
Concept explainers
a.
To draw:
The structure of alanine and mark an asterisk (*) next to any chiral carbon centers.
Introduction:
Amino acids contain a protonated
a.
Explanation of Solution
Alanine is a non essential amino acid. The three letter abbreviation of alanine is “Ala” and the one letter code is “A”. The side chain of alanine contains hydrocarbon as functional group.
Pictorial representation:
The structure of amino acid Alanine.
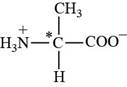
Fig 1: The structure of amino acid Alanine (Ala, A).
b.
To determine:
The structure of lysine and mark an asterisk (*) next to any chiral carbon centers
Introduction:
Amino acids contain a protonated amine and a carboxylic acid in the form of carboxylate ion. These two functional groups are bonded to a central carbon atom called the alpha-carbon. The protonated amine bonded to this carbon is called alpha-amino group and the carboxylate ion as the alpha carboxylate group. The alpha-carbon is also bonded to a hydrogen atom and a large side chain designated as R, which gives unique identification and characteristics to each amino acids.
b.
Explanation of Solution
Lysine is an essential amino acid. The three letter abbreviation of lysine is “Lys” and the one letter code is “K”. The side chain of lysine contains protonated amine as functional group.
Pictorial representation: The structure of amino acid Lysine.
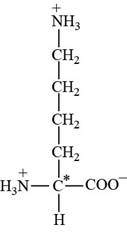
Fig 2: The structure of amino acid Lysine (Lys, K).
c.
To determine:
The structure of tryptophan and mark an asterisk (*) next to any chiral carbon centers
Introduction:
Amino acids contain a protonated amine and a carboxylic acid in the form of carboxylate ion. These two functional groups are bonded to a central carbon atom called the alpha-carbon. The protonated amine bonded to this carbon is called alpha-amino group and the carboxylate ion as the alpha carboxylate group. The alpha-carbon is also bonded to a hydrogen atom and a large side chain designated as R, which gives unique identification and characteristics to each amino acids.
c.
Explanation of Solution
Tryptophan is an essential amino acid. The three letter abbreviation of tryptophan is “Trp” and the one letter code is “W”. The side chain of tryptophan contains
Pictorial representation:
The structure of amino acid Tryptophan.
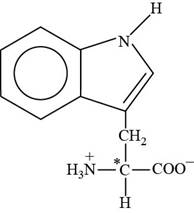
Fig 3: The structure of amino acid Tryptophan (Trp, W).
d.
To determine:
The structure of aspartate and mark an asterisk (*) next to any chiral carbon centers
Introduction:
Amino acids contain a protonated amine and a carboxylic acid in the form of carboxylate ion. These two functional groups are bonded to a central carbon atom called the alpha-carbon. The protonated amine bonded to this carbon is called alpha-amino group and the carboxylate ion as the alpha carboxylate group. The alpha-carbon is also bonded to a hydrogen atom and a large side chain designated as R, which gives unique identification and characteristics to each amino acids.
d.
Explanation of Solution
Aspartate is a non essential amino acid. The three letter abbreviation of aspartate is “Asp” and the one letter code is “D”. The side chain of aspartate contains carboxylate as functional group.
Pictorial representation:
The structure of amino acid Aspartate.
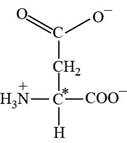
Fig 4: The structure of amino acid Aspartate (Asp, D)
Want to see more full solutions like this?
Chapter 10 Solutions
General, Organic, and Biological Chemistry (3rd Edition)
- Steps and explanations pleasearrow_forwardUse diagram to answer the following: 1.Is the overall rxn endo- or exothermic. Explain briefly your answer____________________2. How many steps in this mechanism?_____________3. Which is the rate determining step? Explain briefly your answer____________________4. Identify (circle and label) the reactants,the products and intermediate (Is a Cation, Anion, or a Radical?) Please explain and provide full understanding.arrow_forwardDraw the entire mechanism and add Curved Arrows to show clearly how electrons areredistributed in the process. Please explain and provide steps clearly.arrow_forward
- Match the denticity to the ligand. Water monodentate ✓ C₂O2 bidentate H₂NCH₂NHCH2NH2 bidentate x EDTA hexadentate Question 12 Partially correct Mark 2 out of 2 Flag question Provide the required information for the coordination compound shown below: Na NC-Ag-CN] Number of ligands: 20 Coordination number: 2✔ Geometry: linear Oxidation state of transition metal ion: +3 x in 12 correct out of 2 question Provide the required information for the coordination compound shown below. Na NC-Ag-CN] Number of ligands: 20 Coordination number: 2 Geometry: linear 0 Oxidation state of transition metal ion: +3Xarrow_forwardCan you explain step by step behind what the synthetic strategy would be?arrow_forwardPlease explain step by step in detail the reasoning behind this problem/approach/and answer. thank you!arrow_forward
- 2. Predict the product(s) that forms and explain why it forms. Assume that any necessary catalytic acid is present. .OH HO H₂N OHarrow_forwardconsider the rate of the reaction below to be r. Whats the rate after each reaction? Br + NaCN CN + NaBr a. Double the concentration of alkyl bromide b. Halve the concentration of the electrophile & triple concentration of cyanide c. Halve the concentration of alkyl chloridearrow_forwardPredict the organic reactant that is involved in the reaction below, and draw the skeletal ("line") structures of the missing organic reactant. Please include all steps & drawings & explanations.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





