
Chemistry: The Central Science (13th Edition)
13th Edition
ISBN: 9780321910417
Author: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10.8, Problem 10.14.2PE
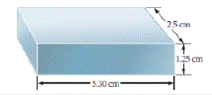
- How many significant figures should be reported for the volume of the metal bar shown here?
- If the mass of the bar is 104.72 g, how many significant figures should be reported when its density is determined using the calculated volume? [Section 1.60
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A student sets up the following equation to convert a measurement.
(The ? stands for a number the student is going to calculate.)
Fill in the missing part of this equation.
0.050
mol
kg
= ?
mol
g
口。
x10
X
μ
0|0
G
A chemistry student needs 30.0 g of diethylamine for an experiment. By consulting the CRC Handbook of Chemistry and Physics, the student discovers that the
density of diethylamine is 0.706 g·cm
-3
Calculate the volume of diethylamine the student should pour out.
Be sure your answer has the correct number of significant digits.
O mL
Methyl benzoate is an oily liquid with a boiling point of 199°C. What volume of methyl benzoate is needed to prepare 5.8 grams of benzoic acid? The density of methyl benzoate is 1.08 g·cm⁻³. Molar masses are given in Figure 4. [Give only the number without the unit. Give the answer in cubic centimetres and round to 1 decimal place. Use a decimal point.] *
Your answer
Chapter 10 Solutions
Chemistry: The Central Science (13th Edition)
Ch. 10.2 - Classify each of the following as a pure substance...Ch. 10.2 - Classify each of the following as a pure substance...Ch. 10.2 - 1.15 Give the chemical symbol or name for the...Ch. 10.2 - 1.16 Give the chemical symbol or name for each of...Ch. 10.3 - A solid white substance A is heated strongly in...Ch. 10.3 - 1.18 You are hiking in the mountains and find a...Ch. 10.4 - 1.19 In the process of attempting to characterize...Ch. 10.4 - 1.20
Read the following description of the element...Ch. 10.4 - Prob. 10.5.1PECh. 10.4 - A match is lit and held under a cold piece of...
Ch. 10.4 - Which separation method is better suited for...Ch. 10.4 - Two beakers contain clear, colorless liquids. When...Ch. 10.5 - Prob. 10.7.1PECh. 10.5 - Prob. 10.7.2PECh. 10.5 - Prob. 10.8.1PECh. 10.5 - Prob. 10.8.2PECh. 10.5 - Prob. 10.9.1PECh. 10.5 - Prob. 10.9.2PECh. 10.6 - Prob. 10.10.1PECh. 10.6 - Prob. 10.10.2PECh. 10.6 - Musical instruments like trumpets and trombones...Ch. 10.6 - Consider the two spheres shown here, one made of...Ch. 10.7 - Is the separation method used in brewing a cup of...Ch. 10.7 - Identify each of the following as measurements of...Ch. 10.8 - Three spheres of equal size are composed of...Ch. 10.8 - The three targets from a rifle range shown below...Ch. 10.8 - What is the length of the pencil in the following...Ch. 10.8 - How many significant figures should be reported...Ch. 10.9 - Consider the jar of jelly beans in the photo. To...Ch. 10.9 - The photo below shows a picture of an agate stone....Ch. 10 - SO Two students deterrmne the percen.ge of lead in...Ch. 10 - 1.70
Is Om use of significant figures in ea. of...Ch. 10 - Water has a density of 0.997 g/cm3 at 25C ; ice...Ch. 10 - Prob. 3ECh. 10 - Practice Exercise 1 A biochemist who is studying...Ch. 10 - Practice Exercise 2
Write the empirical formula...Ch. 10 - Prob. 6ECh. 10 - Hydrogen sulfide is composed of two elements:...Ch. 10 - Consider an atom of "B. a. How many protons,...Ch. 10 - 2.34
a. What is the mass in amu of a carbon-12...Ch. 10 - Prob. 10ECh. 10 - You have a graduated cylinder that contains a...Ch. 10 - The density of air at ordinary atmospheric...Ch. 10 - Prob. 13ECh. 10 - Prob. 14ECh. 10 - Prob. 15ECh. 10 - 165 Classify ea. al the folbwing as a pure...Ch. 10 - Prob. 17ECh. 10 - Prob. 18ECh. 10 - Prob. 19ECh. 10 - What type of quantity (for example, length,...Ch. 10 - 1.72 Give the derived SI units for each of the...Ch. 10 - 1.73 The distance from Earth to the Moon is...Ch. 10 - 1.74 Which of the following would you characterize...Ch. 10 -
