
Interpretation: The meaning of different portion in the cooling curve of water has to be discussed.
Concept introduction: Heating curves represents the change in temperature as a substance is heated. Cooling curves represents the change in temperature as a substance is cooled. Heating and cooling curves have horizontal flat parts that shows the phases changes.
Answer to Problem 15A
The phase changes are given by the flat portions of the curve whereas, the slope represents the changes in temperature.
Explanation of Solution
The heating-cooling curve of water is,
The x-axis represents the amount of heat added at constant rate and the temperature in
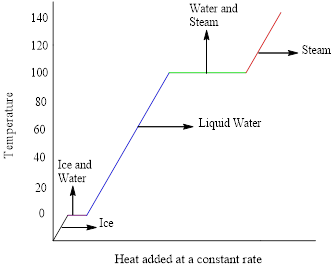
Figure 1: Heating-cooling curve of water
The heating curve (addition of energy) goes from left to right in the above graph and the cooling curve (removal of energy) goes from right to left.
The flat portions of the curve represent the changes in phases and the slope of the curve represents the changes in temperature.
The changes in phase are given by the flat portions of the curve whereas, the slope represents the changes in temperature.
Chapter 14 Solutions
World of Chemistry
- Extra for Experts: Your Future in Chemistry. As you now know, there are countless jobs that involve chemistry! Research a chemistry profession that interests you. In your answer, discuss which aspects of the job most appeal to you.arrow_forwardMISSED THIS? Read Section 19.9 (Pages 878-881); Watch IWE 19.10 Consider the following reaction: CH3OH(g) CO(g) + 2H2(g) (Note that AG,CH3OH(g) = -162.3 kJ/mol and AG,co(g)=-137.2 kJ/mol.) Part A Calculate AG for this reaction at 25 °C under the following conditions: PCH₂OH Pco PH2 0.815 atm = 0.140 atm 0.170 atm Express your answer in kilojoules to three significant figures. Ο ΑΣΦ AG = -150 Submit Previous Answers Request Answer □? kJ × Incorrect; Try Again; 2 attempts remaining Calculate the free energy change under nonstandard conditions (AGrxn) by using the following relationship: AGrxn = AGrxn + RTInQ, AGxn+RTInQ, where AGxn is the standard free energy change, R is the ideal gas constant, T is the temperature in kelvins, a is the reaction quotient. Provide Feedback Next >arrow_forwardIdentify and provide a brief explanation of Gas Chromatography (GC) within the context of chemical analysis of food. Incorporate the specific application name, provide a concise overview of sample preparation methods, outline instrumental parameters and conditions ultilized, and summarise the outcomes and findings achieved through this analytical approach.arrow_forward
- Identify and provide a concise explanation of the concept of signal-to-noise ratio (SNR) in the context of chemical analysis. Provide specific examples.arrow_forwardIdentify and provide a concise explanation of a specific analytical instrument capable of detecting and quantifying trace compounds in food samples. Emphasise the instrumental capabilities relevant to trace compound analysis in the nominated food. Include the specific application name (eg: identification and quantification of mercury in salmon), outline a brief description of sample preparation procedures, and provide a summary of the obtained results from the analytical process.arrow_forwardIdentify and provide an explanation of what 'Seperation Science' is. Also describe its importance with the respect to the chemical analysis of food. Provide specific examples.arrow_forward
- 5. Propose a Synthesis for the molecule below. You may use any starting materials containing 6 carbons or less (reagents that aren't incorporated into the final molecule such as PhзP do not count towards this total, and the starting material can have whatever non-carbon functional groups you want), and any of the reactions you have learned so far in organic chemistry I, II, and III. Your final answer should show each step separately, with intermediates and conditions clearly drawn. H3C CH3arrow_forwardState the name and condensed formula of isooxazole obtained by reacting acetylacetone and hydroxylamine.arrow_forwardState the name and condensed formula of the isothiazole obtained by reacting acetylacetone and thiosemicarbazide.arrow_forward
- Provide the semi-developed formula of isooxazole obtained by reacting acetylacetone and hydroxylamine.arrow_forwardGiven a 1,3-dicarbonyl compound (R1-CO-CH2-CO-R2), indicate the formula of the compound obtaineda) if I add hydroxylamine (NH2OH) to give an isooxazole.b) if I add thiosemicarbazide (NH2-CO-NH-NH2) to give an isothiazole.arrow_forwardAn orange laser has a wavelength of 610 nm. What is the energy of this light?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





