
Concept explainers
Interpretation: An explanation regarding the cleavage of the given amide substrate A by chymotrypsin reveals no burst by using stopped-flow kinetic methods is to be stated.
Concept introduction: The organic compounds that contain
Proteolytic enzymes commonly known as proteases are used for the cleavage of protein. Chymotrypsin is an enzyme that is used for the cleavage of protein through hydrolysis reaction.
Answer to Problem 1P
The cleavage of the given amide substrate, A, by chymotrypsin reveals no burst because the acyl-enzyme intermediate formation occurs at a slower rate as compared to the hydrolysis for the given amide substrate.
Explanation of Solution
Given:
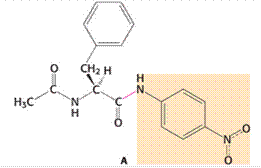
Chymotrypsin is an enzyme that is used for the cleavage of protein through hydrolysis reaction. In case of large hydrophobic amino acids, the cleavage of the peptide bond occurs particularly at the carboxyl-terminal side. In the given amide substrate A, the cleavage by chymotrypsin shows no burst because the hydrolysis for this substrate of amide is faster than the generation of the acyl-enzyme intermediate. In other words, the acyl-enzyme intermediate formation occurs at a slower rate as compared to the hydrolysis for the given amide substrate. Hence, no burst is found. In case of ester substrates, the acyl-enzyme intermediate formation occurs at a faster rate.
Want to see more full solutions like this?
Chapter 9 Solutions
BIOCHEM-ACHIEVE(FIRST DAY DISCOUNTED)
- You’ve isolated a protein and determined that the Native molecular weight of the holoenzyme is 160 kD using size exclusion chromatography. Analysis of this protein using SDS-PAGE revealed 2 bands, one at 100 kD and one at 30 kD. The enzyme was found to be 0.829% NAD (by weight). What further can be said regarding the structure of the polypeptide?arrow_forwardWhat is the formation of glycosylated hemoglobin (the basis for the HbA1c test)? Can you describe it?arrow_forwardPlease analze the gel electrophoresis column of the VRK1 kinase (MW: 39.71 kDa). Also use a ruler to measure the length of the column in centimeters and calculate the MW of each band observed. Lane 1: buffer Lane 2 : Ladder Lane 3: Lysate Lane 4: Flowthrough Lane 5: Wash Lanes 6-8: E1, E2, E3 Lane 9: Dialyzed VRK1 Lane 10: LDHarrow_forward
- Do sensory neurons express ACE2 or only neurolipin-1 receptors for COVID19 virus particle binding?arrow_forwardExplain the process of CNS infiltration of COVID19 through sensory neurons from beginning to end, including processes like endocytosis, the different receptors/proteins that are involved, how they are transported and released, etc.,arrow_forwardH2C CH2 HC-COOO CH2 ܘHO-C-13c-O isocitrate C-S-COA H213c CH2 C-OO 13C-S-COA CH2 C-00 the label will not be present in succinyl CoA C-S-COA succinyl-CoAarrow_forward
- A culture of kidneys cells contains all intermediates of the citric acid cycle. It is treated with an irreversible inhibitor of malate dehydrogenase, and then infused withglucose. Fill in the following list to account for the number of energy molecules that are formed from that one molecule of glucose in this situation. (NTP = nucleotidetriphosphate, e.g., ATP or GTP)Net number of NTP:Net number of NADH:Net number of FADH2:arrow_forward16. Which one of the compounds below is the final product of the reaction sequence shown here? OH A B NaOH Zn/Hg aldol condensation heat aq. HCI acetone C 0 D Earrow_forward2. Which one of the following alkenes undergoes the least exothermic hydrogenation upon treatment with H₂/Pd? A B C D Earrow_forward
- 6. What is the IUPAC name of the following compound? A) (Z)-3,5,6-trimethyl-3,5-heptadiene B) (E)-2,3,5-trimethyl-1,4-heptadiene C) (E)-5-ethyl-2,3-dimethyl-1,5-hexadiene D) (Z)-5-ethyl-2,3-dimethyl-1,5-hexadiene E) (Z)-2,3,5-trimethyl-1,4-heptadienearrow_forwardConsider the reaction shown. CH2OH Ex. CH2 -OH CH2- Dihydroxyacetone phosphate glyceraldehyde 3-phosphate The standard free-energy change (AG) for this reaction is 7.53 kJ mol-¹. Calculate the free-energy change (AG) for this reaction at 298 K when [dihydroxyacetone phosphate] = 0.100 M and [glyceraldehyde 3-phosphate] = 0.00300 M. AG= kJ mol-1arrow_forwardIf the pH of gastric juice is 1.6, what is the amount of energy (AG) required for the transport of hydrogen ions from a cell (internal pH of 7.4) into the stomach lumen? Assume that the membrane potential across this membrane is -70.0 mV and the temperature is 37 °C. AG= kJ mol-1arrow_forward
 BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College
Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning
Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax





