
Concept explainers
(a)
Interpretation:
The R or S configuration of the chiral center in the given molecule is to be designated.
Concept introduction:
When assigning priorities to substituents, the atom having the greater
When the fourth priority substituent is pointing away (it is attached by a dash bond) and the first, second, and third priority substituents are arranged clockwise, the configuration is R.
When the fourth priority substituent is pointing away (it is attached by a dash bond) and the first, second, and third priority substituents are arranged counterclockwise, the configuration is S.
If the fourth priority substituent is attached by a wedge bond, then the clockwise or counterclockwise arrangement of the first, second, and third priority substituents is determined, and that arrangement is reversed before assigning R or S.
Answer to Problem C.10P
The configuration of the chiral carbon atom in the given molecule is designated as S.
Explanation of Solution
The given molecule is
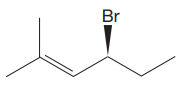
The IUPAC name for the above molecule, without considering the stereochemistry, would be
There is one chiral center in this molecule, and the substituents attached to it are
In the structure, the
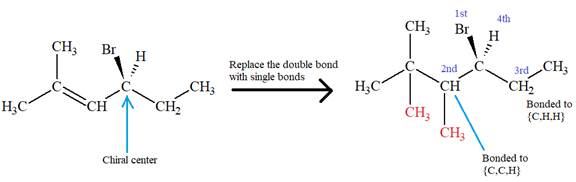
The second and third position priorities are decided by the set of atoms one bond away from the respective points of attachment. The set for the ethyl substituent, which is located on the right, is
The configuration at the chiral center of the molecule is designated as above.
(b)
Interpretation:
The R or S configuration of the chiral center in the given molecule is to be designated.
Concept introduction:
When assigning priorities to substituents, the atom having the greater atomic number has the higher priority. In case of comparison between isotopes, the one having the greater atomic mass gets the higher priority. Substituents with double/triple bonds are treated differently from the substituents having only single bonds. An atom that is doubly bonded to another atom is treated as having two single bonds to the atom – one real (shown in black) and one imaginary (shown in red). An atom that is triply bonded to another atom is treated as having three single bonds to the atom – one real (shown in black) and two imaginary (shown in red).
When the fourth priority substituent is pointing away (it is attached by a dash bond) and the first, second, and third priority substituents are arranged clockwise, the configuration is R.
When the fourth priority substituent is pointing away (it is attached by a dash bond) and the first, second, and third priority substituents are arranged counterclockwise, the configuration is S.
If the fourth priority substituent is attached by a wedge bond, then the clockwise or counterclockwise arrangement of the first, second, and third priority substituents is determined, and that arrangement is reversed before assigning R or S.
If the fourth priority substituent is in the plane of the page, then it is switched with the substituent that points away. Then the clockwise or counterclockwise arrangement of the first, second, and third priority substituents is determined, and that arrangement is reversed before assigning R or S.
Answer to Problem C.10P
The configuration of the chiral carbon atom in the given molecule is designated as R.
Explanation of Solution
The given molecule is
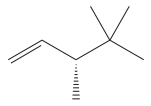
The IUPAC name for the above molecule, without considering the stereochemistry, would be
There is one chiral center in this molecule, and the substituents attached to it are
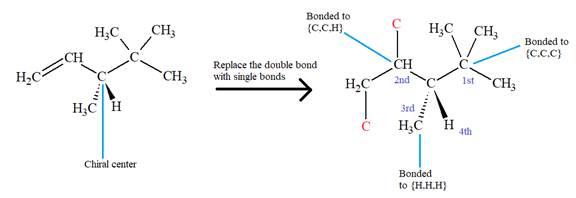
The first three top-priority substituents are decided by the set of atoms one bond away from the respective points of attachment. The set for the substituent, which is located on the right, is
The configuration at the chiral center of the molecule is designated as above.
(c)
Interpretation:
The R or S configuration of the chiral center in the given molecule is to be designated.
Concept introduction:
When assigning priorities to substituents, the atom having the greater atomic number has the higher priority. In case of comparison between isotopes, the one having the greater atomic mass gets the higher priority. Substituents with double/triple bonds are treated differently from the substituents having only single bonds. An atom that is doubly bonded to another atom is treated as having two single bonds to the atom – one real (shown in black) and one imaginary (shown in red). An atom that is triply bonded to another atom is treated as having three single bonds to the atom – one real (shown in black) and two imaginary (shown in red).
When the fourth priority substituent is pointing away (it is attached by a dash bond) and the first, second, and third priority substituents are arranged clockwise, the configuration is R.
When the fourth priority substituent is pointing away (it is attached by a dash bond) and the first, second, and third priority substituents are arranged counterclockwise, the configuration is S.
If the fourth priority substituent is attached by a wedge bond, then the clockwise or counterclockwise arrangement of the first, second, and third priority substituents is determined, and that arrangement is reversed before assigning R or S.
If the fourth priority substituent is in the plane of the page, then it is switched with the substituent that points away. Then the clockwise or counterclockwise arrangement of the first, second, and third priority substituents is determined, and that arrangement is reversed before assigning R or S.
Answer to Problem C.10P
The configuration of the chiral carbon atom in the given molecule is designated as R.
Explanation of Solution
The given molecule is
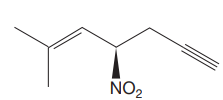
The IUPAC name for the above molecule, without considering the stereochemistry, would be
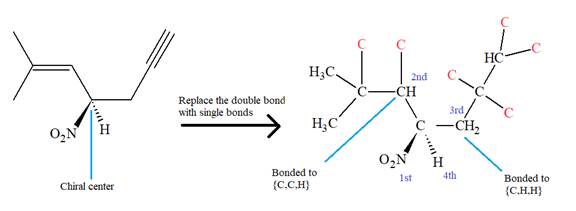
The second and third priority substituents are decided by the set of atoms one bond away from the respective points of attachment. The set for the substituent, which is located on the right, is
The configuration at the chiral center of the molecule is designated as above.
(d)
Interpretation:
The R or S configuration of the chiral center in the given molecule is to be designated.
Concept introduction:
When assigning priorities to substituents, the atom having the greater atomic number has the higher priority. In case of comparison between isotopes, the one having the greater atomic mass gets the higher priority. Substituents with double/triple bonds are treated differently from the substituents having only single bonds. An atom that is doubly bonded to another atom is treated as having two single bonds to the atom – one real (shown in black) and one imaginary (shown in red). An atom that is triply bonded to another atom is treated as having three single bonds to the atom – one real (shown in black) and two imaginary (shown in red).
When the fourth priority substituent is pointing away (it is attached by a dash bond) and the first, second, and third priority substituents are arranged clockwise, the configuration is R.
When the fourth priority substituent is pointing away (it is attached by a dash bond) and the first, second, and third priority substituents are arranged counterclockwise, the configuration is S.
If the fourth priority substituent is attached by a wedge bond, then the clockwise or counterclockwise arrangement of the first, second, and third priority substituents is determined, and that arrangement is reversed before assigning R or S.
If the fourth priority substituent is in the plane of the page, then it is switched with the substituent that points away. Then the clockwise or counterclockwise arrangement of the first, second, and third priority substituents is determined, and that arrangement is reversed before assigning R or S.
Answer to Problem C.10P
The configuration of the chiral carbon atom in the given molecule is designated as R.
Explanation of Solution
The given molecule is
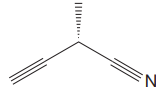
The molecule contains two triple bonds. There is one chiral center in this molecule, and the substituents attached to it are
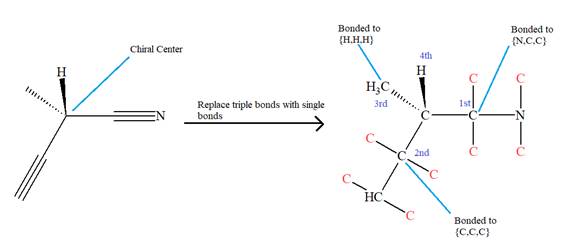
The top three-priorities are decided by the set of atoms one bond away from the respective points of attachment.
The set for the substituent, which is located on the right, is
Given that these substituents are arranged in the counterclockwise manner, but since the fourth-priority substituent is attached by a wedge bond, the arrangement is considered as reverse, that is, clockwise, and the configuration at the chiral center is R. Thus, the configuration at the chiral center for the molecule is R.
The configuration at the chiral center of the molecule is designated as above.
(e)
Interpretation:
The R or S configuration of the chiral center in the given molecule is to be designated.
Concept introduction:
When assigning priorities to substituents, the atom having the greater atomic number has the higher priority. In case of comparison between isotopes, the one having the greater atomic mass gets the higher priority. Substituents with double/triple bonds are treated differently from the substituents having only single bonds. An atom that is doubly bonded to another atom is treated as having two single bonds to the atom – one real (shown in black) and one imaginary (shown in red). An atom that is triply bonded to another atom is treated as having three single bonds to the atom – one real (shown in black) and two imaginary (shown in red).
When the fourth priority substituent is pointing away (it is attached by a dash bond) and the first, second, and third priority substituents are arranged clockwise, the configuration is R.
When the fourth priority substituent is pointing away (it is attached by a dash bond) and the first, second, and third priority substituents are arranged counterclockwise, the configuration is S.
If the fourth priority substituent is attached by a wedge bond, then the clockwise or counterclockwise arrangement of the first, second, and third priority substituents is determined, and that arrangement is reversed before assigning R or S.
If the fourth priority substituent is in the plane of the page, then it is switched with the substituent that points away. Then the clockwise or counterclockwise arrangement of the first, second, and third priority substituents is determined, and that arrangement is reversed before assigning R or S.
Answer to Problem C.10P
The configuration of the chiral carbon atom in the given molecule is designated as R.
Explanation of Solution
The given molecule is
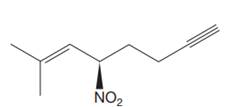
The molecule contains one double bond and one triple bond. There is one chiral center in this molecule, and the substituents attached to it are
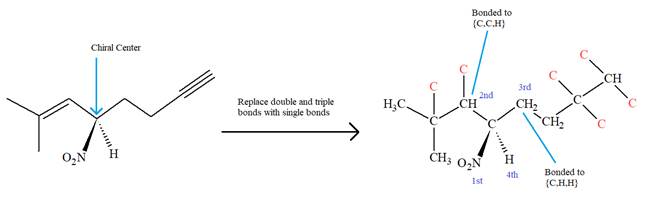
The second and third priority substituents are decided by the set of atoms one bond away from the respective points of attachment. The set for the substituent, which is located on the right, is
The configuration at the chiral center of the molecule is designated as above.
Want to see more full solutions like this?
Chapter C Solutions
EBK ORGANIC CHEMISTRY: PRINCIPLES AND M
- In the phase diagram of steel (two components Fe and C), region A is the gamma austenite solid and region B contains the gamma solid and liquid. Indicate the degrees of freedom that the fields A and B have,arrow_forwardFor a condensed binary system in equilibrium at constant pressure, indicate the maximum number of phases that can exist.arrow_forwardPart V. Label ad match the carbons in compounds Jane and Diane w/ the corresponding peak no. in the Spectra (Note: use the given peak no. To label the carbons, other peak no are intentionally omitted) 7 4 2 -0.13 -0.12 -0.11 -0.10 -0.08 8 CI Jane 1 -0.09 5 210 200 190 180 170 160 150 140 130 120 110 100 -8 90 f1 (ppm) 11 8 172.4 172.0 f1 (ppr HO CI NH Diane 7 3 11 80 80 -80 -R 70 60 60 2 5 -8 50 40 8. 170 160 150 140 130 120 110 100 90 -0 80 70 20 f1 (ppm) 15 30 -20 20 -60 60 -0.07 -0.06 -0.05 -0.04 -0.03 -0.02 -0.01 -0.00 -0.01 10 -0.17 16 15 56 16 -0.16 -0.15 -0.14 -0.13 -0.12 -0.11 -0.10 -0.09 -0.08 -0.07 -0.06 -0.05 -0.04 17.8 17.6 17.4 17.2 17.0 f1 (ppm) -0.03 -0.02 550 106 40 30 20 20 -0.01 -0.00 F-0.01 10 0arrow_forward
- n Feb 3 A T + 4. (2 pts) Draw the structure of the major component of the Limonene isolated. Explain how you confirmed the structure. 5. (2 pts) Draw the fragment corresponding to the base peak in the Mass spectrum of Limonene. 6. (1 pts) Predict the 1H NMR spectral data of R-Limonene. Proton NMR: 5.3 pon multiplet (H Ringarrow_forwardPart VI. Ca H 10 O is the molecular formula of compound Tom and gives the in the table below. Give a possible structure for compound Tom. 13C Signals summarized C1 C2 C3 C4 C5 C6 C7 13C shift (ppm) 23.5 27.0 33.0 35.8 127 162 205 DEPT-90 + DEPT-135 + +arrow_forward2. Using the following data to calculate the value of AvapH o of water at 298K. AvapH o of water at 373K is 40.7 kJ/mol; molar heat capacity of liquid water at constant pressure is 75.2J mol-1 K-1 and molar heat capacity of water vapor at constant pressure is 33.6 J mol-1 K-1.arrow_forward
- Part VII. Below are the 'HNMR 13 3 C-NMR, COSY 2D- NMR, and HSQC 20-NMR (Similar with HETCOR but axes are reversed) spectra of an organic compound with molecular formula C6H13 O. Assign chemical shift values to the H and c atoms of the compound. Find the structure. Show complete solutions. Predicted 1H NMR Spectrum ли 4.7 4.6 4.5 4.4 4.3 4.2 4.1 4.0 3.9 3.8 3.7 3.6 3.5 3.4 3.3 3.2 3.1 3.0 2.9 2.8 2.7 2.6 2.5 2.4 2.3 2.2 2.1 2.0 1.9 1.8 1.7 1.6 1.5 1.4 1.3 1.2 1.1 1.0 0.9 0.8 f1 (ppm)arrow_forward3. Draw the expanded structural formula, the condensed structural formula, and the skeletal structural formula for 2-pentene. expanded structure: Condensed structure: Skeletal formula: 4. Draw the expanded structural formula, the condensed structural formula, and the skeletal structural formula for 2-methyl-3-heptene. expanded structure: Condensed structure: Skeletal formula: following structurearrow_forwardPart IV. Propose a plausible Structure w/ the following descriptions: a) A 5-carbon hydrocarbon w/ a single peak in its proton decoupled the DEPT-135 Spectrum shows a negative peak C-NMR spectrum where b) what cyclohexane dione isomer gives the largest no. Of 13C NMR signals? c) C5H120 (5-carbon alcohol) w/ most deshielded carbon absent in any of its DEPT Spectivaarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
