
Concept explainers
(a)
Interpretation:TheKekulè (straight line) notation for the given condensed formula needs to be determined.
Concept Introduction:The chemical compounds can be shown in different structural formulae. Condense formula, wedge-dashed formula and bond-line formula are some common ways to show the structural of chemical compounds.
In wedge-dashed line formula, the groups or atoms bonded to each C atoms must be shown with dashed and wedges. Here wedge is the solid line that represents the bond in plane of the surface whereas dash line represents the line extended back behind the surface.
In the bond line formula, each C is shown as dot and the each dot is connected with another dot with line. Excluding C and H, all other elements of the compound like N, S, O must be indicted with their atomic symbols. In Kekulè (straight line) notation all atoms and
(b)
Interpretation: The Kekulè (straight line) notation for the given condensed formula needs to be determined.
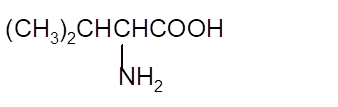
Concept Introduction:The chemical compounds can be shown in different structural formulae. Condense formula, wedge-dashed formula and bond-line formula are some common ways to show the structural of chemical compounds.
In wedge-dashed line formula, the groups or atoms bonded to each C atoms must be shown with dashed and wedges. Here wedge is the solid line that represents the bond in plane of the surface whereas dash line represents the line extended back behind the surface.
In the bond line formula, each C is shown as dot and the each dot is connected with another dot with line. Excluding C and H, all other elements of the compound like N, S, O must be indicted with their atomic symbols. In Kekulè (straight line) notation all atoms and chemical bonds must be shown with any symbol of lone pairs on atoms if any.
(c)
Interpretation: The Kekulè (straight line) notation for the given condensed formula needs to be determined.
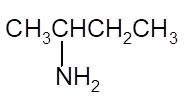
Concept Introduction:The chemical compounds can be shown in different structural formulae. Condense formula, wedge-dashed formula and bond-line formula are some common ways to show the structural of chemical compounds.
In wedge-dashed line formula, the groups or atoms bonded to each C atoms must be shown with dashed and wedges. Here wedge is the solid line that represents the bond in plane of the surface whereas dash line represents the line extended back behind the surface.
In the bond line formula, each C is shown as dot and the each dot is connected with another dot with line. Excluding C and H, all other elements of the compound like N, S, O must be indicted with their atomic symbols. In Kekulè (straight line) notation all atoms and chemical bonds must be shown with any symbol of lone pairs on atoms if any.
(d)
Interpretation: The Kekulè (straight line) notation for the given condensed formula needs to be determined.
Concept Introduction:The chemical compounds can be shown in different structural formulae. Condense formula, wedge-dashed formula and bond-line formula are some common ways to show the structural of chemical compounds.
In wedge-dashed line formula, the groups or atoms bonded to each C atoms must be shown with dashed and wedges. Here wedge is the solid line that represents the bond in plane of the surface whereas dash line represents the line extended back behind the surface.
In the bond line formula, each C is shown as dot and the each dot is connected with another dot with line. Excluding C and H, all other elements of the compound like N, S, O must be indicted with their atomic symbols. In Kekulè (straight line) notation all atoms and chemical bonds must be shown with any symbol of lone pairs on atoms if any.
(e)
Interpretation: The Kekulè (straight line) notation for the given condensed formula needs to be determined.
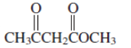
Concept Introduction:The chemical compounds can be shown in different structural formulae. Condense formula, wedge-dashed formula and bond-line formula are some common ways to show the structural of chemical compounds.
In wedge-dashed line formula, the groups or atoms bonded to each C atoms must be shown with dashed and wedges. Here wedge is the solid line that represents the bond in plane of the surface whereas dash line represents the line extended back behind the surface.
In the bond line formula, each C is shown as dot and the each dot is connected with another dot with line. Excluding C and H, all other elements of the compound like N, S, O must be indicted with their atomic symbols. In Kekulè (straight line) notation all atoms and chemical bonds must be shown with any symbol of lone pairs on atoms if any.
(f)
Interpretation: The Kekulè (straight line) notation for the given condensed formula needs to be determined.
Concept Introduction:The chemical compounds can be shown in different structural formulae. Condense formula, wedge-dashed formula and bond-line formula are some common ways to show the structural of chemical compounds.
In wedge-dashed line formula, the groups or atoms bonded to each C atoms must be shown with dashed and wedges. Here wedge is the solid line that represents the bond in plane of the surface whereas dash line represents the line extended back behind the surface.
In the bond line formula, each C is shown as dot and the each dot is connected with another dot with line. Excluding C and H, all other elements of the compound like N, S, O must be indicted with their atomic symbols. In Kekulè (straight line) notation all atoms and chemical bonds must be shown with any symbol of lone pairs on atoms if any.
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
EBK STUDY GUIDE/SOLUTIONS MANUAL FOR OR
- if the answer is no reaction than state that and please hand draw!arrow_forward"I have written solutions in text form, but I need experts to rewrite them in handwriting from A to Z, exactly as I have written, without any changes."arrow_forwardDon't used hand raiting and don't used Ai solutionarrow_forward
- Please correct answer and don't used hand raitingarrow_forwardreciprocal lattices rotates along with the real space lattices of the crystal. true or false?arrow_forwardDeducing the reactants of a Diels-Alder reaction vn the molecule on the right-hand side of this organic reaction be made in good yield from no more than two reactants, in one step, by moderately heating the reactants? ? Δ O If your answer is yes, then draw the reactant or reactants in the drawing area below. You can draw the reactants in any arrangement you like. • If your answer is no, check the box under the drawing area instead. Click and drag to start drawing a structure. Product can't be made in one step. Explanation Checkarrow_forward
- Predict the major products of the following organic reaction: Δ ? Some important notes: • Draw the major product, or products, of the reaction in the drawing area below. • If there aren't any products, because no reaction will take place, check the box below the drawing area instead. • Be sure to use wedge and dash bonds when necessary, for example to distinguish between major products that are enantiomers. Explanation Check Click and drag to start drawing a structure. Larrow_forward> Can the molecule on the right-hand side of this organic reaction be made in good yield from no more than two reactants, in one step, by moderately heating the reactants? ? Δ • If your answer is yes, then draw the reactant or reactants in the drawing area below. You can draw the reactants in any arrangement you like. If your answer is no, check the box under the drawing area instead. Explanation Check Click and drag to start drawing a structure. Х © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accesarrow_forwardPredict the major products of the following organic reaction: O O + A ? Some important notes: • Draw the major product, or products, of the reaction in the drawing area below. • If there aren't any products, because no reaction will take place, check the box below the drawing area instead. • Be sure to use wedge and dash bonds when necessary, for example to distinguish between major products that are enantiomers. Explanation Check Click and drag to start drawing a structure. eserved. Terms of Use | Privacy Center >arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning





