EP INTRO.TO GENERAL,ORGANIC...-OWL ACCE
12th Edition
ISBN: 9781337915984
Author: Bettelheim
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 15, Problem 51P
Interpretation Introduction
Interpretation:
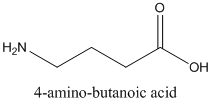
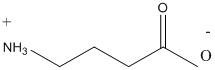
Following are the two structural formulas for 4-amino-butanoic acid, a neurotransmitter. The better structural formula for the above molecule is (A) or (B).
Concept introduction:
The spatial arrangement of atom within a compound that are joined together is identified with the help of structural formula.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
ΗΝ,
Draw Final Product
C
cyclohexanone
pH 4-5
Edit Enamine
H3O+
CH3CH2Br
THF, reflux
H
Edit Iminium Ion
How many hydrogen atoms are connected to the indicated carbon atom?
Identify the compound with the longest carbon - nitrogen bond.
O CH3CH2CH=NH
O CH3CH2NH2
CH3CH2C=N
CH3CH=NCH 3
The length of all the carbon-nitrogen bonds are the same
Chapter 15 Solutions
EP INTRO.TO GENERAL,ORGANIC...-OWL ACCE
Ch. 15.1 - Prob. 15.1QCCh. 15.2 - Problem 16-2 Write a structural formula for each...Ch. 15.2 - Prob. 15.3QCCh. 15.3 - Problem 16-4 Select the stronger base from each...Ch. 15.3 - Prob. 15.5QCCh. 15 - 16-6 Answer true or false. te/7-Butylamine is a 3°...Ch. 15 - Prob. 2PCh. 15 - Prob. 3PCh. 15 - 16-9 In what way are pyridine and pyrimidine...Ch. 15 - Prob. 5P
Ch. 15 - Prob. 6PCh. 15 - Prob. 7PCh. 15 - 16-13 Classify each amino group as primary,...Ch. 15 - Prob. 9PCh. 15 - 16-15 There are eight primary amines with the...Ch. 15 - Prob. 11PCh. 15 - 16-17 Propylamine (bp 48°C), ethylmethylamine (bp...Ch. 15 - 16-18 Account for the fact that 1-butanamine (bp...Ch. 15 - 16-19 2-Me thy 1 propane (bp -12°C), 2-propanol...Ch. 15 - Prob. 15PCh. 15 - Prob. 16PCh. 15 - Prob. 17PCh. 15 - Prob. 18PCh. 15 - Prob. 19PCh. 15 - Prob. 20PCh. 15 - 16-26 The p/fb of amphetamine is approximately 3.2...Ch. 15 - 16-27 Guanidine, p/Ca 13.6, is a very strong base,...Ch. 15 - 16-28 Following is the structural formula of...Ch. 15 - Prob. 24PCh. 15 - Prob. 25PCh. 15 - Prob. 26PCh. 15 - 16*32 Many tumors of the breast are correlated...Ch. 15 - Prob. 28PCh. 15 - Prob. 29PCh. 15 - Prob. 30PCh. 15 - (Chemical Connections 15B ) Identify all...Ch. 15 - Prob. 32PCh. 15 - Prob. 33PCh. 15 - Prob. 34PCh. 15 - Prob. 35PCh. 15 - Prob. 36PCh. 15 - (Chemical Connections 15D ) Suppose you saw this...Ch. 15 - Prob. 38PCh. 15 - Prob. 39PCh. 15 - Prob. 40PCh. 15 - 16-46 Arrange these three compounds in order of...Ch. 15 - Prob. 42PCh. 15 - Prob. 43PCh. 15 - Prob. 44PCh. 15 - Prob. 45PCh. 15 - Prob. 46PCh. 15 - Prob. 47PCh. 15 - Prob. 48PCh. 15 - 16-54 Several poisonous plants, including Atropa...Ch. 15 - Prob. 50PCh. 15 - Prob. 51PCh. 15 - Prob. 52PCh. 15 - 16-58 Following is a structural formula of...Ch. 15 - Prob. 54P
Knowledge Booster
Similar questions
- Identify any polar covalent bonds in epichlorohydrin with S+ and 8- symbols in the appropriate locations. Choose the correct answer below. Η H's+ 6Η Η Η Η Η Ηδ Η Ο Ο HH +Η Η +Η Η Η -8+ CIarrow_forwardH H:O::::H H H HH H::O:D:D:H HH HH H:O:D:D:H .. HH H:O:D:D:H H H Select the correct Lewis dot structure for the following compound: CH3CH2OHarrow_forwardRank the following compounds in order of decreasing boiling point. ннннн -С-С-Н . н-с- ННННН H ΗΤΗ НННН TTTĪ н-с-с-с-с-о-н НННН НН C' Н н-с-с-с-с-н НН || Ш НННН H-C-C-C-C-N-H ННННН IVarrow_forward
- Rank the following compounds in order of decreasing dipole moment. |>||>||| ||>|||>| |>|||>|| |||>||>| O ||>>||| H F H F H c=c || H c=c F F IIIarrow_forwardchoose the description that best describes the geometry for the following charged species ch3-arrow_forwardWhy isn't the ketone in this compound converted to an acetal or hemiacetal by the alcohol and acid?arrow_forward
- What is the approximate bond angle around the nitrogen atom? HNH H Harrow_forwardOH 1. NaOCH2CH3 Q 2. CH3CH2Br (1 equiv) H3O+ Select to Draw 1. NaOCH2 CH3 2. CH3Br (1 equiv) heat Select to Edit Select to Drawarrow_forwardComplete and balance the following half-reaction in acidic solution. Be sure to include the proper phases for all species within the reaction. S₂O₃²⁻(aq) → S₄O₆²⁻(aq)arrow_forward
- Q Select to Edit NH3 (CH3)2CHCI (1 equiv) AICI 3 Select to Draw cat. H2SO4 SO3 (1 equiv) HO SOCl2 pyridine Select to Edit >arrow_forwardComplete and balance the following half-reaction in basic solution. Be sure to include the proper phases for all species within the reaction. Zn(s) → Zn(OH)₄²⁻(aq)arrow_forwardb. ὋΗ CH3CH2OH H2SO4arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co