
Concept explainers
a.
Interpretation: The molecules that contain polar bonds; the polar molecules and the non-polar molecules from the given molecules are to be identified.
Concept introduction: Polarity in bond arises due to the difference in electronegativity of the bonded atoms.
If the net dipole moment of the molecule is zero, the molecule is non-polar.
If the molecule has some net dipole moment than the molecule is polar.
The overall polarity of the molecule is determined by calculating the vector sum of the individual bond polarities.
To identify: The molecules that contain polar bonds.
a.
Answer to Problem 9.45QP
Solution
All the molecules contain polar bonds in there structure.
Explanation of Solution
Explanation
Polarity in bond arises due to the difference in electronegativity of the bonded atoms.
For molecule (a)
In 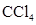 , there is difference in electronegativity of carbon and chlorine atoms because chlorine is more electronegative than carbon.
, there is difference in electronegativity of carbon and chlorine atoms because chlorine is more electronegative than carbon.
Therefore, the bonds in 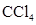 is polar.
is polar.
For molecule (b)
In 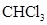 , there is difference in electronegativity of carbon and hydrogen atom because carbon is more electronegative than hydrogen.
, there is difference in electronegativity of carbon and hydrogen atom because carbon is more electronegative than hydrogen.
Therefore, the 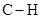 bond in
bond in 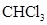 is polar.
is polar.
In 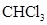 , there is difference in electronegativity of carbon and chlorine atom because chlorine is more electronegative than carbon.
, there is difference in electronegativity of carbon and chlorine atom because chlorine is more electronegative than carbon.
Therefore, the 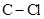 bond in
bond in 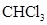 is polar.
is polar.
For molecule (c)
In 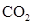 , there is difference in electronegativity of carbon and oxygen atoms because oxygen is more electronegative than carbon.
, there is difference in electronegativity of carbon and oxygen atoms because oxygen is more electronegative than carbon.
Therefore, the 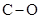 bonds in
bonds in 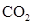 is polar.
is polar.
For molecule (d)
In 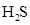 , there is difference in electronegativity of sulphur and hydrogen atom because sulphur is more electronegative than hydrogen.
, there is difference in electronegativity of sulphur and hydrogen atom because sulphur is more electronegative than hydrogen.
Therefore, the 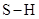 bond in
bond in 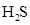 is polar.
is polar.
For molecule (e)
In 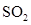 , there is difference in electronegativity of sulphur and oxygen atoms because oxygen is more electronegative than sulphur.
, there is difference in electronegativity of sulphur and oxygen atoms because oxygen is more electronegative than sulphur.
Therefore, the 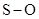 bond in
bond in 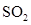 is polar.
is polar.
b.
To identify: The molecules that are polar.
b.
Answer to Problem 9.45QP
Solution
The polar molecules are 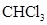 ,
,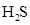 and
and 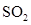 .
.
Explanation of Solution
Explanation
Polarity in bond arises due to the difference in electronegativity of the bonded atoms.
If the net dipole moment of the molecule is zero, the molecule is non polar in nature.
If the molecule has some net dipole moment than the molecule is polar in nature.
The overall polarity of the molecule is determined by calculating the vector sum of the individual bond polarities.
For molecule (a)
In 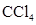 , there is difference in electronegativity of carbon and chlorine atoms because chlorine is more electronegative than carbon.
, there is difference in electronegativity of carbon and chlorine atoms because chlorine is more electronegative than carbon.
Therefore, the bonds in 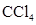 are polar.
are polar.
The dipole moment of 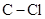 bonds get cancelled out to give zero net dipole moment to the molecule.
bonds get cancelled out to give zero net dipole moment to the molecule.
Therefore, the 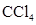 is non polar.
is non polar.
For molecule (b)
In 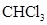 , there is difference in electronegativity of carbon and hydrogen atom because carbon is more electronegative than hydrogen.
, there is difference in electronegativity of carbon and hydrogen atom because carbon is more electronegative than hydrogen.
Therefore, the 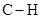 bond in
bond in 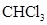 is polar.
is polar.
In 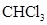 , there is difference in electronegativity of carbon and chlorine atom because chlorine is more electronegative than carbon.
, there is difference in electronegativity of carbon and chlorine atom because chlorine is more electronegative than carbon.
Therefore, the 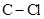 bonds in
bonds in 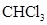 is polar.
is polar.
The sum of dipole moment of one 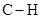 bond and three
bond and three 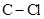 give a net dipole moment to the molecule.
give a net dipole moment to the molecule.
Therefore, the 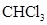 molecule is polar.
molecule is polar.
For molecule (c)
In 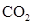 , there is difference in electronegativity of carbon and oxygen atoms because oxygen is more electronegative than carbon.
, there is difference in electronegativity of carbon and oxygen atoms because oxygen is more electronegative than carbon.
Therefore, the 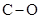 bonds in
bonds in 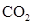 is polar.
is polar.
