.
The volume flow rate at the engine’s inlet m3/s.
The mass flow rate at the engine’s exit is kg/s.
Chapter 15 Solutions
THERMODYNAMICS (LL)-W/ACCESS >CUSTOM<
Ch. 15.7 - What are the approximate chemical compositions of...Ch. 15.7 - How does the presence of N2 in air affect the...Ch. 15.7 - Prob. 3PCh. 15.7 - Prob. 4PCh. 15.7 - Is the airfuel ratio expressed on a mole basis...Ch. 15.7 - How does the presence of moisture in air affect...Ch. 15.7 - Prob. 7PCh. 15.7 - Prob. 8PCh. 15.7 - Prob. 9PCh. 15.7 - Are complete combustion and theoretical combustion...
Ch. 15.7 - What does 100 percent theoretical air represent?Ch. 15.7 - Consider a fuel that is burned with (a) 130...Ch. 15.7 - What are the causes of incomplete combustion?Ch. 15.7 - Which is more likely to be found in the products...Ch. 15.7 - Methane (CH4) is burned with the stoichiometric...Ch. 15.7 - Prob. 16PCh. 15.7 - n-Butane fuel (C4H10) is burned with the...Ch. 15.7 - Prob. 18PCh. 15.7 - Propane (C3H8) is burned with 75 percent excess...Ch. 15.7 - Propane fuel (C3H8) is burned with 30 percent...Ch. 15.7 - In a combustion chamber, ethane (C2H6) is burned...Ch. 15.7 - Prob. 22PCh. 15.7 - Prob. 23PCh. 15.7 - Ethane (C2H6) is burned with 20 percent excess air...Ch. 15.7 - Octane (C8H18) is burned with 250 percent...Ch. 15.7 - Prob. 26PCh. 15.7 - A fuel mixture of 60 percent by mass methane (CH4)...Ch. 15.7 - Prob. 28PCh. 15.7 - A certain natural gas has the following volumetric...Ch. 15.7 - Prob. 30PCh. 15.7 - A gaseous fuel with a volumetric analysis of 45...Ch. 15.7 - Prob. 33PCh. 15.7 - The fuel mixer in a natural gas burner mixes...Ch. 15.7 - Prob. 35PCh. 15.7 - Prob. 36PCh. 15.7 - Determine the fuelair ratio when coal from...Ch. 15.7 - Prob. 38PCh. 15.7 - Prob. 39PCh. 15.7 - Prob. 40PCh. 15.7 - Prob. 41PCh. 15.7 - When are the enthalpy of formation and the...Ch. 15.7 - Prob. 43PCh. 15.7 - Prob. 44PCh. 15.7 - Prob. 45PCh. 15.7 - Prob. 46PCh. 15.7 - Prob. 48PCh. 15.7 - Repeat Prob. 1546 for liquid octane (C8H18).Ch. 15.7 - Ethane (C2H6) is burned at atmospheric pressure...Ch. 15.7 - Reconsider Prob. 1550. What minimum pressure of...Ch. 15.7 - Calculate the HHV and LHV of gaseous n-octane fuel...Ch. 15.7 - Prob. 53PCh. 15.7 - Consider a complete combustion process during...Ch. 15.7 - Prob. 56PCh. 15.7 - Prob. 57PCh. 15.7 - Prob. 58PCh. 15.7 - Propane fuel (C3H8) is burned with an airfuel...Ch. 15.7 - Prob. 60PCh. 15.7 - Prob. 61PCh. 15.7 - Prob. 62PCh. 15.7 - Octane gas (C8H18) at 25C is burned steadily with...Ch. 15.7 - Liquid ethyl alcohol [C2H5OH(l)] at 25C is burned...Ch. 15.7 - Prob. 66PCh. 15.7 - A gaseous fuel mixture that is 40 percent propane...Ch. 15.7 - A constant-volume tank contains a mixture of 120 g...Ch. 15.7 - Prob. 70PCh. 15.7 - Prob. 71PCh. 15.7 - Prob. 72PCh. 15.7 - A fuel is completely burned first with the...Ch. 15.7 - Prob. 74PCh. 15.7 - Prob. 75PCh. 15.7 - What is the adiabatic flame temperature of methane...Ch. 15.7 - Octane gas (C8H18) at 25C is burned steadily with...Ch. 15.7 - Acetylene gas (C2H2) at 25C is burned during a...Ch. 15.7 - Ethyl alcohol [C2H5OH(g)] is burned with 200...Ch. 15.7 - Prob. 81PCh. 15.7 - Prob. 82PCh. 15.7 - Reconsider Prob. 1582. The combustion products are...Ch. 15.7 - Express the increase of entropy principle for...Ch. 15.7 - Prob. 85PCh. 15.7 - What does the Gibbs function of formation gf of a...Ch. 15.7 - Liquid octane (C8H18) enters a steady-flow...Ch. 15.7 - Prob. 88PCh. 15.7 - Reconsider Prob. 1588. The automobile engine is to...Ch. 15.7 - Benzene gas (C6H6) at 1 atm and 77F is burned...Ch. 15.7 - Prob. 91PCh. 15.7 - n-Octane [C8H18(l)] is burned in the...Ch. 15.7 - A steady-flow combustion chamber is supplied with...Ch. 15.7 - Prob. 94RPCh. 15.7 - Prob. 95RPCh. 15.7 - Prob. 96RPCh. 15.7 - Prob. 97RPCh. 15.7 - Prob. 98RPCh. 15.7 - Prob. 99RPCh. 15.7 - n-Butane (C4H10) is burned with the stoichiometric...Ch. 15.7 - A gaseous fuel mixture of 60 percent propane...Ch. 15.7 - Calculate the higher and lower heating values of...Ch. 15.7 - Prob. 103RPCh. 15.7 - Methane gas (CH4) at 25C is burned steadily with...Ch. 15.7 - A 6-m3 rigid tank initially contains a mixture of...Ch. 15.7 - Propane gas (C3H8) enters a steady-flow combustion...Ch. 15.7 - Determine the highest possible temperature that...Ch. 15.7 - Liquid propane [C3H8(l)] enters a combustion...Ch. 15.7 - Prob. 109RPCh. 15.7 - Prob. 110RPCh. 15.7 - Prob. 111RPCh. 15.7 - A steam boiler heats liquid water at 200C to...Ch. 15.7 - Repeat Prob. 15112 using a coal from Utah that has...Ch. 15.7 - Liquid octane (C8H18) enters a steady-flow...Ch. 15.7 - Prob. 115RPCh. 15.7 - Consider the combustion of a mixture of an...Ch. 15.7 - Prob. 117RPCh. 15.7 - A fuel is burned steadily in a combustion chamber....Ch. 15.7 - A fuel is burned with 70 percent theoretical air....Ch. 15.7 - Prob. 126FEPCh. 15.7 - One kmol of methane (CH4) is burned with an...Ch. 15.7 - The higher heating value of a hydrocarbon fuel...Ch. 15.7 - Acetylene gas (C2H2) is burned completely during a...Ch. 15.7 - An equimolar mixture of carbon dioxide and water...Ch. 15.7 - A fuel is burned during a steady-flow combustion...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Similar questions
