
Chemistry In Context
9th Edition
ISBN: 9781259638145
Author: Fahlman, Bradley D., Purvis-roberts, Kathleen, Kirk, John S., Bentley, Anne K., Daubenmire, Patrick L., ELLIS, Jamie P., Mury, Michael T., American Chemical Society
Publisher: Mcgraw-hill Education,
expand_more
expand_more
format_list_bulleted
Question
Chapter 9, Problem 16Q
Interpretation Introduction
Interpretation:
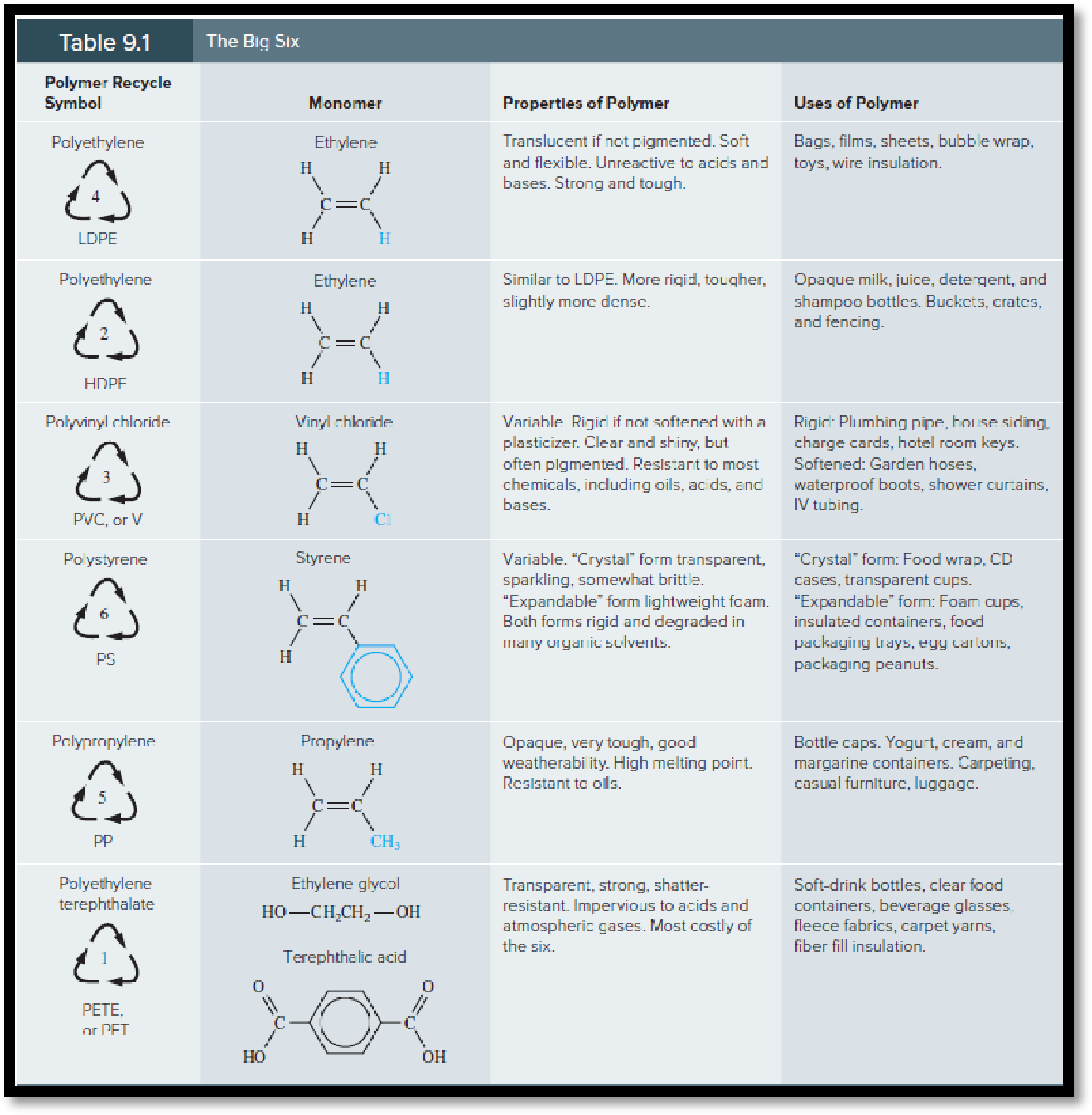
Taking 10 plastic containers of one’s choice it should be identified that which polymer is most frequently encountered in the sample.
