
EBK ORGANIC CHEMISTRY
7th Edition
ISBN: 9780133556186
Author: Bruice
Publisher: VST
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 10.10, Problem 31P
Why is a cumulated diene not formed in the reaction shown above?
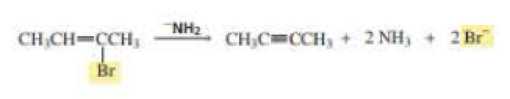
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Indicate what metal clusters are.
55. The photoelectric threshold energy for ytterbium
metal is 4.16 × 10-19 J/atom.
a. Calculate the wavelength of light that this
energy corresponds to (in nm).
b. Which region of the electromagnetic spectrum
does this wavelength fall in?
c. Would light of wavelength 490 nm produce a
photoelectric effect in ytterbium? Why or why
not?
14.50 Explain why methyl vinyl ether (CH2=CHOCH 3) is not a reactive
dienophile in the Diels-Alder reaction.
Chapter 10 Solutions
EBK ORGANIC CHEMISTRY
Ch. 10.2 - Prob. 1PCh. 10.2 - Prob. 2PCh. 10.2 - Prob. 3PCh. 10.2 - Prob. 4PCh. 10.3 - Four alkenes are formed from the E1 reaction of...Ch. 10.3 - If 2-fluoropentane could undergo an E1 reaction,...Ch. 10.3 - Prob. 7PCh. 10.3 - Propose a mechanism for the following reaction:Ch. 10.4 - Prob. 9PCh. 10.4 - What products will be obtained from the El...
Ch. 10.4 - Prob. 11PCh. 10.5 - Prob. 12PCh. 10.6 - Prob. 14PCh. 10.7 - Why do cis-1-bromo-2-ethylcyclohexane and...Ch. 10.7 - Which isomer reacts more rapidly in an E2...Ch. 10.7 - Prob. 18PCh. 10.8 - Prob. 19PCh. 10.8 - Prob. 20PCh. 10.9 - Prob. 21PCh. 10.9 - Explain why only a substitution product and no...Ch. 10.9 - Prob. 23PCh. 10.9 - Prob. 24PCh. 10.9 - Prob. 25PCh. 10.9 - a. Explain why 1-bromo-2,2-dimethylpropane has...Ch. 10.10 - A small amount of another organic product is...Ch. 10.10 - What is the best way to prepare the following...Ch. 10.10 - Prob. 29PCh. 10.10 - Prob. 30PCh. 10.10 - Why is a cumulated diene not formed in the...Ch. 10.10 - What product is obtained when the following...Ch. 10.11 - Prob. 33PCh. 10.11 - Prob. 34PCh. 10 - Draw the major product obtained when each of the...Ch. 10 - Prob. 36PCh. 10 - a. Indicate how each of the following factors...Ch. 10 - Prob. 38PCh. 10 - A chemist wanted to synthesize the...Ch. 10 - Prob. 40PCh. 10 - Prob. 41PCh. 10 - Prob. 42PCh. 10 - Starting with an alkyl halide, how could the...Ch. 10 - Indicate which species in each pair gives a higher...Ch. 10 - Prob. 45PCh. 10 - For each of the following alkyl halides, indicate...Ch. 10 - Prob. 47PCh. 10 - When 2-bromo-2,3-dimethylbutane reacts with a...Ch. 10 - Prob. 49PCh. 10 - When the following compound undergoes solvolysis...Ch. 10 - cis-1-Bromo-4-tert-butylcyclohexane and...Ch. 10 - Draw the substitution and elimination products.Ch. 10 - Prob. 53PCh. 10 - Prob. 54PCh. 10 - Which of the following hexachlorocyclohexanes is...Ch. 10 - Explain why the rate of the reaction of...Ch. 10 - Prob. 57PCh. 10 - Two elimination products are obtained from the...Ch. 10 - Draw the structures or the product of the obtained...Ch. 10 - How could you prepare the following compounds from...Ch. 10 - cis-4-Bromocyclohexanol and...Ch. 10 - Prob. 62PCh. 10 - Prob. 63P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Show work with explanation needed. don't give Ai generated solutionarrow_forward14.49 From what you have learned about the reaction of conjugated dienes in Section 14.10, predict the products of each of the following electrophilic additions. a. H₂O H2SO4 Br2 b. H₂Oarrow_forward14.46 Draw a stepwise mechanism for the following reaction. HBr ROOR Br + Brarrow_forward
- Show work..don't give Ai generated solution....arrow_forward14.47 Addition of HCI to alkene X forms two alkyl halides Y and Z. exocyclic C=C X HCI CI Y + CI Z a. Label Y and Z as a 1,2-addition product or a 1,4-addition product. b.Label Y and Z as the kinetic or thermodynamic product and explain why. c. Explain why addition of HCI occurs at the indicated C=C (called an exocyclic double bond), rather than the other C=C (called an endocyclic double bond).arrow_forward14.44 Ignoring stereoisomers, draw all products that form by addition of HBr to (E)-hexa-1,3,5-triene.arrow_forward
- Include stereochemistry Leven though the solutions manual does 14.43 Draw the products formed when each compound is treated with one not) equivalent of HBr. a. b. C.arrow_forward14.41 Label each pair of compounds as stereoisomers, conformations, or constitutional isomers: (a) A and B; (b) A and C; (c) A and D; (d) C and D. A B C Darrow_forwardSteps and detailed explanation for work. Thanks!arrow_forward
- 14.39 Draw the structure of each compound. a. (Z)-penta-1,3-diene in the s-trans conformation b. (2E,4Z)-1-bromo-3-methylhexa-2,4-diene c. (2E,4E,6E)-octa-2,4,6-triene d. (2E,4E)-3-methylhexa-2,4-diene in the s-cis conformationarrow_forwardPLEASE ANSWER ALL PARTS!!arrow_forwardpls help on all, inlcude all steps.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Seven Name Reactions in One - Palladium Catalysed Reaction (047 - 053); Author: Rasayan Academy - Jagriti Sharma;https://www.youtube.com/watch?v=5HEKTpDFkqI;License: Standard YouTube License, CC-BY