
Concept explainers
(a)
Interpretation:
The most polar bond of methanol should be identified and polarity described with symbols
Concept Introduction:
Polarity of any molecule depends on electronegativity of atoms that forms covalent bonds. Depending upon the difference in electronegativity of atoms, polar or nonpolar molecule is determined. Polar molecule has electronegativity between 0.5 and 1.4 whereas in nonpolar molecule, it is less than 0.5.
Answer to Problem 10.49P
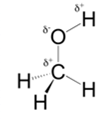
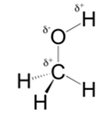
In methanol, most polar bond is O-H bond with electronegativity difference of 1.4 and oxygen atom becomes partial negative charge atom whereas hydrogen becomes partial positive charge atom.
Explanation of Solution
Polar covalent bonds contains more atoms with high electronegativity and acquire more partial negative charge represented as delta minus
In methanol, most polar bond is O-H with electronegativity difference of 1.4 and oxygen atom becomes partial negative charge atom whereas hydrogen becomes partial positive charge atom.
C-O bond have 1.0 electronegativity but it is less polar when compare to O-H bond.
(b)
Interpretation:
The most polar bond of methylamine should be identified and polarity described with symbols
Concept Introduction:
Polarity of any molecule depends on electronegativity of atoms that forms covalent bonds. Depending upon the difference in electronegativity of atoms, polar or nonpolar molecule is determined. Polar molecule has electronegativity between 0.5 and 1.4 whereas in nonpolar molecule, it is less than 0.5.
Answer to Problem 10.49P
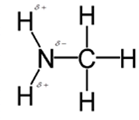
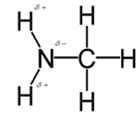
In methylamine, most polar bond is N-H bond with electronegativity difference of 0.9 and nitrogen atom becomes partial negative charge atom whereas hydrogen becomes partial positive charge atom.
Explanation of Solution
Polar covalent bonds contains more atoms with high electronegativity and acquire more partial negative charge represented as delta minus
In methylamine, most polar bond is N-H with electronegativity difference of 0.9 and nitrogen atom becomes partial negative charge atom whereas hydrogen becomes partial positive charge atom.
C-H bond is nonpolar because of electronegativity of 0.4 only.
(c)
Interpretation:
The most polar bond of 2-aminoethathiol should be identified and polarity described with symbols
Concept Introduction:
Polarity of any molecule depends on electronegativity of atoms that forms covalent bonds. Depending upon the difference in electronegativity of atoms, polar or nonpolar molecule is determined. Polar molecule has electronegativity between 0.5 and 1.4 whereas in nonpolar molecule, it is less than 0.5.
Answer to Problem 10.49P
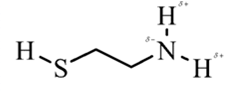
In 2-aminoethathiol, most polar bond is N-Hbond with electronegativity difference of 0.9 and nitrogen atom becomes partial negative charge atom whereas hydrogen becomes partial positive charge atom.
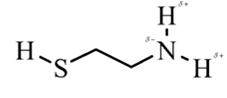
Explanation of Solution
Polar covalent bonds contains more atoms with high electronegativity and acquire more partial negative charge represented as delta minus
In 2-aminoethanethiol, most polar bond is N-H with electronegativity difference of 0.9 and nitrogen atom becomes partial negative charge atom whereas hydrogen becomes partial positive charge atom.
The bonds S-H, C-C and C-H have electronegativity difference are 0.4, 0, and 0.4 respectively.
(d)
Interpretation:
The most polar bond of acetone should be identified and polarity described with symbols
Concept Introduction:
Polarity of any molecule depends on electronegativity of atoms that forms covalent bonds. Depending upon the difference in electronegativity of atoms, polar or nonpolar molecule is determined. Polar molecule has electronegativity between 0.5 and 1.4 whereas in nonpolar molecule, it is less than 0.5.
Answer to Problem 10.49P
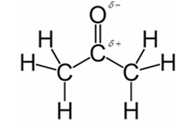
In acetone, most polar bond is C=O with electronegativity difference of 1.0 and oxygen atom becomes partial negative charge atom whereas carbon becomes partial positive charge atom.
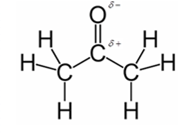
Explanation of Solution
Polar covalent bonds contains more atoms with high electronegativity and acquire more partial negative charge represented as delta minus
In acetone, most polar bond is C=O with electronegativity difference of 1.0 and oxygen atom becomes partial negative charge atom whereas carbon becomes partial positive charge atom.
Other bonds present in acetone are C-H and C-C with electronegativity of 0.4 and 0 respectively.
(e)
Interpretation:
The most polar bond of formaldehyde should be identified and polarity described with symbols
Concept Introduction:
Polarity of any molecule depends on electronegativity of atoms that forms covalent bonds. Depending upon the difference in electronegativity of atoms, polar or nonpolar molecule is determined. Polar molecule has electronegativity between 0.5 and 1.4 whereas in nonpolar molecule, it is less than 0.5.
Answer to Problem 10.49P
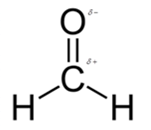
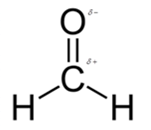
In formaldehyde, most polar bond is C=O with electronegativity difference of 1.0 and oxygen atom becomes partial negative charge atom whereas carbon becomes partial positive charge.
Explanation of Solution
Polar covalent bonds contains more atoms with high electronegativity and acquire more partial negative charge represented as delta minus
In formaldehyde, most polar bond is C=O with electronegativity difference of 1.0 and oxygen atom becomes partial negative charge atom whereas carbon becomes partial positive charge atom.
The bond C-H has electronegativity difference of 0.4 and it is a nonpolar bond.
(f)
Interpretation:
The most polar bond of acetic acid should be identified and polarity described with symbols
Concept Introduction:
Polarity of any molecule depends on electronegativity of atoms that forms covalent bonds. Depending upon the difference in electronegativity of atoms, polar or nonpolar molecule is determined. Polar molecule has electronegativity between 0.5 and 1.4 whereas in nonpolar molecule, it is less than 0.5.
Answer to Problem 10.49P
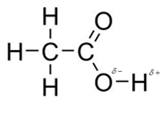
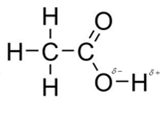
In acetic acid, most polar bond is O-H with electronegativity difference of 1.4 and oxygen atom becomes partial negative charge atom whereas hydrogen becomes partial positive charge atom.
Explanation of Solution
Polar covalent bonds contains more atoms with high electronegativity and acquire more partial negative charge represented as delta minus
In acetic acid, most polar bond is O-H with electronegativity difference of 1.4 and oxygen atom becomes partial negative charge atom whereas hydrogen becomes partial positive charge atom.
The other bonds are C-C, C=O and C-H with electronegativity of 0, 1.0 and 0.4 but bond O-H has more electronegativity as compared to other bonds.
Want to see more full solutions like this?
Chapter 10 Solutions
Bundle: Introduction to General, Organic and Biochemistry, 11th + OWLv2, 4 terms (24 months) Printed Access Card
- Indicate the products obtained by mixing 2,2-dimethylpropanal with acetaldehyde and sodium ethoxide in ethanol.arrow_forwardSynthesize 2-Ethyl-3-methyloxirane from dimethyl(propyl)sulfonium iodide using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize 2-Hydroxy-2-phenylacetonitrile from phenylmethanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forward
- Synthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardIf possible, please provide the formula of the compound 3,3-dimethylbut-2-enal.arrow_forward
- Synthesize 1,4-dibromobenzene from acetanilide (N-phenylacetamide) using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardIndicate the products obtained by mixing (3-oxo-3-phenylpropyl)triphenylphosphonium bromide with sodium hydride.arrow_forwardWe mix N-ethyl-2-hexanamine with excess methyl iodide and followed by heating with aqueous Ag2O. Indicate the major products obtained.arrow_forward
- Indicate the products obtained by mixing acetophenone with iodine and NaOH.arrow_forwardIndicate the products obtained by mixing 2-Propanone and ethyllithium and performing a subsequent acid hydrolysis.arrow_forwardIndicate the products obtained if (E)-2-butenal and 3-oxo-butanenitrile are mixed with sodium ethoxide in ethanol.arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

