A mixture of dichloromethane (1) and ethyl acetate (2) and is separated in a flash drum at 1 bar. a) Feed rate is 150 kmol/h. Feed enters at 27 mol % dichloromethane and 36% of the feed is vaporized. What are the flowrates and composition of the vapor and liquid fractions? b) Feed rate is 750 lb mol/h. Feed is 20 mol% dichloromethane and a liquid product. of 13 mol % dichloromethane is desired. What V/F must be used? Find product flow rates and composition. c) Feed rate is 200 lb mol/h. Feed is 79 mol% dichloromethane and a vapor product of 6 mol % ethyl acetate is desired. What V/F must be used? Find product flow rates and composition. The equilibrium data for ethyl acetate (1)/dichloromethane (2) is given: Dichloromethane/Ethyl Acetate y (Dichloromethane), mol. frac. 1.0 0.9 0.8 0.7 0.6 0.5 0.4 0.3 0.2 0.1 0.0 0.0 0.1 0.2 0.3 04 0.5 0.6 0.7 0.8 x (Dichloromethane), mol. frac. 0.9 1.0 80 75 70 65 00 T(°C) 8 55 50 45 40 35 0.0 Txy Plot for Dichloromethane/Ethyl Acetate 0.1 0.2 0.3 0.4 0.5 0.6 -Y 0.7 08 0.9 10 x,y (Dichloromethane), mol.frac.
A mixture of dichloromethane (1) and ethyl acetate (2) and is separated in a flash drum at 1 bar. a) Feed rate is 150 kmol/h. Feed enters at 27 mol % dichloromethane and 36% of the feed is vaporized. What are the flowrates and composition of the vapor and liquid fractions? b) Feed rate is 750 lb mol/h. Feed is 20 mol% dichloromethane and a liquid product. of 13 mol % dichloromethane is desired. What V/F must be used? Find product flow rates and composition. c) Feed rate is 200 lb mol/h. Feed is 79 mol% dichloromethane and a vapor product of 6 mol % ethyl acetate is desired. What V/F must be used? Find product flow rates and composition. The equilibrium data for ethyl acetate (1)/dichloromethane (2) is given: Dichloromethane/Ethyl Acetate y (Dichloromethane), mol. frac. 1.0 0.9 0.8 0.7 0.6 0.5 0.4 0.3 0.2 0.1 0.0 0.0 0.1 0.2 0.3 04 0.5 0.6 0.7 0.8 x (Dichloromethane), mol. frac. 0.9 1.0 80 75 70 65 00 T(°C) 8 55 50 45 40 35 0.0 Txy Plot for Dichloromethane/Ethyl Acetate 0.1 0.2 0.3 0.4 0.5 0.6 -Y 0.7 08 0.9 10 x,y (Dichloromethane), mol.frac.
Introduction to Chemical Engineering Thermodynamics
8th Edition
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Chapter1: Introduction
Section: Chapter Questions
Problem 1.1P
Related questions
Question
100%
please answer all. show all the solution. thank you... do not use excel

Transcribed Image Text:A mixture of dichloromethane (1) and ethyl acetate (2) and is separated in a flash drum
at 1 bar.
a) Feed rate is 150 kmol/h. Feed enters at 27 mol% dichloromethane and 36% of
the feed is vaporized. What are the flowrates and composition of the vapor and
liquid fractions?
b) Feed rate is 750 lb mol/h. Feed is 20 mol% dichloromethane and a liquid product
of 13 mol% dichloromethane is desired. What V/F must be used? Find product
flow rates and composition.
c) Feed rate is 200 lb mol/h. Feed is 79 mol% dichloromethane and a vapor product
of 6 mol% ethyl acetate is desired. What V/F must be used? Find product flow
rates and composition.
The equilibrium data for ethyl acetate (1)/dichloromethane (2) is given:
Dichloromethane/Ethyl Acetate
y (Dichloromethane), mol. frac.
1.0
0.9
0.8
0.7
0.6
0.5
0.4
0.3
0.2
0.1
0.0
0.0
0.1
0.2
0.3 0.4 0.5
0.6 0.7 0.8
x (Dichloromethane), mol. frac.
0.9 1.0
2 2 2 2 2 2 2
T(°C)
80
8 8 8
75
65
55
50
45
40
35
0.0
Txy Plot for Dichloromethane/Ethyl Acetate
0.1
0.2
-x-y
0.3 0.4 0.5 0.6 0.7 0.8
x,y (Dichloromethane), mol.frac.
0.9 1.0
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 5 images

Follow-up Questions
Read through expert solutions to related follow-up questions below.
Follow-up Question
please answer all. show all the solution. thank you... do not use excel.,
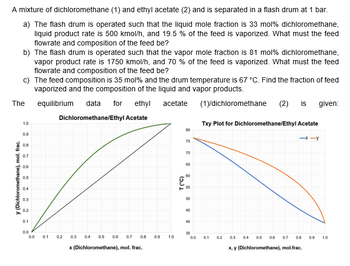
Transcribed Image Text:A mixture of dichloromethane (1) and ethyl acetate (2) and is separated in a flash drum at 1 bar.
a) The flash drum is operated such that the liquid mole fraction is 33 mol % dichloromethane,
liquid product rate is 500 kmol/h, and 19.5 % of the feed is vaporized. What must the feed
flowrate and composition of the feed be?
b) The flash drum is operated such that the vapor mole fraction is 81 mol % dichloromethane,
vapor product rate is 1750 kmol/h, and 70 % of the feed is vaporized. What must the feed
flowrate and composition of the feed be?
c) The feed composition is 35 mol% and the drum temperature is 67 °C. Find the fraction of feed
vaporized and the composition of the liquid and vapor products.
(1)/dichloromethane (2) is given:
equilibrium data for ethyl acetate
Dichloromethane/Ethyl Acetate
The
y (Dichloromethane), mol. frac.
1.0
0.9
0.8
0.7
0.6
0.5
0.4
0.3
0.2
0.1
0.0
0.0
0.1
0.2
0.3 0.4
0.5
0.6
0.7 0.8
x (Dichloromethane), mol. frac.
0.9
1.0
T (°C)
80
75
70
65
60
56
50
45
40
35
0.0
Txy Plot for Dichloromethane/Ethyl Acetate
0.1
0.2
0.3
0.4
0.5
x, y (Dichloromethane), mol.frac.
-x-y
0.6 0.7 0.8
0.9 1.0
Solution
Recommended textbooks for you

Introduction to Chemical Engineering Thermodynami…
Chemical Engineering
ISBN:
9781259696527
Author:
J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:
McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind…
Chemical Engineering
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY

Elements of Chemical Reaction Engineering (5th Ed…
Chemical Engineering
ISBN:
9780133887518
Author:
H. Scott Fogler
Publisher:
Prentice Hall

Introduction to Chemical Engineering Thermodynami…
Chemical Engineering
ISBN:
9781259696527
Author:
J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:
McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind…
Chemical Engineering
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY

Elements of Chemical Reaction Engineering (5th Ed…
Chemical Engineering
ISBN:
9780133887518
Author:
H. Scott Fogler
Publisher:
Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:
9781285061238
Author:
Lokensgard, Erik
Publisher:
Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:
9780072848236
Author:
Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:
McGraw-Hill Companies, The