Concept Introduction:
Monomer: A molecule is considered as monomer when this molecule bonds with another identical molecule which results to form polymer.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Select the product for the following reaction.
HO
HO
PCC
OH
○
OH
O HO
○ HO
HO
HO
5:45
Х
Select the final product for the following reaction sequence.
O
O
1. Mg. ether
2.D.O
Based on the chart
Two similarities between the molecule with alpha glycosidic linkages.
Two similarities between the molecules with beta glycosidtic linkages.
Two differences between the alpha and beta glycosidic linkages.
Chapter 9 Solutions
Chemistry In Context
Ch. 9.1 - Scientific Practices Tennis Anyone? Examine this...Ch. 9.3 - Prob. 9.2YTCh. 9.3 - Prob. 9.3YTCh. 9.4 - Prob. 9.4YTCh. 9.4 - Prob. 9.5YTCh. 9.4 - Prob. 9.6YTCh. 9.4 - Prob. 9.7YTCh. 9.4 - Prob. 9.8YTCh. 9.4 - Prob. 9.9YTCh. 9.5 - Prob. 9.10YT
Ch. 9.5 - Skill Building Benzene and Phenyl The difference...Ch. 9.5 - Prob. 9.13YTCh. 9.5 - Skill Building Polystyrene Possibilities Show the...Ch. 9.6 - Skill Building Esters and Polyesters You have seen...Ch. 9.6 - Prob. 9.16YTCh. 9.7 - Skill Building Kevlar Kevlar is a polyamide used...Ch. 9.8 - Prob. 9.20YTCh. 9.8 - Your Turn 9.22 Skill Building Burning a Plastic...Ch. 9.8 - Your Turn 9.23 Scientific Practices Landfill...Ch. 9.9 - Examine the values in Table 9.4 from the American...Ch. 9.9 - Prob. 9.25YTCh. 9.9 - Prob. 9.26YTCh. 9.9 - Prob. 9.28YTCh. 9.10 - Skill Building The Chemistry of PLA We dont show...Ch. 9.11 - Your Turn 9.31 Scientific Practices Glass or...Ch. 9.11 - Prob. 9.32YTCh. 9.11 - Skill Building Meet DEHP DEHP belongs to a common...Ch. 9 - Prob. 1QCh. 9 - Prob. 2QCh. 9 - Prob. 3QCh. 9 - Prob. 4QCh. 9 - Prob. 5QCh. 9 - Prob. 6QCh. 9 - Prob. 7QCh. 9 - Prob. 8QCh. 9 - Prob. 9QCh. 9 - Prob. 10QCh. 9 - Prob. 11QCh. 9 - Prob. 12QCh. 9 - Prob. 13QCh. 9 - Prob. 14QCh. 9 - Prob. 15QCh. 9 - Prob. 16QCh. 9 - Prob. 17QCh. 9 - Prob. 18QCh. 9 - Prob. 19QCh. 9 - Prob. 20QCh. 9 - Prob. 21QCh. 9 - Prob. 22QCh. 9 - Prob. 23QCh. 9 - Prob. 24QCh. 9 - Prob. 25QCh. 9 - Prob. 26QCh. 9 - Prob. 27QCh. 9 - Prob. 28QCh. 9 - Prob. 29QCh. 9 - Prob. 30QCh. 9 - Prob. 31QCh. 9 - Prob. 32QCh. 9 - Prob. 33QCh. 9 - Prob. 34QCh. 9 - Prob. 35QCh. 9 - Prob. 36QCh. 9 - Prob. 37QCh. 9 - Prob. 38QCh. 9 - Prob. 39QCh. 9 - Prob. 40QCh. 9 - Prob. 41QCh. 9 - Prob. 42QCh. 9 - Prob. 43QCh. 9 - Prob. 44QCh. 9 - Prob. 45QCh. 9 - Prob. 46QCh. 9 - Prob. 47QCh. 9 - Prob. 48QCh. 9 - Prob. 49QCh. 9 - Prob. 50QCh. 9 - Prob. 51QCh. 9 - Prob. 52QCh. 9 - Prob. 53QCh. 9 - Prob. 54QCh. 9 - Prob. 55QCh. 9 - Prob. 56QCh. 9 - Prob. 57QCh. 9 - Prob. 58QCh. 9 - Prob. 59Q
Knowledge Booster
Similar questions
- please help fill in the tablearrow_forwardAnswer F pleasearrow_forward4. Refer to the data below to answer the following questions: The octapeptide saralasin is a specific antagonist of angiotensin II. A derivative of saralasin is used therapeutically as an antihypertensive. Amino acid analysis of saralasin show the presence of the following amino acids: Ala, Arg, His, Pro, Sar, Tyr, Val, Val A.Sar is the abbreviation for sarcosine, N-methyl aminoethanoic acid. Draw the structure of sarcosine. B. N-Terminal analysis by the Edman method shows saralasin contains sarcosine at the N-terminus. Partial hydrolysis of saralasin with dilute hydrochloric acid yields the following fragments: Tyr-Val-His Sar-Arg-Val His-Pro-Ala Val-Tyr-Val Arg-Val-Tyr What is the structure of saralasin?arrow_forward
- What is the structure of the DNA backbone?arrow_forwardPLEASE PLEASE PLEASE use hand drawn structures when possarrow_forward. M 1- MATCH each of the following terms to a structure from the list below. There is only one correct structure for each term and structures may be used more than once. Place the letter of the structure in the blank to the left of the corresponding term. A. Sanger dideoxy method C. Watson-Crick B. GAUCGUAAA D. translation E. HOH2C OH OH G. transcription I. AUGGCUGAG 0 K. OPOH2C 0- OH N- H NH2 F. -OPOH2C 0- OH OH H. Maxam-Gilbert method J. replication N L. HOH2C a. b. C. d. e. f. g. B M. AGATCGCTC a pyrimidine nucleoside RNA base sequence with guanine at the 3' end. DNA base sequence with cytosine at the 3' end. a purine nucleoside DNA sequencing method for the human genome 2'-deoxyadenosine 5'-phosphate process by which mRNA directs protein synthesis OH NH2arrow_forward
- Please use hand drawn structures when neededarrow_forwardB. Classify the following amino acid. Atoms other than carbon and hydrogen are labeled. a. acidic b. basic C. neutral C. Consider the following image. Which level of protein structure is shown here? a. primary b. secondary c. tertiary d. quaternary D. Consider the following image. H RH H HR H R HR HR RH Which level of protein structure is shown in the box? a. primary b. secondary R c. tertiary d. quaternary コー Rarrow_forwardBriefly answer three from the followings: a. What are the four structures of the protein? b. Why is the side chain (R) attached to the alpha carbon in the amino acids is important for the function? c. What are the types of amino acids? And how is it depend on the (R) structure? d. Write a reaction to prepare an amino acid. prodarrow_forward
- Answe Answer A and B pleasearrow_forward3. Refer to the data below to answer the following questions: Isoelectric point Amino Acid Arginine 10.76 Glutamic Acid 3.22 Tryptophan 5.89 A. Define isoelectric point. B. The most basic amino acid is C. The most acidic amino acid is sidizo zoarrow_forward3. A gas mixture contains 50 mol% H2 and 50 mol% He. 1.00-L samples of this gas mixture are mixed with variable volumes of O2 (at 0 °C and 1 atm). A spark is introduced to allow the mixture to undergo complete combustion. The final volume is measured at 0 °C and 1 atm. Which graph best depicts the final volume as a function of the volume of added O2? (A) 2.00 1.75 Final Volume, L 1.50 1.25 1.00 0.75 0.50 0.25 0.00 0.00 0.25 0.50 2.00 (B) 1.75 1.50 Final Volume, L 1.25 1.00 0.75 0.50- 0.25 0.00 0.75 1.00 0.00 0.25 Volume O₂ added, L 2 0.50 0.75 1.00 Volume O₂ added, L 2 2.00 2.00 (C) (D) 1.75 1.75 1.50 1.50 Final Volume, L 1.25 1.00 0.75 0.50 Final Volume, L 1.25 1.00 0.75 0.50 0.25 0.25 0.00 0.00 0.00 0.25 0.50 0.75 1.00 0.00 0.25 Volume O₂ added, L 0.50 0.75 1.00 Volume O₂ added, L 2arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781285199030
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER