
The Volcanic Explosivity Index (VEI) is based primarily on the amount of material ejected from a volcano, although other factors play a role as well, such as height of plume in the atmosphere. The following table shows the number of volcanic eruptions (N) over the past 10,000 years having a VEI of between 2 and 7.
There are also VEI values of 0, 1, and 8. There is a level 0 volcano erupting somewhere on the Earth essentially all the time. There are one or more level 1 volcanoes essentially every day. The last known level 8 volcano was about 26 000 years ago
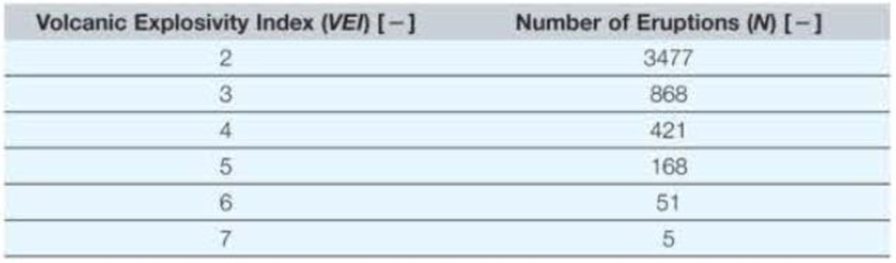
- a. Show the resulting data trendline, with equation and R2 value, on the appropriate graph type (xy scatter, semilog, or log–log) to make the data appear linear
- b. How many level 1 volcanoes does the model predict should have occurred in the last 10,000 years?
- c. How many level 8 volcanoes does the model predict should have occurred in the last 10,000 years?
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Thinking Like an Engineer: An Active Learning Approach (3rd Edition)
Additional Engineering Textbook Solutions
Engineering Mechanics: Statics & Dynamics (14th Edition)
INTERNATIONAL EDITION---Engineering Mechanics: Statics, 14th edition (SI unit)
Automotive Technology: Principles, Diagnosis, and Service (5th Edition)
Applied Fluid Mechanics (7th Edition)
Automotive Technology: Principles, Diagnosis, And Service (6th Edition) (halderman Automotive Series)
Applied Statics and Strength of Materials (6th Edition)
- The table below shows the melting point, specific heat, and heat of fusion of different solids. Suppose the same mass of these solids are at their respective melting point temperatures, which solid requires most amount of heat to melt? Substance Melting Point Heat of Fusion Specific Heat 79.71 cal/g 5.85 cal/g 21.07 cal/g 15.39 cal/g 31.98 cal/g 0.50 cal/gC° 0.033 cal/gC° 0.056 cal/gC° 0.030 cal/gCo 0.093 cal/gC° Ice 0°C Lead 327°C Silver 961°C Gold 1063°C Copper 1083°C O lead ice O gold O silver O O O Oarrow_forwardConsider a process to prepare a metal for a certain application. There are five parameters that must be considered: temperature, quenching rate, cooling time, carbon content, CO₂ concentration. It is desired to determine which of these parameters has the most influence on the process. There are two levels for each parameter as shown below. Temperature (°C) Quenching Rate(°C/s) Cooling time (s) Carbon Content (wt% C) CO₂ Concentration (%) Eight experiments were defined as follows: Experiment Carbon Content (wt% C) 1 2 3 4 5 6 7 8 1) What size orthogonal array should be used for evaluation (assume noise is negligible)? 2) Generate the array with the level values (i.e. Level 1 and Level 2). 1 1 1 1 6 6 6 6 Quenching Rate (°C/s) 35 35 140 140 35 35 140 140 Four trials were run for the experiments defined above: Experiment 1 2 3 4 5 6 7 8 Level 1 760 35 1 1 5 T1 68.00 69.84 74.36 71.71 91.27 54.39 64.65 60.31 Cooling Time 1 1 300 300 300 300 1 1 T2 61.41 64.76 61.30 58.42 90.89 Level 2 900…arrow_forwardIn measuring the surface tension of a liquid (drop weight method), 20 drops of the liquid (r = 0.2cm) falling apart from the tip whose diameter is 0.4 cm were found to weight 0.95 gram. What is the surface tension of the liquid?arrow_forward
- Please explain and answerarrow_forwardIn a power plant operating with water and steam, the following measurements and observations were done at various stages along the system. the pressure is 150 kPa and the enthalpy, h is 1580.115 (kJ/kg) For this case, the Temperature, in oC, is most likely: 250 105.97 111.35 130 116.04arrow_forward9arrow_forward
- Draw a figure or FBD that will support the problem. Explain each step by step formula.arrow_forwardHypothesis: EF=ma , f=MkfN Question: Make a judgment about whether or not your hypothesis (I used EF=ma , f=MkfN) can be rejected or if it is supported based on this experiment. Justify your judgment with evidence from this experiment. What assumptions did you make? Help me with this question pleasearrow_forwardTHERMODYNAMICS UPVOTE WILL BE GIVEN. PLEASE WRITE THE COMPLETE SOLUTIONS LEGIBLY. SHOW THE STEP-BY-STEP PROCESS WITH SHORT COMMENT/EXPLANATION. ANSWER IN 3 DECIMAL PLACES. Note: Use values of MW, Cp, Cv, etc., from the Tables provided below.arrow_forward
- A 30 wt% Ni-70 wt% Cu alloy (Animated Figure 9.3a) is slowly cooled from 1400°C (2550°F) to 1150°C (2100°F). (a) At what temperature does the first solid phase form? i ! °C (b) What is the composition of this solid phase? i %wt Ni (c) At what temperature does the last of the liquid solidify? i °℃ (d) What is the composition of this last remaining liquid phase? i %wt Niarrow_forwardb) The variation in the experimental density of water, p, with temperature T, in the range of 20°Carrow_forwardSelect two saturated and two unsaturated samples of air from the dataset of pressure and temperature given below: Pressure (mb): 10, 20, 30 Temperature (°C): 10, 20, 30 Let A and B be two air samples, where A: (T=30°C, P=25 mb) and B: (T=30°C, P=30 mb). For each sample, determine the following: a. Saturation vapor pressure b. Dew point c. Relative humidity d. If samples A and B were cooled to 15°C. What would be their relative humidity? What would be their dew point temperature?arrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





