
Pearson eText for Essential Organic Chemistry -- Instant Access (Pearson+)
3rd Edition
ISBN: 9780137533268
Author: Paula Bruice
Publisher: PEARSON+
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 55P
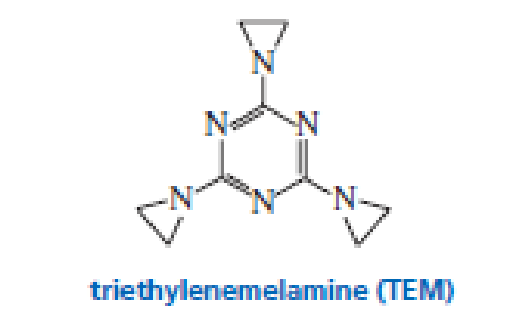
Triethylenemelamine (TEM) is an antitumor agent. Its activity is due to its ability to cross-link DNA
- a. Explain why it can be used only under slightly acidic conditions.
- b. Explain why it can cross-link DNA.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1. The Ability of a substance to exist in different crystalline form
a. Lattice
b. Polymorphism
c. Crystallization
d. Amphoterism
2. A drug can exert its pharmacological effect only if it is
a. Protein bound
b. Protein unbound
c. Free drug
d. Both B & C
e. Both A & C
3. In order for the drug to be ready and available for absorption, it must be release first from its dosage form with the exception of:
a. Capsule
b. Tablet
c. Solution
d. Suspension
4. All of the following are true, except
a. Solubility increase with decrease particle size
b. Solubility increase with increase surface area
c. Solubility increase with increase particle size
d. Solubility decrease with decrease surface area
5. The rate in which the drug appears in the bloodstream is also known as
a. Half-life
b. Potency
c. Bioavailability
d. Area under the curve
1. An inactive or much less active form of drug which transformed to active drug in the body is
a. Coated drug
b. Specialized dosage form
c. Prodrug
d. Reservoir
2. Sodium pump is an example of
a. G-protein receptors
b. Ion channels
c. Gene Transcription linked receptor
d. Enzyme-linked tyrosine kinase3. As the length of a non-polar chain of aliphatic alcohol increases, its solubility in water
a. Increases
b. Decreases
c. No effect
4. More common force of attraction for drugs
a. Covalent
b. Electrostatic
c. Hydrogen bonding
d. Ionic5. In organs and tissues that are well-perfuse, drug distribution is
a. Faster
b. Slower
c. Negligible
d. The same
22. Your 9 year-old son wants to show his friends how cool he is. So he designs an
experiment that makes his friends' tee shirts "magically' turn black, metallic green. The
caused this color change.
presence of
and
a. sucrose, neutral red
b. glucose. Phenol red
c. lactose, eosin methylene blue
d. lactose, neutral red
e. glucose, eosin methylene blue
23. Your husband is fully amazed when his dinner plate "magically" turned black when
you put potatoes on his plate. The presence of
on the plate and
in the potatoes caused this color change.
a. iron, amylase
b. trypthophan, indole
c. iron, hydrogen sulfide
d. lipase, fats
e. decarboxylase, indole
24. The role of safranin in the gram stain is similar to the role of
fast stain.
a. methylene blue
b. carbolfuschin
c. acetone alcohol
d. heat
e. crystal violet
25. Immersion oil was used in conjunction with the
the spread of light to increase the microscope's resolution.
