
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1.3, Problem 1.11P
Characterize each of the following structures as aromatic, nonaromatic, or antiaromatic:
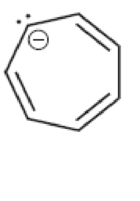
Answer: _____
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
determine if the compounds are aromatic. are my answers correct?
In the blank for each molecule, enter A, B or C according to the code below:
A = aromatic if planar
Banti-aromatic if planar
C=non-aromatic (does not matter if it is planar or not because other conditions for aromaticity or anti-
aromaticity are not met)
$0 $
blank 1 blank 2
blank 3
blank 4
blank 5
m
blank 6
blank 7
blank 8
blank 9
5
Chapter 1 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 1.2 - Prob. 1.2PCh. 1.2 - Prob. 1.3PCh. 1.2 - Prob. 1.4PCh. 1.2 - Prob. 1.5PCh. 1.2 - Prob. 1.6PCh. 1.3 - Characterize each of the following structures as...Ch. 1.3 - Characterize each of the following structures as...Ch. 1.3 - Characterize each of the following structures as...Ch. 1.3 - Characterize each of the following structures as...Ch. 1.3 - Characterize each of the following structures as...
Additional Science Textbook Solutions
Find more solutions based on key concepts
How many electrons are in each energy level of the following elements? a. H b. F c. Ar d. K
General, Organic, and Biological Chemistry (3rd Edition)
You prepare a buffer solution by dissolving 2.00 g each of benzoic acid C2H2COOH, and sodium benzoate, NaC8H8CO...
General Chemistry: Principles and Modern Applications (11th Edition)
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Refrigerant- 134a is being transported a 0.1 kg/s through a Teflon tube of inside diameter D0=28 mm and outside...
Fundamentals of Heat and Mass Transfer
Q1. Which wavelength of light has the highest frequency?
a) 10 nm
b) 10 mm
c) 1 nm
d) 1 mm
Chemistry: A Molecular Approach (4th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Aromatic molecules must be Aromatic Rules: суclic planar fully conjugated contain 4n + 2n electrons Answer Bank contain an odd number of pairs of a electrons асyclic contain 4n a electrons contain an even number of pairs of a electrons non-planar Incorrectarrow_forwardClassify the following compounds as aromatic, antiaromatic, or nonaromatic.arrow_forwardanswer all questionsarrow_forward
- 5) Indicate whether the following charged compounds are aromatic (A), anti-aromatic (AA) or non-aromatic (NA). A B C CH₂ Aromatic?arrow_forward10. Identify the following as aromatic, non-aromatic, or anti-aromatic. List answers below. N'arrow_forwardClassify each of the following compounds as aromatic or non-aromatic. SH D C Structure A V[Choose ] Aromatic Non-aromatic Structure B Choose] Structure C [Choose ] Structure D [ Choose ) Structure E [ Choose] Structure F Choose1 B.arrow_forward
- Aromaticity and Properties Identify the properties that describe aromatic compounds. Aromatic Answer Bank planar all single bonds flexible rigid twisted alternating double and single bonds electrons fully delocalized in a ring all bond lengths between double and single bond lengths all double bonds reactive unpaired electrons very unstable very stablearrow_forwardÔ https://app.101edu.co Question 3 of 24 A substance which is cyclic, conjugated, and has 4n + 2 pi electrons but contains a sp³ hybridized atom in the ring is classified as A) aromatic. B) non-aromatic. C) anti-aromatic. e here to search 82% 54°F C F5 F8 F9 F10 F11 F12 F3 F4 F6 F7 %23 $4 & 3. 4. 6. 17 8. 9. Y U H JK Larrow_forwardCharacterize each of the following structures as aromatic, nonaromatic, or antiaromatic: + +arrow_forward
- Determine if the following compounds are not aromatic, aromatic or anti-aromatic. :O: 00 IZ:arrow_forwardClassify each of the following molecules as aromatic, antiaromatic, or nonaromatic. O aromatic O antiaromatic O nonaromatic O aromatic O antiaromatic O nonaromatic O aromatic O antiaromatic O nonaromaticarrow_forwardDefine if AROMATIC, ANTI AROMATIC, OR NON AROMATICarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
CBSE Class 12 Chemistry || Polymers || Full Chapter || By Shiksha House; Author: Best for NEET;https://www.youtube.com/watch?v=OxdJlS0xZ0Y;License: Standard YouTube License, CC-BY