
Concept explainers
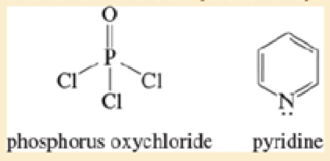
Some alcohols undergo rearrangement or other unwanted side reactions when they dehydrate in acid Alcohols may be dehydrated under mildly basic conditions using phosphorus oxychloride (POCl3) in pyridine. The alcohol reacts with phosphorus oxychloride much like it reacts with tosyl chloride (Section11-5), displacing a chloride ion from phosphorus to give an alkyl dichloroptrosphate ester. The dichlorophosphate group is an outstanding leaving group. Pyridine reacts as a base with the dichlorophosphate ester to give an E2 elimination. Propose a mechanism for the dehydration of cyclohexanol by POCl3 in pyridine.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
ORGANIC CHEMISTRY
Additional Science Textbook Solutions
Fundamentals of Physics Extended
Biology: Life on Earth (11th Edition)
MARINE BIOLOGY
Laboratory Experiments in Microbiology (12th Edition) (What's New in Microbiology)
Organic Chemistry
Campbell Essential Biology (7th Edition)
- 2. 200 LOD For an unknown compound with a molecular ion of 101 m/z: a. Use the molecular ion to propose at least two molecular formulas. (show your work) b. What is the DU for each of your possible formulas? (show your work) C. Solve the structure and assign each of the following spectra. 8 6 4 2 (ppm) 150 100 50 ō (ppm) 4000 3000 2000 1500 1000 500 HAVENUMBERI-11arrow_forwardComplete the spectroscopy with structurearrow_forwardComplete the spectroscopy with structurearrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