a. 100x, condensed
b. 40x, increased
c. 100x total magnification, increased…
Chapter 9 Solutions
Pearson eText for Essential Organic Chemistry -- Instant Access (Pearson+)
Ch. 9.1 - Draw the structures of straight-chain alcohols...Ch. 9.1 - Prob. 2PCh. 9.1 - Prob. 3PCh. 9.2 - Why are NH3 and CH3NH2 no longer nucleophiles when...Ch. 9.2 - Prob. 5PCh. 9.2 - The observed relative reactivities of primary,...Ch. 9.4 - Which of the following alcohols would dehydrate...Ch. 9.4 - Prob. 10PCh. 9.4 - Prob. 11PCh. 9.4 - Prob. 12P
Ch. 9.4 - Prob. 13PCh. 9.5 - What product will be obtained from the reaction of...Ch. 9.5 - Prob. 15PCh. 9.6 - a. What is each ethers systematic name? 1....Ch. 9.8 - Draw the structure of the following: a....Ch. 9.8 - Prob. 20PCh. 9.8 - Would you expect the reactivity of a five-membered...Ch. 9.9 - Explain why the two arene oxides in Problem 22...Ch. 9.9 - Which compound is more likely to be...Ch. 9.11 - The following three nitrogen mustards were studied...Ch. 9 - What are the common and systematic names of the...Ch. 9 - Prob. 28PCh. 9 - Prob. 29PCh. 9 - Prob. 30PCh. 9 - Prob. 31PCh. 9 - What is the major product obtained from the...Ch. 9 - Draw structures for the following: a....Ch. 9 - Prob. 34PCh. 9 - Prob. 35PCh. 9 - Prob. 36PCh. 9 - Prob. 37PCh. 9 - Ethylene oxide reacts readily with HO.because of...Ch. 9 - Propose a mechanism for each of the following...Ch. 9 - Which of the following ethers would be obtained in...Ch. 9 - Show how each of the following syntheses could be...Ch. 9 - Prob. 42PCh. 9 - Prob. 43PCh. 9 - Prob. 44PCh. 9 - Propose a mechanism for each of the following...Ch. 9 - a. Propose a mechanism for the following reaction:...Ch. 9 - Three arene oxides can be obtained from...Ch. 9 - Prob. 48PCh. 9 - The following reaction takes place several times...Ch. 9 - Show how each of the following compounds could be...Ch. 9 - Propose a mechanism for the following reaction:Ch. 9 - Propose a mechanism for the following reaction:Ch. 9 - What alkenes would you expect to be obtained from...Ch. 9 - Triethylenemelamine (TEM) is an antitumor agent....Ch. 9 - When a diol that has OH groups on adjacent carbons...Ch. 9 - What product is obtained when...Ch. 9 - Prob. 58P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- which has a positive result in ninhydrin test? a. ethyl ethanoate b. sucrose c. proline d. 1-butanol which is more soluble in water? a. 1-aminohexane b. tripopylaminearrow_forwardthe analysis of a mixture of hydrocarbon cracking products, all of which are able to be distilled, but distillation is unable to separate them cleanly. A. a pharmaceutical product containing 2 stereoisomers of the active ingredient, both of which are fairly polar, aromatic, water soluble, and decompose rather than boil. B. C. The separation of a mixture of water-soluble globular proteins of various sizes. The analysis of a mixture of reactor gases that includes methane, hydrogen, carbon dioxide, carbon monoxide, and nitrogen. D.arrow_forwardA. 2,4-dihydroxybenzaldehyde reacts with ethylamine to form an imine. B. 2,4-dihydroxybenzaldehyde reacts with ethanamide to form an ester. Are the statements True?arrow_forward
- Proteins with a quaternary structure have A. A protein and a nucleic acid bonded together. B. A protein and a lipid bonded together. C.Two or more proteins subunits bonded together. D. A protein and a carbohydrate bonded together. E. Answers A, B, and C are correct.arrow_forwardd) 1.EtMgBr PCC HO- CH2CI2 2. H20 Aarrow_forwardRESET 4. A peptide bond is classified as a(n) C. phosphate bond. A. ester B. amine D. amide E. none of these Cancel Done 中。arrow_forward
- Nonearrow_forwardPRODUCTS. Match the name of the product with the biochemical reaction. Products may be used more than once. Tests may require more than one answer.a. 2,3-butanediol 1.catalase b. ammonia 2.phenylalanine deamination c.fatty acids 3.triglyceride hydrolysis d. indole 4.tryptophan degradation e.molecular oxygen 5.urea hydrolysis f. phenylpyruvic acid 6.Voges-Proskauer testarrow_forwardDisadvantages of using cisplatin as an anti-cancer drugarrow_forward
- II. Odd-One-Out. Find which of the following is not included in the group. b. Tyrosine b. Histidine b. Amide b. Glysine b. Cys, C b. Tyr, T c. Phenylalanine c. Tryptophan c. Basic c. Methionine c. Val, V c. Lys, K 36. a. Tryptophan 37. a. Arginine 38. a. Alcohol d. Histidine d. Lysine d. Sulfur d. Leucine d. Pro, P d. Leu, L 39. a. Alanine 40. a. Glu, G 41. a. Asp, Darrow_forward13. Artificial sweetening agent that has a bitter aftertaste, especially at high concentrations. A. Aspartame B. Saccharin C. Glucose D. Sucralose C. 14. TRUE or FALSE: When digested, aspartame breaks down into three components: aspartic acid, phenylalanine, and methanol. A. True B. False 15. An artificial sweetening agent commonly used in dairy dessert. A. Aspartame В. Saccharin C. Glucose D. Sucralosearrow_forwardcan you answer activity 7?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT  Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License