
Concept explainers
(a)
Interpretation:
The R or S configuration of the chiral center in the given molecule is to be designated.
Concept introduction:
When assigning priorities to substituents, the atom having the greater
When the fourth priority substituent is pointing away (it is attached by a dash bond) and the first, second, and third priority substituents are arranged clockwise, the configuration is R.
When the fourth priority substituent is pointing away (it is attached by a dash bond) and the first, second, and third priority substituents are arranged counterclockwise, the configuration is S.
If the fourth priority substituent is attached by a wedge bond, then the clockwise or counterclockwise arrangement of the first, second, and third priority substituents is determined, and that arrangement is reversed before assigning R or S.
Answer to Problem C.10P
The configuration of the chiral carbon atom in the given molecule is designated as S.
Explanation of Solution
The given molecule is
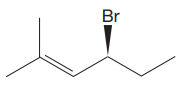
The IUPAC name for the above molecule, without considering the stereochemistry, would be
There is one chiral center in this molecule, and the substituents attached to it are
In the structure, the
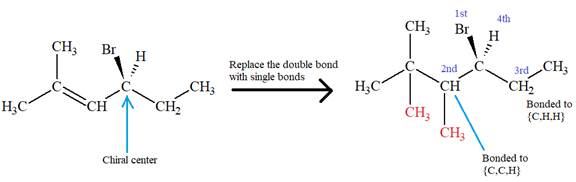
The second and third position priorities are decided by the set of atoms one bond away from the respective points of attachment. The set for the ethyl substituent, which is located on the right, is
The configuration at the chiral center of the molecule is designated as above.
(b)
Interpretation:
The R or S configuration of the chiral center in the given molecule is to be designated.
Concept introduction:
When assigning priorities to substituents, the atom having the greater atomic number has the higher priority. In case of comparison between isotopes, the one having the greater atomic mass gets the higher priority. Substituents with double/triple bonds are treated differently from the substituents having only single bonds. An atom that is doubly bonded to another atom is treated as having two single bonds to the atom – one real (shown in black) and one imaginary (shown in red). An atom that is triply bonded to another atom is treated as having three single bonds to the atom – one real (shown in black) and two imaginary (shown in red).
When the fourth priority substituent is pointing away (it is attached by a dash bond) and the first, second, and third priority substituents are arranged clockwise, the configuration is R.
When the fourth priority substituent is pointing away (it is attached by a dash bond) and the first, second, and third priority substituents are arranged counterclockwise, the configuration is S.
If the fourth priority substituent is attached by a wedge bond, then the clockwise or counterclockwise arrangement of the first, second, and third priority substituents is determined, and that arrangement is reversed before assigning R or S.
If the fourth priority substituent is in the plane of the page, then it is switched with the substituent that points away. Then the clockwise or counterclockwise arrangement of the first, second, and third priority substituents is determined, and that arrangement is reversed before assigning R or S.
Answer to Problem C.10P
The configuration of the chiral carbon atom in the given molecule is designated as R.
Explanation of Solution
The given molecule is
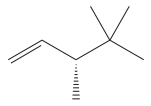
The IUPAC name for the above molecule, without considering the stereochemistry, would be
There is one chiral center in this molecule, and the substituents attached to it are
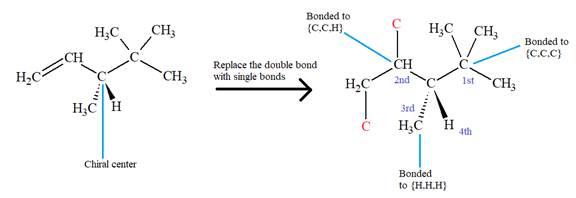
The first three top-priority substituents are decided by the set of atoms one bond away from the respective points of attachment. The set for the substituent, which is located on the right, is
The configuration at the chiral center of the molecule is designated as above.
(c)
Interpretation:
The R or S configuration of the chiral center in the given molecule is to be designated.
Concept introduction:
When assigning priorities to substituents, the atom having the greater atomic number has the higher priority. In case of comparison between isotopes, the one having the greater atomic mass gets the higher priority. Substituents with double/triple bonds are treated differently from the substituents having only single bonds. An atom that is doubly bonded to another atom is treated as having two single bonds to the atom – one real (shown in black) and one imaginary (shown in red). An atom that is triply bonded to another atom is treated as having three single bonds to the atom – one real (shown in black) and two imaginary (shown in red).
When the fourth priority substituent is pointing away (it is attached by a dash bond) and the first, second, and third priority substituents are arranged clockwise, the configuration is R.
