
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
3rd Edition
ISBN: 9781259298424
Author: SMITH
Publisher: VST
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 9.112P
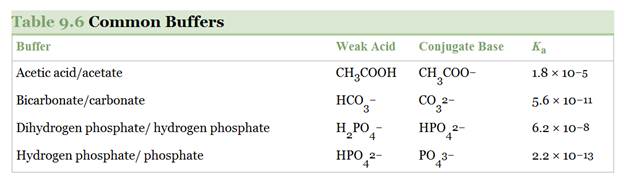
Using the
a.
b.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
None
Q1: For each molecule, assign each stereocenter as R or S. Circle the meso compounds. Label
each compound as chiral or achiral.
+
CI
Br
:
Н
OH
H
wo་ཡིག་ཐrow
HO
3
D
။။ဂ
CI
Br
H,
CI
Br
Br H₂N
OMe
R
IN
I
I
N
S
H
Br
ជ័យ
CI
CI
D
OH
Please correct answer and don't use hand rating
Chapter 9 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
Ch. 9.1 - Name each acid: (a)HF;(b)HNO3;(c)HCN.Ch. 9.1 - If the polyatomic anion C1O2- is called chlorite,...Ch. 9.1 - Which of the following species can be...Ch. 9.1 - Which of the following species can be...Ch. 9.1 - Classify each reactant as a Brønsted-Lowry acid or...Ch. 9.2 - Determine the conjugate acid of each species:...Ch. 9.2 - Determine the conjugate base of each species:...Ch. 9.2 - Draw the structure of the conjugate base of each...Ch. 9.2 - Label the acid and the base and the conjugate acid...Ch. 9.2 - Ammonia, NH3, is amphoteric. Draw the conjugate...
Ch. 9.2 - Prob. 9.11PCh. 9.3 - Diagrams represent three acids (HA) dissolved in...Ch. 9.3 - Label the stronger acid in each pair. Which acid...Ch. 9.3 - Are the reactants or products favored at...Ch. 9.3 - If lactic acid is similar in strength to acetic...Ch. 9.4 - Rank the acids in each group in order of...Ch. 9.4 - Use the acid dissociation constants in Table 9.3...Ch. 9.4 - Consider the weak acids, HCN and H2CO3. Which acid...Ch. 9.5 - Calculate the value of [OH-] from the given [H3O+]...Ch. 9.5 - Calculate the value of [H3O+] from the given [OH-]...Ch. 9.5 - Calculate the value of [H3O+] and [H3O-] in each...Ch. 9.6 - Convert each H3O+ concentration to a pH value. a....Ch. 9.6 - What H3O+ concentration corresponds to each pH...Ch. 9.6 - Prob. 9.24PCh. 9.6 - Convert each H3O+ concentration to a pH value....Ch. 9.6 - Prob. 9.26PCh. 9.7 - Write a balanced equation for each acid-base...Ch. 9.7 - Prob. 9.28PCh. 9.7 - The acid in acid rain is generally sulfuric acid...Ch. 9.7 - Write a balanced equation for the reaction of...Ch. 9.8 - Determine whether each salt forms an acidic,...Ch. 9.8 - Which of the following salts forms an aqueous...Ch. 9.9 - What is the molarity of an HCI solution if 25.5 mL...Ch. 9.9 - How many milliliters of 2.0MNaOH are needed to...Ch. 9.10 - Determine whether a solution containing each of...Ch. 9.10 - Consider a buffer prepared from the weak acid HCO3...Ch. 9.10 - Calculate the pH of a dihydrogen...Ch. 9.10 - What is the pH of a buffer that contains...Ch. 9 - Which of the following species can be...Ch. 9 - Which of the following species can be...Ch. 9 - Prob. 9.41PCh. 9 - Which of the following species can be...Ch. 9 - Prob. 9.43PCh. 9 - Draw the conjugate acid of each base. a. Br- b....Ch. 9 - Draw the conjugate base of each acid. HNO2 NH4+...Ch. 9 - Draw the conjugate base of each acid. H3O+ H2Se...Ch. 9 - Prob. 9.47PCh. 9 - Prob. 9.48PCh. 9 - Prob. 9.49PCh. 9 - Prob. 9.50PCh. 9 - Label the conjugate acid-base pairs in each...Ch. 9 - Label the conjugate acid-base pairs in each...Ch. 9 - Prob. 9.53PCh. 9 - Prob. 9.54PCh. 9 - Fill in the missing product in each acid-base...Ch. 9 - Fill in the missing product in each acid-base...Ch. 9 - Prob. 9.57PCh. 9 - Write the equation for the acid-base reaction that...Ch. 9 - Prob. 9.59PCh. 9 - Which diagram represents what happens when HCN...Ch. 9 - Prob. 9.61PCh. 9 - Prob. 9.62PCh. 9 - Prob. 9.63PCh. 9 - Use the data in and 9.2 and 9.3 to label the...Ch. 9 - Prob. 9.65PCh. 9 - Which acid, A or B, is stronger in each part? a. B...Ch. 9 - Fill in the missing terms (strong or weak) and...Ch. 9 - Fill in the missing terms (strong or weak) and...Ch. 9 - For each pair of acids: [1] Label the stronger...Ch. 9 - For each pair of acids: [1] Label the stronger...Ch. 9 - Prob. 9.71PCh. 9 - Prob. 9.72PCh. 9 - Prob. 9.73PCh. 9 - Label the acid in the reactants and the conjugate...Ch. 9 - Calculate the value of [OH-] from the given and...Ch. 9 - Calculate