The dipole moment of 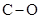 bonds get cancelled out to give zero net dipole moment to the molecule.
bonds get cancelled out to give zero net dipole moment to the molecule.
Therefore, the 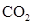 is non polar.
is non polar.
For molecule (d)
In 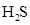 , there is difference in electronegativity of sulphur and hydrogen atom because sulphur is more electronegative than hydrogen.
, there is difference in electronegativity of sulphur and hydrogen atom because sulphur is more electronegative than hydrogen.
Therefore, the 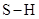 bondS in
bondS in 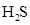 is polar.
is polar.
The sum of dipole moment of two 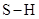 bonds gives a net dipole moment to the molecule.
bonds gives a net dipole moment to the molecule.
Therefore, the 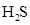 molecule is polar.
molecule is polar.
For molecule (e)
In 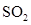 , there is difference in electronegativity of sulphur and oxygen atoms because oxygen is more electronegative than sulphur.
, there is difference in electronegativity of sulphur and oxygen atoms because oxygen is more electronegative than sulphur.
Therefore, the 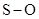 bonds in
bonds in 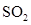 is polar.
is polar.
The structure of 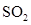 is bent. The sum of dipole moment of two
is bent. The sum of dipole moment of two 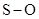 bonds gives a net dipole moment to the molecule.
bonds gives a net dipole moment to the molecule.
Therefore, the 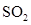 molecule is polar.
molecule is polar.
c.
To identify: The molecules that are non polar.
c.
Answer to Problem 9.45QP
Solution
The polar molecules are 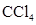 and
and 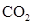 .
.
Explanation of Solution
Explanation
Polarity in bond arises due to the difference in electronegativity of the bonded atoms.
If the net dipole moment of the molecule is zero, the molecule is non polar in nature.
If the molecule has some net dipole moment than the molecule is polar in nature.
The overall polarity of the molecule is determined by calculating the vector sum of the individual bond polarities.
For molecule (a)
In 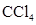 , there is difference in electronegativity of carbon and chlorine atoms because chlorine is more electronegative than carbon.
, there is difference in electronegativity of carbon and chlorine atoms because chlorine is more electronegative than carbon.
Therefore, the bonds in 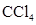 is polar.
is polar.
The dipole moment of 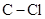 bonds get cancelled out to give zero net dipole moment to the molecule.
bonds get cancelled out to give zero net dipole moment to the molecule.
Therefore, the 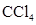 is non polar.
is non polar.
For molecule (b)
In 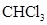 , there is difference in electronegativity of carbon and hydrogen atom because carbon is more electronegative than hydrogen.
, there is difference in electronegativity of carbon and hydrogen atom because carbon is more electronegative than hydrogen.
Therefore, the 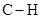 bond in
bond in 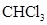 is polar.
is polar.
In 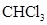 , there is difference in electronegativity of carbon and chlorine atom because chlorine is more electronegative than carbon.
, there is difference in electronegativity of carbon and chlorine atom because chlorine is more electronegative than carbon.
Therefore, the 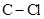 bonds in
bonds in 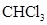 is polar.
is polar.
The sum of dipole moment of one 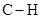 bond and three
bond and three 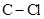 give a net dipole moment to the molecule.
give a net dipole moment to the molecule.
Therefore, the 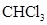 molecule is polar.
molecule is polar.
For molecule (c)
In 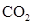 , there is difference in electronegativity of carbon and oxygen atoms because oxygen is more electronegative than carbon.
, there is difference in electronegativity of carbon and oxygen atoms because oxygen is more electronegative than carbon.
Therefore, the 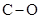 bonds in
bonds in 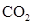 is polar.
is polar.
The dipole moment of 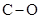 bonds get cancelled out to give zero net dipole moment to the molecule.
bonds get cancelled out to give zero net dipole moment to the molecule.
Therefore, the 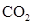 is non polar.
is non polar.
For molecule (d)
In 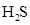 , there is difference in electronegativity of sulphur and hydrogen atom because sulphur is more electronegative than hydrogen.
, there is difference in electronegativity of sulphur and hydrogen atom because sulphur is more electronegative than hydrogen.
Therefore, the 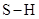 bond in
bond in 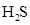 is polar.
is polar.
The sum of dipole moment of two 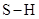 bonds gives a net dipole moment to the molecule.
bonds gives a net dipole moment to the molecule.
Therefore, the 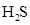 molecule is polar.
molecule is polar.
For molecule (e)
In 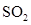 , there is difference in electronegativity of sulphur and oxygen atoms because oxygen is more electronegative than sulphur.
, there is difference in electronegativity of sulphur and oxygen atoms because oxygen is more electronegative than sulphur.
Therefore, the 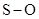 bonds in
bonds in 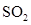 is polar.
is polar.
The structure of 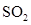 is bent. The sum of dipole moment of two
is bent. The sum of dipole moment of two 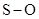 bonds gives a net dipole moment to the molecule.
bonds gives a net dipole moment to the molecule.
Therefore, the 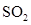 molecule is polar.
molecule is polar.
