
FUND.OF GEN CHEM CHAP 1-13 W/ACCESS
16th Edition
ISBN: 9781323406038
Author: McMurry
Publisher: PEARSON C
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11.9, Problem 11.20KCP
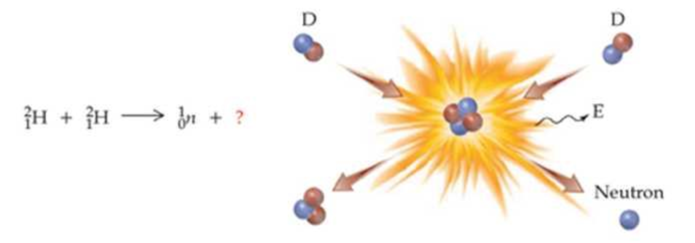
One of the possible reactions for nuclear fusion involves the collision of 2 deuterium nuclei. Complete the reaction by identifying the missing particle:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Show the mechanism for the acid-catalyzed formation of an [α-1,6] glycosidic linkagebetween two molecules of α-D-glucopyranose.
Please sketch the structure and use arrows showing electron flow!
I am a Biochemistry student and I am confused on how to analyze FRAP Analysis using Excel Spread Sheets.
The following spread sheet has my 0 minute data listed at top and the 4 minute data listed on the bottom.
Sheet: https://mnscu-my.sharepoint.com/:x:/g/personal/vi2163ss_go_minnstate_edu/EXjrCizWiXRPmpittqZA12IB8EkB5eE8iaRqj_iun-IAtg?rtime=Wo9zPHFY3Ug
The formula for FRAP Analysis is:
FRAP value = A (4 min sample) - A (0 min sample) over A (4 min 30 uM ascorbic acid) - A (0 min 30 uM ascorbic acid) multiplied by 30 uM and the dilution factor of 1/10
HO
Fill in the missing boxes.
ON
800
NO
NO
Glucose
ATP
NADH
Hexokinase
1,3-Bisphosphoglycerate
Mg2+
ADP
NAD+, Pi
Phosphoglucose
Isomerase
Glucose-6-Phosphate
ON
沁
Glyceraldehyde-3-Phosphate
HO
حلمة
ADP
ADP Phospho
Mg2+
glycerate
Dihydroxyacetone Phosphate
ATP
kinase
ATP
Phosphoglycerate
3-phosphoglycerate
Mutase
H₁₂O
Fructose-6-Phosphate
ATP
Mg2+
ADP
Fructose-1,6-Bisphosphate
2-phosphoglycerate
H₂O
Phosphoenolpyruvate
ADP
Mg2+
ATP
Pyruvate
Chapter 11 Solutions
FUND.OF GEN CHEM CHAP 1-13 W/ACCESS
Ch. 11.4 - Prob. 11.1PCh. 11.4 - Prob. 11.2PCh. 11.4 - Prob. 11.3PCh. 11.4 - Prob. 11.4PCh. 11.4 - Prob. 11.5PCh. 11.4 - Prob. 11.6PCh. 11.4 - Prob. 11.7KCPCh. 11.5 - What are the three main classes of techniques used...Ch. 11.5 - Prob. 11.2CIAPCh. 11.5 - The half-life of carbon-14, an isotope used in...
Ch. 11.5 - Prob. 11.9PCh. 11.5 - Prob. 11.10PCh. 11.5 - Prob. 11.11KCPCh. 11.6 - A -emitting radiation source gives 250 units of...Ch. 11.7 - What is the purpose of food irradiation, and how...Ch. 11.7 - What kind of radiation is used to treat food?Ch. 11.7 - A typical food irradiation application for the...Ch. 11.7 - A solution of selenium-75, a radioisotope used in...Ch. 11.7 - A typical chest X ray exposes a patient to an...Ch. 11.8 - Prob. 11.6CIAPCh. 11.8 - What advantages does MRI have over CT and PET...Ch. 11.8 - Prob. 11.8CIAPCh. 11.8 - Prob. 11.15PCh. 11.8 - The element berkelium, first prepared at the...Ch. 11.8 - Write a balanced nuclear equation for the reaction...Ch. 11.9 - What other isotope besides tellurium-137 is...Ch. 11.9 - Prob. 11.19PCh. 11.9 - One of the possible reactions for nuclear fusion...Ch. 11 - Magnesium-28 decays by emission to give...Ch. 11 - Prob. 11.22UKCCh. 11 - Prob. 11.23UKCCh. 11 - Prob. 11.24UKCCh. 11 - Prob. 11.25UKCCh. 11 - Prob. 11.26UKCCh. 11 - Prob. 11.27UKCCh. 11 - Prob. 11.28UKCCh. 11 - Prob. 11.29UKCCh. 11 - Prob. 11.30APCh. 11 - Describe how radiation, radiation, radiation,...Ch. 11 - Prob. 11.32APCh. 11 - Prob. 11.33APCh. 11 - Prob. 11.34APCh. 11 - Prob. 11.35APCh. 11 - Prob. 11.36APCh. 11 - Prob. 11.37APCh. 11 - Prob. 11.38APCh. 11 - Prob. 11.39APCh. 11 - Prob. 11.40APCh. 11 - Prob. 11.41APCh. 11 - Prob. 11.42APCh. 11 - What characteristic of uranium-235 fission causes...Ch. 11 - What products result from radioactive decay of the...Ch. 11 - Prob. 11.45APCh. 11 - Prob. 11.46APCh. 11 - Prob. 11.47APCh. 11 - Balance the following equations for the nuclear...Ch. 11 - Complete the following nuclear equations and...Ch. 11 - Prob. 11.50APCh. 11 - Cobalt-60 (half-life = 5.3 years) is used to...Ch. 11 - Prob. 11.52APCh. 11 - Prob. 11.53APCh. 11 - Prob. 11.54APCh. 11 - Prob. 11.55APCh. 11 - Selenium-75, a emitter with a half-life of 120...Ch. 11 - Prob. 11.57APCh. 11 - The half-life of mercury-197 is 64.1 hours. If a...Ch. 11 - Gold-198, a emitter used to treat leukemia, has a...Ch. 11 - Describe how a Geiger counter works.Ch. 11 - Prob. 11.61APCh. 11 - Prob. 11.62APCh. 11 - Prob. 11.63APCh. 11 - Prob. 11.64APCh. 11 - Match each unit in the left column with the...Ch. 11 - Prob. 11.66APCh. 11 - Sodium-24 is used to study the circulatory system...Ch. 11 - Prob. 11.68APCh. 11 - Prob. 11.69APCh. 11 - Prob. 11.70CPCh. 11 - Prob. 11.71CPCh. 11 - Prob. 11.72CPCh. 11 - Prob. 11.73CPCh. 11 - Prob. 11.74CPCh. 11 - Prob. 11.75CPCh. 11 - Prob. 11.76CPCh. 11 - Prob. 11.77CPCh. 11 - Prob. 11.78CPCh. 11 - Prob. 11.79CPCh. 11 - Prob. 11.80CPCh. 11 - Prob. 11.81CPCh. 11 - Prob. 11.82CPCh. 11 - Although turning lead into gold in a nuclear...Ch. 11 - Prob. 11.84CPCh. 11 - Prob. 11.85CPCh. 11 - Prob. 11.86CPCh. 11 - Prob. 11.87CPCh. 11 - Prob. 11.88CPCh. 11 - One way to demonstrate the dose factor of ionizing...Ch. 11 - One approach for treating cancerous tumors is...Ch. 11 - Prob. 11.91GP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- In a diffraction experiment of a native crystal, intensity of reflection (-1 0 6) is equivalent to the intensity of reflection (1 0 -6). true or false?arrow_forwardin an x-ray diffraction experiment, moving the detector farther away from the crystal will allow collection of reflection of reflections with high Miller indices. true or false?arrow_forwardShow the mechanism for the acid-catalyzed formation of an [α-1,6] glycosidic linkagebetween two molecules of α-D-glucopyranose.arrow_forward
- Label the following polysaccharide derivatives as reducing or nonreducing. a. C. b. HO CH₂OH CH2OH OH OH OH OH OH HOCH₂ OH OH OH HOCH₂ HO HO HO OH OH ΙΟ CH₂OH OH OH "OH OHarrow_forwardFor a red blood cell (erythrocyte) undergoing active glycolysis, briefly explain how increases in concentration of the following factors are likely to affect glycolytic flux. a. ATP b. AMP c. F-1,6-BP d. F-2,6-BP e. Citrate f. Glucose-6-phosphatearrow_forwardThe ∆G°’ for hydrolysis of phosphoenol pyruvate is -62.2 kJ/mol. The standard freeenergy of ATP hydrolysis is -30.5 kJ/mol. A. What is the standard free energy and K eq of the spontaneous reaction betweenADP/ATP and phosphoenol pyruvate. B. Repeat A for F-1,6-BP (∆G°’=-16.7 kJ/mol) and 1,3-BPG (∆G°’=-49.6 kJ/mol)hydrolysis. C. If the ATP and ADP concentrations are 8mM and 1mM respectively, what would bethe ratio of pyruvate/phosphoenolpyruvate at equilibrium?arrow_forward
- Answerarrow_forward13. Which one is the major organic product of the following sequence of reactions? A OH (CH3)2CHCH2COOH SOCI2 CH3OH 1. CH3MgBr 2. H₂O, H+ B C D OH E OHarrow_forward14. Which one is the major organic product of the following sequence of reactions? (CH3)2CH-COCI CH3OH 1. DIBALH, -78°C 1. PhCH2MgBr ? 2. H2O, HCI 2. H2O, HCI OH OMe A Ph B Ph OH Ph C OMe Ph D E OH .Pharrow_forward
- 6. Which one is the major organic product obtained from the following reaction? CO₂Me 1. LiAlH4 2. H₂O CH₂OH CH₂OCH3 5555 HO A B HO C HO D CH₂OH E ?arrow_forward1. (10 points) Pulverized coal pellets, which may be ° approximated as carbon spheres of radius r = 1 mm, are burned in a pure oxygen atmosphere at 1450 K and 1 atm. Oxygen is transferred to the particle surface by diffusion, where it is consumed in the reaction C + O₂ →> CO₂. The reaction rate is first order and of the form No2 = k₁C₁₂(r), where k₁ = 0.1 m/s. Neglecting changes in r, determine the steady-state O₂ molar consumption rate in kmol/s. At 1450 K, the binary diffusion coefficient for O2 and CO2 is 1.71 x 10ª m²/s.arrow_forward2. (20 points) Consider combustion of hydrogen gas in a mixture of hydrogen and oxygen adjacent to the metal wall of a combustion chamber. Combustion occurs at constant temperature and pressure according to the chemical reaction 2H₂+ O₂→ 2H₂O. Measurements under steady-state conditions at 10 mm from the wall indicate that the molar concentrations of hydrogen, oxygen, and water vapor are 0.10, 0.10, and 0.20 kmol/m³, respectively. The generation rate of water vapor is 0.96x102 kmol/m³s throughout the region of interest. The binary diffusion coefficient for each of the species (H, O̟, and H₂O) in the remaining species is 0.6 X 10-5 m²/s. (a) Determine an expression for and make a qualitative plot of C as a function of distance from the wall. H2 (b) Determine the value of C2 at the wall. H2 (c) On the same coordinates used in part (a), sketch curves for the concentrations of oxygen and water vapor. This will require you to calculate Co, and C. 02 H20 (d) What is the molar flux of water…arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning- Essentials Health Info Management Principles/Prac...Health & NutritionISBN:9780357191651Author:BowiePublisher:Cengage

Principles Of Radiographic Imaging: An Art And A ...
Health & Nutrition
ISBN:9781337711067
Author:Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:Cengage Learning




Essentials Health Info Management Principles/Prac...
Health & Nutrition
ISBN:9780357191651
Author:Bowie
Publisher:Cengage

DNA vs RNA (Updated); Author: Amoeba Sisters;https://www.youtube.com/watch?v=JQByjprj_mA;License: Standard youtube license