
Chemistry: The Central Science (14th Edition)
14th Edition
ISBN: 9780134414232
Author: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 9, Problem 11E
(a)
Interpretation Introduction
To determine: The atomic orbital used to construct the given MO.
(b)
Interpretation Introduction
To determine: The type of MOs 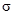 or
or 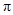
(c)
Interpretation Introduction
To determine: If the MO is bonding or anti bonding.
(d)
Interpretation Introduction
To determine: The location of nodal planes in the given molecular orbital diagram.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Protecting Groups and Carbonyls
6) The synthesis generates allethrolone that exhibits high insect toxicity but low mammalian toxicity. They are used in pet
shampoo, human lice shampoo, and industrial sprays for insects and mosquitos. Propose detailed mechanistic steps to
generate the allethrolone label the different types of reagents (Grignard, acid/base protonation, acid/base deprotonation,
reduction, oxidation, witting, aldol condensation, Robinson annulation, etc.)
III + VI
HS
HS
H+
CH,CH,Li
III
I
II
IV
CI + P(Ph)3
V
༼
Hint: no strong base added
VI
S
VII
IX
HO
VIII
-MgBr
HgCl2,HgO
HO.
isomerization
aqeuous solution
H,SO,
༽༽༤༽༽
X
MeOH
Hint: enhances selectivity for reaction at the S
X
☑
Draw the complete mechanism for the acid-catalyzed hydration of this alkene.
esc
田
Explanation
Check
1
888
Q
A
slock
Add/Remove step
Q
F4
F5
F6
A
བྲA
F7
$
%
5
@
4
2
3
&
6
87
Click and drag to start
drawing a structure.
© 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Ce
W
E
R
T
Y
U
S
D
LL
G
H
IK
DD
요
F8
F9
F10
F1
*
(
8
9
0
O
P
J
K
L
Z
X
C
V
B
N
M
H
He
command
Explanation
Check
F1
H₂O
H₂
Pd
1) MCPBA
2) H3O+
1) Hg(OAc)2, H₂O
2) NaBH4
OH
CI
OH OH
OH
hydration
halohydrin formation
addition
halogenation
hydrogenation
inhalation
hydrogenation
hydration
☐ halohydrin formation
addition
halogenation
formation chelation
hydrogenation
halohydrin formation
substitution
hydration
halogenation
addition
Ohalohydrin formation
subtraction
halogenation
addition
hydrogenation
hydration
F2
80
F3
σ
F4
F5
F6
1
!
2
#
3
$
4
%
05
Q
W
&
Å
© 2025 McGraw Hill LLC. All Rights Reserved.
F7
F8
(
6
7
8
9
LU
E
R
T
Y
U
A
F9
Chapter 9 Solutions
Chemistry: The Central Science (14th Edition)
Ch. 9.2 - Consider the following AB3 molecules and ions-...Ch. 9.2 - Prob. 9.1.2PECh. 9.2 - Prob. 9.2.1PECh. 9.2 - Prob. 9.2.2PECh. 9.2 - Prob. 9.3.1PECh. 9.2 - Prob. 9.3.2PECh. 9.3 - Prob. 9.4.1PECh. 9.3 - Determine whether the following molecules are...Ch. 9.5 - Prob. 9.5.1PECh. 9.5 - Prob. 9.5.2PE
Ch. 9.6 - Prob. 9.6.1PECh. 9.6 - Prob. 9.6.2PECh. 9.6 - Prob. 9.7.1PECh. 9.6 - Prob. 9.7.2PECh. 9.7 - Prob. 9.8.1PECh. 9.7 - Prob. 9.8.2PECh. 9.8 - Prob. 9.9.1PECh. 9.8 - Prob. 9.9.2PECh. 9 - Prob. 1DECh. 9 - 9.1 A certain AB4, molecule has a "seesaw" shape...Ch. 9 - Prob. 2ECh. 9 - Prob. 3ECh. 9 - Prob. 4ECh. 9 - Prob. 5ECh. 9 - Prob. 6ECh. 9 - In the hydrocarbon a. What is the hybridization at...Ch. 9 - The drawing below shows the overlap of two hybrid...Ch. 9 - Prob. 9ECh. 9 -