Conclusion
- a. All the molecules contains polar bonds in there structure.
- b. The polar molecules are
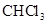 ,
, 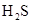 and
and 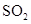 .
. - c. The polar molecules are
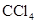 and
and 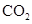 .
.
Want to see more full solutions like this?
Chapter 9 Solutions
Chemistry
- 4. For the following complexes, draw the structures and give a d-electron count of the metal: a) Tris(acetylacetonato)iron(III) b) Hexabromoplatinate(2-) c) Potassium diamminetetrabromocobaltate(III) (6 points)arrow_forward2. Calculate the overall formation constant for [Fe(CN)6]³, given that the overall formation constant for [Fe(CN)6] 4 is ~1032, and that: Fe3+ (aq) + e = Fe²+ (aq) E° = +0.77 V [Fe(CN)6]³ (aq) + e¯ = [Fe(CN)6] (aq) E° = +0.36 V (4 points)arrow_forward5. Consider the compounds shown below as ligands in coordination chemistry and identify their denticity; comment on their ability to form chelate complexes. (6 points) N N A B N N N IN N Carrow_forward
- 1. Use standard reduction potentials to rationalize quantitatively why: (6 points) (a) Al liberates H2 from dilute HCl, but Ag does not; (b) Cl2 liberates Br2 from aqueous KBr solution, but does not liberate C12 from aqueous KCl solution; c) a method of growing Ag crystals is to immerse a zinc foil in an aqueous solution of AgNO3.arrow_forwardWhat would be the best choices for the missing reagents 1 and 3 in this synthesis? 1 1. PPh3 2. n-BuLi 3 2 • Draw the missing reagents in the drawing area below. You can draw them in any arrangement you like. • Do not draw the missing reagent 2. If you draw 1 correctly, we'll know what it is. • Note: if one of your reagents needs to contain a halogen, use bromine. Click and drag to start drawing a structure. Xarrow_forwardWhat is the missing reactant R in this organic reaction? N N H3O+ +R + • Draw the structure of R in the drawing area below. • Be sure to use wedge and dash bonds if it's necessary to draw one particular enantiomer. Click and drag to start drawing a structure. fmarrow_forward
- The product on the right-hand side of this reaction can be prepared from two organic reactants, under the conditions shown above and below the arrow. Draw 1 and 2 below, in any arrangement you like. 1+2 NaBH3CN H+ N Click and drag to start drawing a structure. 5arrow_forwardAssign this HSQC Spectrum ( please editing clearly on the image)arrow_forward(a 4 shows scanning electron microscope (SEM) images of extruded actions of packing bed for two capillary columns of different diameters, al 750 (bottom image) and b) 30-μm-i.d. Both columns are packed with the same stationary phase, spherical particles with 1-um diameter. A) When the columns were prepared, the figure shows that the column with the larger diameter has more packing irregularities. Explain this observation. B) Predict what affect this should have on band broadening and discuss your prediction using the van Deemter terms. C) Does this figure support your explanations in application question 33? Explain why or why not and make any changes in your answers in light of this figure. Figure 4 SEM images of sections of packed columns for a) 750 and b) 30-um-i.d. capillary columns.³arrow_forward
- fcrip = ↓ bandwidth Il temp 32. What impact (increase, decrease, or no change) does each of the following conditions have on the individual components of the van Deemter equation and consequently, band broadening? Increase temperature Longer column Using a gas mobile phase instead of liquid Smaller particle stationary phase Multiple Paths Diffusion Mass Transferarrow_forward34. Figure 3 shows Van Deemter plots for a solute molecule using different column inner diameters (i.d.). A) Predict whether decreasing the column inner diameters increase or decrease bandwidth. B) Predict which van Deemter equation coefficient (A, B, or C) has the greatest effect on increasing or decreasing bandwidth as a function of i.d. and justify your answer. Figure 3 Van Deemter plots for hydroquinone using different column inner diameters (i.d. in μm). The data was obtained from liquid chromatography experiments using fused-silica capillary columns packed with 1.0-μm particles. 35 20 H(um) 큰 20 15 90 0+ 1500 100 75 550 01 02 594 05 μ(cm/sec) 30 15 10arrow_forwardelow are experimentally determined van Deemter plots of column efficiency, H, vs. flow rate. H is a quantitative measurement of band broadening. The left plot is for a liquid chromatography application and the night is for gas chromatography. Compare and contrast these two plots in terms of the three band broadening mechanisms presented in this activity. How are they similar? How do they differ? Justify your answers.? 0.4 H (mm) 0.2 0.1- 0.3- 0 0.5 H (mm) 8.0 7.0 6.0 5.0 4.0- 3.0 T +++ 1.0 1.5 0 2.0 4.0 Flow Rate, u (cm/s) 6.0 8.0 Flow Rate, u (cm/s)arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