- The ventilating fan of the bathroom of a building has a volume flow rate of 33 L/s and runs continuously. If the density of air inside is 1.20 kg/m3, determine the mass of air vented out in one day. The mass of air is kg.arrow_forwardA steady-flow compressor is used to compress helium from 15 psia and 70°F at the inlet to 200 psia and 600°F at the outlet. The outlet area and velocity are 0.01 ft2 and 100 ft/s, respectively, and the inlet velocity is 53 ft/s. Determine the mass flow rate and the inlet area. The gas constant of helium is R = 2.6809 psia·ft3/lbm·R. The mass flow rate is lbm/s. The inlet area is ft2.arrow_forward1. The maximum and minimum stresses as well as the shear stress seen subjected the piece in plane A-A. Assume it is a cylinder with a diameter of 12.7mm 2. Draw the Mohr circle for the stress state using software. 3. Selection of the material for the prosthesis, which must be analyzed from the point of safety and cost view.arrow_forward
- 1. The maximum and minimum stresses as well as the shear stress seen subjected the piece in plane A-A. Assume it is a cylinder with a diameter of 12.7mm 2. Draw the Mohr circle for the stress state using software. 3. Selection of the material for the prosthesis, which must be analyzed from the point of safety and cost view.arrow_forwardFirst, define the coordinate system XY with its origin at O2 and X-axis passing through O4 asshown above, then based on the provided steps Perform coordinate transformation from XY to xy to get the trajectory of point P. Show all the steps and calcualtionsarrow_forwardI don't know how to solve thisarrow_forward
- Question 2 (40 Points) Consider the following double pendulum-like system with links ₁ and 12. The angles 0 and & could have angular velocities ėêk and êk, respectively, where ②k is a unit vector that points out of the page and is perpendicular to x and y. They could also have angular accelerations Ök and êk. The angle is defined relative to the angle 0. The link 12 is a spring and can extend or compress at a rate of 12. It can also have a rate of extension or compression Ï2. li y êr1 êe 12 χ 3 еф er2 ده لج 1) Express the velocity of the mass in terms of the unit vectors ê0, êr1, êø, and êr2, and any extension/contraction of the links (e.g.,. i; and Ï¿) (12 Points) 2) Express the acceleration of the mass in terms of the unit vectors ê¤, ê×1, êp, and êÃ2, and any extension/contraction of the links (e.g.,. İ; and Ï¿) (12 Points) 3) Express the velocity of the mass in terms of unit vectors î and ĵ that point in the x and y directions, respectively. Also include the appropriate,…arrow_forwardprovide step by step solutions for angles teta 3 and teta 4 by the vector loopmethod. Show work in: vector loop, vector equations, solution procedure.arrow_forward(Manometer) A tank is constructed of a series of cylinders having diameters of 0.35, 0.30, and 0.20 m as shown in the figure below. The tank contains oil, water, and glycerin and a mercury manometer is attached to the bottom as illustrated. Calculate the manometer reading, h. 0.11 m + SAE 30 Oil 0.13 m + Water 0.10 m Glycerin + 0.10 m Mercury h = marrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY

Elements Of Electromagnetics
Mechanical Engineering
ISBN:9780190698614
Author:Sadiku, Matthew N. O.
Publisher:Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:9780134319650
Author:Russell C. Hibbeler
Publisher:PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:9781259822674
Author:Yunus A. Cengel Dr., Michael A. Boles
Publisher:McGraw-Hill Education

Control Systems Engineering
Mechanical Engineering
ISBN:9781118170519
Author:Norman S. Nise
Publisher:WILEY

Mechanics of Materials (MindTap Course List)
Mechanical Engineering
ISBN:9781337093347
Author:Barry J. Goodno, James M. Gere
Publisher:Cengage Learning

Engineering Mechanics: Statics
Mechanical Engineering
ISBN:9781118807330
Author:James L. Meriam, L. G. Kraige, J. N. Bolton
Publisher:WILEY
Extent of Reaction; Author: LearnChemE;https://www.youtube.com/watch?v=__stMf3OLP4;License: Standard Youtube License