When the fourth priority substituent is pointing away (it is attached by a dash bond) and the first, second, and third priority substituents are arranged counterclockwise, the configuration is S.
If the fourth priority substituent is attached by a wedge bond, then the clockwise or counterclockwise arrangement of the first, second, and third priority substituents is determined, and that arrangement is reversed before assigning R or S.
If the fourth priority substituent is in the plane of the page, then it is switched with the substituent that points away. Then the clockwise or counterclockwise arrangement of the first, second, and third priority substituents is determined, and that arrangement is reversed before assigning R or S.
Answer to Problem C.10P
The configuration of the chiral carbon atom in the given molecule is designated as R.
Explanation of Solution
The given molecule is
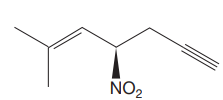
The IUPAC name for the above molecule, without considering the stereochemistry, would be
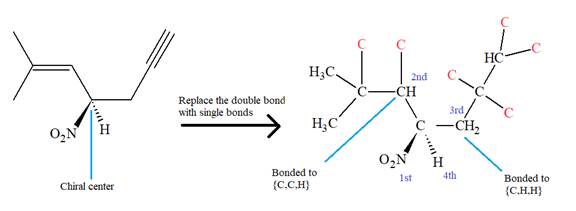
The second and third priority substituents are decided by the set of atoms one bond away from the respective points of attachment. The set for the substituent, which is located on the right, is
The configuration at the chiral center of the molecule is designated as above.
(d)
Interpretation:
The R or S configuration of the chiral center in the given molecule is to be designated.
Concept introduction:
When assigning priorities to substituents, the atom having the greater atomic number has the higher priority. In case of comparison between isotopes, the one having the greater atomic mass gets the higher priority. Substituents with double/triple bonds are treated differently from the substituents having only single bonds. An atom that is doubly bonded to another atom is treated as having two single bonds to the atom – one real (shown in black) and one imaginary (shown in red). An atom that is triply bonded to another atom is treated as having three single bonds to the atom – one real (shown in black) and two imaginary (shown in red).
When the fourth priority substituent is pointing away (it is attached by a dash bond) and the first, second, and third priority substituents are arranged clockwise, the configuration is R.
When the fourth priority substituent is pointing away (it is attached by a dash bond) and the first, second, and third priority substituents are arranged counterclockwise, the configuration is S.
If the fourth priority substituent is attached by a wedge bond, then the clockwise or counterclockwise arrangement of the first, second, and third priority substituents is determined, and that arrangement is reversed before assigning R or S.
If the fourth priority substituent is in the plane of the page, then it is switched with the substituent that points away. Then the clockwise or counterclockwise arrangement of the first, second, and third priority substituents is determined, and that arrangement is reversed before assigning R or S.
Answer to Problem C.10P
The configuration of the chiral carbon atom in the given molecule is designated as R.
Explanation of Solution
The given molecule is
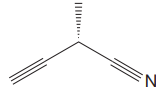
The molecule contains two triple bonds. There is one chiral center in this molecule, and the substituents attached to it are
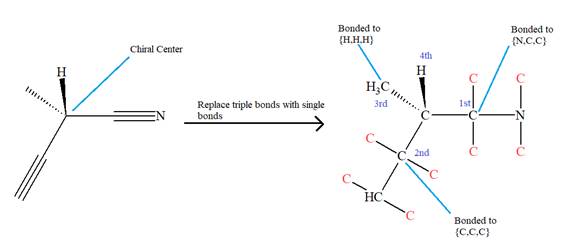
The top three-priorities are decided by the set of atoms one bond away from the respective points of attachment.
The set for the substituent, which is located on the right, is
Given that these substituents are arranged in the counterclockwise manner, but since the fourth-priority substituent is attached by a wedge bond, the arrangement is considered as reverse, that is, clockwise, and the configuration at the chiral center is R. Thus, the configuration at the chiral center for the molecule is R.
The configuration at the chiral center of the molecule is designated as above.
(e)
Interpretation:
The R or S configuration of the chiral center in the given molecule is to be designated.
Concept introduction:
When assigning priorities to substituents, the atom having the greater atomic number has the higher priority. In case of comparison between isotopes, the one having the greater atomic mass gets the higher priority. Substituents with double/triple bonds are treated differently from the substituents having only single bonds. An atom that is doubly bonded to another atom is treated as having two single bonds to the atom – one real (shown in black) and one imaginary (shown in red). An atom that is triply bonded to another atom is treated as having three single bonds to the atom – one real (shown in black) and two imaginary (shown in red).