the value of [OH-] from the given [H3O+]...Ch. 9 - Calculate the value of [OH-] from the given [HO-]...Ch. 9 - Calculate the value of [H3O+] from the given [OH-]...Ch. 9 - Prob. 9.79PCh. 9 - Prob. 9.80PCh. 9 - Prob. 9.81PCh. 9 - Complete the following table with the needed...Ch. 9 - Prob. 9.83PCh. 9 - Prob. 9.84PCh. 9 - Prob. 9.85PCh. 9 - If pancreaticfluids have a pH of 8.2, calculate...Ch. 9 - Calculate the concentrations of H3O+ and OH in the...Ch. 9 - Prob. 9.88PCh. 9 - Prob. 9.89PCh. 9 - Prob. 9.90PCh. 9 - Prob. 9.91PCh. 9 - Prob. 9.92PCh. 9 - Write a balanced equation for each reaction. a....Ch. 9 - Prob. 9.94PCh. 9 - Prob. 9.95PCh. 9 - Prob. 9.96PCh. 9 - Prob. 9.97PCh. 9 - Prob. 9.98PCh. 9 - Prob. 9.99PCh. 9 - Prob. 9.100PCh. 9 - Whatisthe molarityofanaceticacid (CH3COOH)...Ch. 9 - What is the molarity of an H2SO4 solution if 18.5...Ch. 9 - How many milliliters of 1.0MNaOH solution are...Ch. 9 - How many milliliters of 2.0MNaOH solution are...Ch. 9 - Prob. 9.105PCh. 9 - Prob. 9.106PCh. 9 - Prob. 9.107PCh. 9 - Prob. 9.108PCh. 9 - Prob. 9.109PCh. 9 - Prob. 9.110PCh. 9 - Using the Ka values in Table9.6, calculate the pH...Ch. 9 - Using the Ka values in Table9.6, calculate the pH...Ch. 9 - Calculate the pH of an acetic acid/acetate buffer...Ch. 9 - Calculate the pH of a bicarbonate/carbonate buffer...Ch. 9 - Why is the pH of unpolluted rainwater lower than...Ch. 9 - Prob. 9.116PCh. 9 - Prob. 9.117PCh. 9 - A sample of rainwater has a pH of 4.18. Calculate...Ch. 9 - Prob. 9.119PCh. 9 - Prob. 9.120PCh. 9 - Prob. 9.121PCh. 9 - Prob. 9.122PCh. 9 - Prob. 9.123CPCh. 9 - Prob. 9.124CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Nonearrow_forward%Reflectance 95 90- 85 22 00 89 60 55 50 70 65 75 80 50- 45 40 WA 35 30- 25 20- 4000 3500 Date: Thu Feb 06 17:21:21 2025 (GMT-05:0(UnknownD Scans: 8 Resolution: 2.000 3000 2500 Wavenumbers (cm-1) 100- 2981.77 1734.25 2000 1500 1000 1372.09 1108.01 2359.09 1469.82 1181.94 1145.20 1017.01 958.45 886.97 820.49 668.25 630.05 611.37arrow_forwardNonearrow_forward
- CH3 CH H3C CH3 H OH H3C- -OCH2CH3 H3C H -OCH3 For each of the above compounds, do the following: 1. List the wave numbers of all the IR bands in the 1350-4000 cm-1 region. For each one, state what bond or group it represents. 2. Label equivalent sets of protons with lower-case letters. Then, for each 1H NMR signal, give the 8 value, the type of splitting (singlet, doublet etc.), and the number protons it represents. of letter δ value splitting # of protons 3. Redraw the compound and label equivalent sets of carbons with lower-case letters. Then for each set of carbons give the 5 value and # of carbons it represents. letter δ value # of carbonsarrow_forwardDraw the correct ionic form(s) of arginine at the pKa and PI in your titration curve. Use your titration curve to help you determine which form(s) to draw out.arrow_forwardPlease correct answer and don't use hand ratingarrow_forward
- Nonearrow_forwardNonearrow_forwardCarbohydrates- Draw out the Hawthorne structure for a sugar from the list given in class. Make sure to write out all atoms except for carbons within the ring. Make sure that groups off the carbons in the ring are in the correct orientation above or below the plane. Make sure that bonds are in the correct orientation. Include the full name of the sugar. You can draw out your curve within the text box or upload a drawing below.arrow_forward
- How many milliliters of 97.5(±0.5) wt% H2SO4 with a density of 1.84(±0.01) g/mL will you need to prepare 2.000 L of 0.110 M H2SO4? If the uncertainty in delivering H2SO4 is ±0.01 mL, calculate the absolute uncertainty in the molarity (0.110 M). Assume there is negligible uncertainty in the formula mass of H2SO4 and in the final volume (2.000 L) and assume random error.arrow_forwardDon't used hand raiting and don't used Ai solutionarrow_forwardWhat should be use to complete the reaction? CN CNarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Acid-Base Titration | Acids, Bases & Alkalis | Chemistry | FuseSchool; Author: FuseSchool - Global Education;https://www.youtube.com/watch?v=yFqx6_Y6c2M;License: Standard YouTube License, CC-BY