1.75 The U.S. quarter has a mass of 5.67 g and is...Ch. 10 -
1.76 In the United States, water used for...Ch. 10 -
1.77 By using estimation techniques, determine...Ch. 10 - Suppose you decide to define your own temperature...Ch. 10 -
1.79 The liquid substances mercury (density =...Ch. 10 -
1.80 Two spheres of equal volume are placed on...Ch. 10 - A 32.65-g sample of a solid is placed in a flask....Ch. 10 - A thief plans to steal a gold sphere with a radius...Ch. 10 - Automobile batteries contain sulfuric acid, which...Ch. 10 - A 40-lb container of peat moss measures 14 x 20 x...Ch. 10 - A package of aluminum foil contains 50 ft2of foil,...Ch. 10 - Prob. 35ECh. 10 -
1.88 In 2005, J. Robin Warren and Barry J....Ch. 10 -
1 89 A 25 0-cm.long cylindrical glass tube,...Ch. 10 -
1.90 Gold is alloyed (mixed) with other metals to...Ch. 10 -
1.91 Paper chromatography is a simple but...Ch. 10 -
1.93 You are assigned the task of separating a...Ch. 10 - Prob. 41ECh. 10 - Which of the following factors determines the size...Ch. 10 - Practice Exercise 2 The diameter of a cartoon atom...Ch. 10 - Practice Exercise 1 Which of these atoms has the...Ch. 10 - Practice Exercise 2
How many protons, neutrons,...Ch. 10 - Prob. 46ECh. 10 - Which is mode at 1.00 atm and 298K: CO2,,N2O,or...Ch. 10 - Practice Exercise 1 There are two stable isotopes...Ch. 10 - Practice Exercise 2
Three isotopes of silicon...Ch. 10 - Practice Exercise 2 Locate Na (sodium) and Br...Ch. 10 - Practice Exercise 1 Tetra carbon dioxide is an...Ch. 10 - Practice Exercise 2 Give the empirical formula for...Ch. 10 - Practice Exercise 1 In which of the following...Ch. 10 - Practice Exercise 2 How many protons, neutrons,...Ch. 10 - Practice Exercise 1
Although it is helpful to...Ch. 10 - Prob. 56ECh. 10 - Prob. 57ECh. 10 - Prob. 58ECh. 10 - Prob. 59ECh. 10 - Practice Exercise 1 Which of the follow-mg ox...Ch. 10 - Prob. 61ECh. 10 - Prob. 62ECh. 10 - Prob. 63ECh. 10 - Prob. 64ECh. 10 - Prob. 65ECh. 10 - Prob. 66ECh. 10 - Practice Exercise 2
Give the chemical fomi uias...Ch. 10 - Prob. 68ECh. 10 - Prob. 69ECh. 10 - The followmg diagram is a representation of 20...Ch. 10 - 2 3 Four of the boxes in the following periodic...Ch. 10 -
24 Does the following drawing represent a neutral...Ch. 10 - 2.5 Which of the following diagrams most likely...Ch. 10 - Write the chemical formula for the following...Ch. 10 - Prob. 75ECh. 10 - Prob. 76ECh. 10 - Prob. 77ECh. 10 - Prob. 78ECh. 10 - Prob. 79ECh. 10 - Prob. 80ECh. 10 - Prob. 81ECh. 10 - Prob. 82ECh. 10 - Prob. 83ECh. 10 - Prob. 84ECh. 10 - Explain the difference between effusion and...Ch. 10 - Prob. 86ECh. 10 - Prob. 87ECh. 10 - Prob. 88ECh. 10 - Prob. 89ECh. 10 - Prob. 90ECh. 10 - Prob. 91ECh. 10 - Prob. 92ECh. 10 - Prob. 93ECh. 10 - Prob. 94ECh. 10 - In Sample Exercise 10.16, we found that one mole...Ch. 10 - Prob. 96ECh. 10 - Prob. 97ECh. 10 - Prob. 98ECh. 10 - Prob. 99AECh. 10 - Prob. 100AECh. 10 - Prob. 101AECh. 10 - Prob. 102AECh. 10 - Prob. 103AECh. 10 - Prob. 104AECh. 10 - Prob. 105AECh. 10 - Prob. 106AECh. 10 - Prob. 107AECh. 10 - Prob. 108AECh. 10 - Prob. 109AECh. 10 - Prob. 110AECh. 10 - Prob. 111AECh. 10 - Prob. 112AECh. 10 - Prob. 113AECh. 10 - Prob. 114AECh. 10 - Prob. 115AECh. 10 - Prob. 116AECh. 10 - Prob. 117AECh. 10 - Prob. 118AECh. 10 - Prob. 119IECh. 10 - Prob. 120IECh. 10 - Prob. 121IECh. 10 - Prob. 122IECh. 10 - Prob. 123IECh. 10 - Prob. 124IECh. 10 - Chlorine dioxide gas (ClO2) is used as a...Ch. 10 - Natural gas is very abundant us many Middle...Ch. 10 -
[10.127] Gaseous iodine pentafluoride. IF3 can be...Ch. 10 - [10.128]A 6.53-g sample of mixture of magnesium...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Suppose a room is 18 m long, 15 m wide, and the distance from floor to ceiling is 2.9 m. What is the room’s volume in cubic meters? In cubic centimeters? In liters?arrow_forwardThe quantitative significance of “take a deep breath" varies, of course, with the individual. When one person did so, she found that she inhaled 3.62x10^22 molecules of the mixture of mostly nitrogen and oxygen we call air. Assuming this mixture has an average molar mass of 29.0 g/mol, what is her apparent lung capacity in grams of airarrow_forwardA chemist must prepare 475. mL of 1.00 M aqueous sodium nitrate (NaNO3) working solution. He'll do this by pouring out some 5.64 nitrate stock solution into a graduated cylinder and diluting it with distilled water. Calculate the volume in mL of the sodium nitrate stock solution that the chemist should pour out. Round your answer to 3 significant digits. mL mol L X aqueous sodiumarrow_forward
- A chemistry student needs 35.0 mL of dimethyl sulfoxide for an experiment. By consulting the CRC Handbook of Chemistry and Physics, the student discovers that the density of dimethyl sulfoxide is 1.10 g·cm -3 Calculate the mass of dimethyl sulfoxide the student should weigh out. Be sure your answer has the correct number of significant digits. ?arrow_forwardThe volume of water in large scale reservoirs is often measured in units of an acre-foot, which is the amount of water covering one acre of surface to a depth of one foot. An acre-foot is equivalent to 43,560 ft³. A particular water tower in town was constructed to hold 2.55 acre-feet of water. What is the volume of water in liters that the water tower will hold? ( 1 US liquid gallon = 231 cubic inches, 3.785 liters = 1 US liquid gallon)arrow_forwardCarbon tetrachloride, CCl4, was a popular dry-cleaning agent until it was shown to be carcinogenic. It has a density of 1.589 g/cm3. What volume of carbon tetrachloride will contain a total of 3.00 × 1025 molecules of CCl4? Volume = ___cm3arrow_forward
- A chemistry student needs 40.0 mL of glycerol for an experiment. By consulting the CRC Handbook of Chemistry and Physics, the student discovers that the density of glycerol is 1.26 g·cm -3 Calculate the mass of glycerol the student should weigh out. Round your answer to 3 significant digits.arrow_forwardA chemistry student needs 60.0 g of tetrahydrofuran for an experiment. By consulting the CRC Handbook of Chemistry and Physics, the student discovers that the density of tetrahydrofuran em ¯³. Calculate the volume of tetrahydrofuran the student should pour out. is 0.889 g·cm Be sure your answer has the correct number of significant digits. I mLarrow_forwardIf a sample of unknown substance has a mass of 3.10 kg and a volume of 1.80 L, what is the density of the substance when expressed in conventional (but non-SI) units of g/cm3?arrow_forward
- A chemistry student needs 25.0g of diethylamine for an experiment. By consulting the CRC Handbook of Chemistry and Physics, the student discovers that the density of diethylamine is ·0.706g cm−3. Calculate the volume of diethylamine the student should pour out. Round your answer to 3 significant digits.arrow_forwardCalculate the volume of tetrahydrofuran the student should pour out. Round your answer to 3 significant digits.arrow_forwardwhat mass (in g) of solid lead would displace exactly 234.6 liters of water? The density of the lead that is given is 11.4 g/cm^3. the correct answer must contain the appropriate use of significant figures.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
Measurement and Significant Figures; Author: Professor Dave Explains;https://www.youtube.com/watch?v=Gn97hpEkTiM;License: Standard YouTube License, CC-BY
Trigonometry: Radians & Degrees (Section 3.2); Author: Math TV with Professor V;https://www.youtube.com/watch?v=U5a9e1J_V1Y;License: Standard YouTube License, CC-BY