When the fourth priority substituent is pointing away (it is attached by a dash bond) and the first, second, and third priority substituents are arranged clockwise, the configuration is R.
When the fourth priority substituent is pointing away (it is attached by a dash bond) and the first, second, and third priority substituents are arranged counterclockwise, the configuration is S.
If the fourth priority substituent is attached by a wedge bond, then the clockwise or counterclockwise arrangement of the first, second, and third priority substituents is determined, and that arrangement is reversed before assigning R or S.
If the fourth priority substituent is in the plane of the page, then it is switched with the substituent that points away. Then the clockwise or counterclockwise arrangement of the first, second, and third priority substituents is determined, and that arrangement is reversed before assigning R or S.
Answer to Problem C.10P
The configuration of the chiral carbon atom in the given molecule is designated as R.
Explanation of Solution
The given molecule is
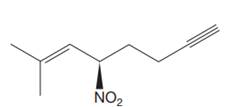
The molecule contains one double bond and one triple bond. There is one chiral center in this molecule, and the substituents attached to it are
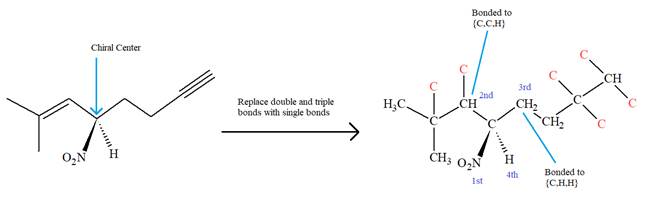
The second and third priority substituents are decided by the set of atoms one bond away from the respective points of attachment. The set for the substituent, which is located on the right, is
The configuration at the chiral center of the molecule is designated as above.
Want to see more full solutions like this?
Chapter C Solutions
EBK ORGANIC CHEMISTRY: PRINCIPLES AND M
- curved arrows are used to illustrate the flow of electrons. using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s)arrow_forwardIdentify the 'cartoon' drawing of the acceptor orbital in the first mechanistic step of an electrophilic addition reaction of butadiene with HBr. Pleasearrow_forwardH- H H H H H H Identify and select all structures below that represent a constitutional isomer(s) of the compound shown above. H- H H H A. H H H H-C CI H H D. H H H H H H C C -H H C C H H H H B. H CI H H- C C H H H H E. H CI H C.arrow_forward
- Why doesn't this carry on to form a ring by deprotonating the alpha carbon and the negatively-charged carbon attacking the C=O?arrow_forward6. A solution (0.0004 M) of Fe(S2CNEt2)3 (see the structural drawing below) in chloroform has absorption bands at: 350 nm (absorbance A = 2.34); 514 nm(absorbance A = 0.0532); Calculate the molar absorptivity values for these bands. Comment on their possible nature (charge transfer transitions or d-d S N- transitions?). (4 points)arrow_forwardWhat is the mechanism for this?arrow_forward
- For questions 1-4, consider the following complexes: [Co(CN)6], [COC14]², [Cr(H2O)6]²+ 4. Room temperature (20°C) measurement of molar magnetic susceptibility (Xm) for Fe(NH4)2(SO4)2×6H2O is 1.1888 x 102 cgs (Gaussian units). Calculate effective magnetic moment and provide a number of unpaired electrons for the iron ion. Use this number to rationalize the coordination geometry around iron center. (4 points)arrow_forward7. Describe the expected 31P and 19F (where applicable) NMR spectral patterns for the following compounds (indicate number of signals and their splitting patterns). a) tetraphenyldiphosphine Ph Ph P-P Ph Ph Ph Ph ' b) tetraphenyldiphosphine monoxide P-P-Ph Ph (2 points) (2 points c) tetrafluorophosphonium hexafluorophosphate [PF4]*[PF6]¯ (4 points)arrow_forward3. For questions 1-4, consider the following complexes: [Co(CN)6]4, [COC14]², [Cr(H2O)6]²+ Which (if any) of these complexes would be expected to display Jahn-Teller distortion? (2 points)arrow_forward
- What is Instrumental Neutron Activation and what are the advantages and disadvantages in using its applications? (I'm doing an in class assignment and need better understanding of what the instrument can be used for) Please include references so that I can better understand the application of how the instrument works!arrow_forwardWhat is Isotope Analysis and what are the advantages and disadvantages in using its applications and instrumentalization? Please include references so that I can better understand how the instrument works!arrow_forward5. Count the electrons on the following complexes and state whether they follow the 18- electron rule: (3 points) Fe(CO)5 Ni(PMe3)4 PMe3 is trimethylphosphine Mn(CO)5Brarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
