
(a)
Interpretation:
The equations for formation constants
Concept Introduction:
Formation Constant:
In the reaction of metal with ligand, the equilibrium constant is called as formation constant or the stability constant.
The formation constant for above complex reaction is,
Where,
(a)
Answer to Problem 12.18P
The equations for formation constants
The equations for formation constants
Explanation of Solution
To give the equations for formation constants
Given,
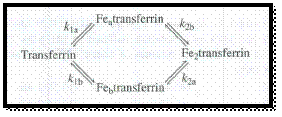
Figure 1
We know the formation constant for forward reaction is,
Apply the respective reactant and product in above equation to gives the equations for formation constants
The equations for formation constants
The equations for formation constants
The equations for formation constants
(b)
Interpretation:
The given respected equations should be explained.
Concept Introduction:
Formation Constant:
In the reaction of metal with ligand, the equilibrium constant is called as formation constant or the stability constant.
The formation constant for above complex reaction is,
Where,
(b)
Answer to Problem 12.18P
The equations for formation constants
The equations for formation constants
Explanation of Solution
To give the equations for formation constants
Given,

Figure 1
We know the formation constant for forward reaction is,
Apply the respective reactant and product in above equation to gives the equations for formation constants
The equations for formation constants
The equations for formation constants
The above equations are combined to give,
The reciprocal of
From the above derivation s the given equations are derived and they are shown correct relationship with corresponding reactions.
The given respected equations should be explained.
(c)
Interpretation:
Respect with the given reaction, the given expression should be explained.
Concept Introduction:
Formation Constant:
In the reaction of metal with ligand, the equilibrium constant is called as formation constant or the stability constant.
The formation constant for above complex reaction is,
Where,
(c)
Explanation of Solution
To give the equations for formation constants
Given,

Figure 1
The given equation is,
We know the formation constant for forward reaction is,
Apply the respective reactant and product in above equation to gives the equations for formation constants
The equations for formation constants
The equations for formation constants
The above equations are combined to give,
To cancel the common the terms,
Respect with the given reaction, the given expression was explained.
(d)
Interpretation:
From the given equations, the three unknown values are should be determined.
Concept Introduction:
Formation Constant:
In the reaction of metal with ligand, the equilibrium constant is called as formation constant or the stability constant.
The formation constant for above complex reaction is,
Where,
(d)
Answer to Problem 12.18P
Three unknown values are,
Explanation of Solution
To give the equations for formation constants
Given,

Figure 1
The given equation is,
We know the formation constant for forward reaction is,
Apply the respective reactant and product in above equation to gives the equations for formation constants
The equations for formation constants
The equations for formation constants
The above equations are combined to give,
Plugged the equation 1 and in 3 and equation 2 in 4,
To solve the quadratic equation to give the
The given value of
Using the calculated
From the above calculations three unknown values are,
From the given equations, the three unknown values are determined.
Want to see more full solutions like this?
Chapter 12 Solutions
Solution Manual for Quantitative Chemical Analysis
- Indicate the products obtained by reacting benzenesulfonic acid with a sulfonitric acid mixture (HNO3 + H2SO4). Indicate the majority if necessary.arrow_forwardIndicate the products obtained by reacting ethylbenzene with a sulfonitric acid mixture (HNO3 + H2SO4). Indicate the majority if necessary.arrow_forwardIndicate the products obtained when tert-butylbenzene reacts with a sulfonitric acid mixture (HNO3 + H2SO4). Indicate the majority if necessary.arrow_forward
- Indicate the products obtained when acetophenone reacts with a sulfonitric acid mixture (HNO3 + H2SO4). Indicate the majority if necessary.arrow_forwardIndicate the products obtained from the reaction of N-(4-methylphenyl)acetamide with a sulfonitric acid mixture (H2SO4 + HNO3). Indicate the majority if necessary.arrow_forwardIndicate the products obtained from the reaction of 4-(trifluoromethyl)benzonitrile with a sulfonitric mixture (H2SO4 + HNO3). Indicate the majority if necessary.arrow_forward
- Indicate the products obtained in the reaction of p-Toluidine with a sulfonitric acid mixture (H2SO4 + HNO3). Indicate the majority if necessary.arrow_forwardIndicate the products obtained from the reaction of 4-methylbenzonitrile with a sulfonitric acid mixture (H2SO4 + HNO3). Indicate the majority if necessary.arrow_forwardIndicate the products obtained from the reaction of 2-nitrophenol with a sulfonitric acid mixture (H2SO4 + HNO3). Indicate the majority if necessary.arrow_forward
- In organic chemistry, what is the correct name for the mixture H2SO4 + HNO3 used in reactions: sulphonitric mixture or sulfonitric mixture?arrow_forwardFormulate the products obtained by reacting p-toluidine with a sulfonate mixture. Indicate the majority if necessary.arrow_forwardConsider this organic reaction: OH Draw the major products of the reaction in the drawing area below. If there won't be any major products, because this reaction won't happen at a significant rate, check the box under the drawing area instead. Click and drag to start drawing a structure. x 0: の Carrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





