
Concept explainers
(a)
Interpretation:
The correct number of hydrogens has to be added for the possible skeletons for a six C compound with a five C chain and two double bonds.
Concept Introduction:
The hydrocarbon is said to be a carbon atom bonded with hydrogen. The carbon atom contains four bonds. The bonds may be four single bonds or two single bonds and one double bond or one single bond and one double bond.
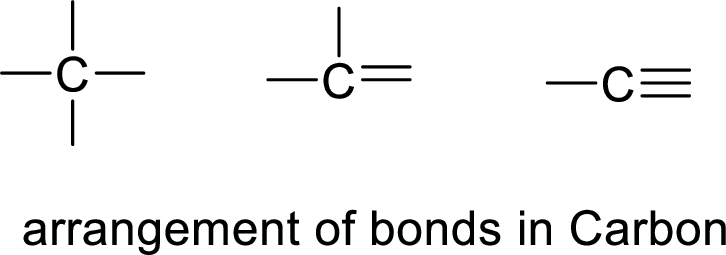
If the straight chain is branched, we get one type of hydrocarbon. If double bond is present, it restricts the rotation of the bond and forms cis and trans hydrocarbons.
(b)
Interpretation:
The correct number of hydrogens has to be added for the possible skeletons for a six C compound with a five C chain and one triple bond.
Concept Introduction:
The hydrocarbon is said to be a carbon atom bonded with hydrogen. The carbon atom contains four bonds. The bonds may be four single bonds or two single bonds and one double bond or one single bond and one double bond.
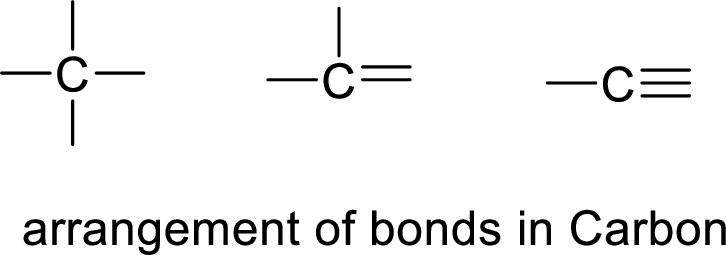
If the straight chain is branched, we get one type of hydrocarbon. If double bond is present, it restricts the rotation of the bond and forms cis and trans hydrocarbons.
(c)
Interpretation:
The correct number of hydrogens has to be added for the possible skeletons for a six C compound with a four C ring and no double bonds.
Concept Introduction:
The hydrocarbon is said to be a carbon atom bonded with hydrogen. The carbon atom contains four bonds. The bonds may be four single bonds or two single bonds and one double bond or one single bond and one double bond.
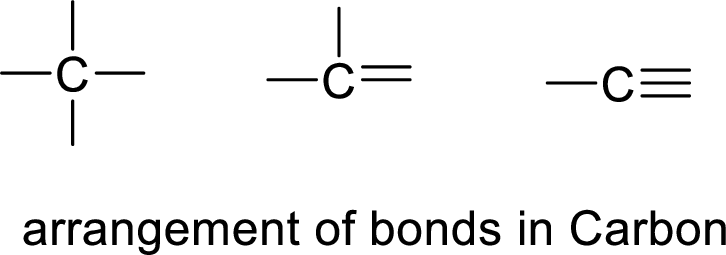
If the straight chain is branched, we get one type of hydrocarbon. If double bond is present, it restricts the rotation of the bond and forms cis and trans- hydrocarbons.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
CHEMISTRY:MOLECULAR...V.2 W/ACCESS
- Write the calculate the reaction quotient for the following system, if the partial pressure of all reactantsand products is 0.15 atm: NOCl (g) ⇌ NO (g) + Cl2 (g) H = 20.5 kcalarrow_forwardComplete the spectroscopy with structurearrow_forwardcould you answer the questions and draw the complete mechanismarrow_forward
- Complete the spectroscopy with structurearrow_forwardCalculate the reaction quotient for the reaction:NaOH (s) ⇌ Na+ (aq)+ OH- (aq) + 44.4 kJ [Na+] = 4.22 M [OH-] = 6.41 Marrow_forwardGiven the following concentrations for a system, calculate the value for the reaction quotient: Cl2(g)+ CS2(g) ⇌ CCl4(g)+ S2Cl2(g) Cl2 = 31.1 atm CS2 = 91.2 atm CCl4 = 2.12 atm S2Cl2 = 10.4 atmarrow_forward
- Match each chemical or item with the proper disposal or cleanup mwthod, Not all disposal and cleanup methods will be labeled. Metal sheets C, calcium, choroide solutions part A, damp metal pieces Part B, volumetric flask part A. a.Return to correct lables”drying out breaker. Place used items in the drawer.: Rinse with deionized water, dry as best you can, return to instructor. Return used material to the instructor.: Pour down the sink with planty of running water.: f.Pour into aqueous waste container. g.Places used items in garbage.arrow_forwardWrite the equilibrium constant expression for the following reaction: HNO2(aq) + H2O(l) ⇌ H3O+(aq) + NO2-(aq)arrow_forwardWrite the reaction quotient for: Pb2+(aq) + 2 Cl- (aq) ⇌ PbCl2(s)arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