9.10 The following is part of a molecular...Ch. 9 - Prob. 11ECh. 9 - Prob. 12ECh. 9 -
9.13
a. An AB2 molecule is linear. How...Ch. 9 - a. Methane (CH4) and the perchlorate ion (C104-)...Ch. 9 - Prob. 15ECh. 9 - Prob. 16ECh. 9 - Prob. 17ECh. 9 - Prob. 18ECh. 9 - In which of these molecules or ions does the...Ch. 9 - Prob. 20ECh. 9 - How many nonbonding electron pairs are there in...Ch. 9 - Prob. 22ECh. 9 - Prob. 23ECh. 9 - Prob. 24ECh. 9 - Give the electron-domain and molecular geometries...Ch. 9 - Prob. 26ECh. 9 - Prob. 27ECh. 9 - Prob. 28ECh. 9 - Prob. 29ECh. 9 - Prob. 30ECh. 9 - Ammonia, NH3 reacts with incredibly strong bases...Ch. 9 - In which of the following AFn molecules or ions is...Ch. 9 - a. Explain why BrF4 is square planar, whereas...Ch. 9 -
9.34 Name the proper three-dimensional molecule...Ch. 9 - Prob. 35ECh. 9 - Prob. 36ECh. 9 - Prob. 37ECh. 9 - Prob. 38ECh. 9 - a. (a) Is the molecule BF3 polar or nonpolar? b....Ch. 9 - Prob. 40ECh. 9 - Predict whether each of the following molecules is...Ch. 9 - Prob. 42ECh. 9 - Prob. 43ECh. 9 - Prob. 44ECh. 9 - For each statement, irldicate whether it is true...Ch. 9 - Draw sketches illustrating the overlap between the...Ch. 9 - For each statement, indicate whether it is true or...Ch. 9 - Prob. 48ECh. 9 - Prob. 49ECh. 9 - Consider the SC12 molecule. a. What IS the...Ch. 9 - Indicate the hybridization of the central atom in...Ch. 9 - Prob. 52ECh. 9 - Prob. 53ECh. 9 - Prob. 54ECh. 9 - Prob. 55ECh. 9 - Prob. 56ECh. 9 - a. Draw Lewis structures for ethane (C2He),...Ch. 9 - a. Draw Lewis structures for ethane (C2He),...Ch. 9 - Prob. 59ECh. 9 - Ethyl acetate. C4H802, is a fragrant substance...Ch. 9 - Prob. 61ECh. 9 - Prob. 62ECh. 9 - Prob. 63ECh. 9 - Prob. 64ECh. 9 - In the formate ion, HC02- , the carbon atom is the...Ch. 9 - Prob. 66ECh. 9 - Prob. 67ECh. 9 - Prob. 68ECh. 9 - Prob. 69ECh. 9 - a. If you combine two atomic orbitals on two...Ch. 9 - Prob. 71ECh. 9 - Prob. 72ECh. 9 - Prob. 73ECh. 9 - Indicate whether each statement is true or false....Ch. 9 - Prob. 75ECh. 9 - Prob. 76ECh. 9 - Prob. 77ECh. 9 - Prob. 78ECh. 9 - Prob. 79ECh. 9 - Prob. 80ECh. 9 - Determine the electron configurations for CN+, CN,...Ch. 9 - Prob. 82ECh. 9 - Consider the molecular orbitals of the P2...Ch. 9 - The iodine bromide molecule, IBr, is an...Ch. 9 - Prob. 85AECh. 9 - Prob. 86AECh. 9 - Consider the following XF4 ions: PF4, BrF4-,...Ch. 9 -
9.88 Consider the molecule PF4Cl....Ch. 9 - Prob. 89AECh. 9 - Fill in the blank spaces in the following chart....Ch. 9 - Prob. 91AECh. 9 - Prob. 92AECh. 9 - Prob. 93AECh. 9 - Prob. 94AECh. 9 - Prob. 95AECh. 9 - Prob. 96AECh. 9 - Prob. 97AECh. 9 - Prob. 98AECh. 9 - Prob. 99AECh. 9 - Prob. 100AECh. 9 - In ozone, 03, the two oxygen atoms on the ends Of...Ch. 9 - Butadiene, C4H6, is a planar molecule that has the...Ch. 9 - The structure of borazine, B3N3H6, is a...Ch. 9 - Prob. 104AECh. 9 - Prob. 105AECh. 9 - Prob. 106AECh. 9 - Prob. 107AECh. 9 - Prob. 108AECh. 9 - Azo dyes are organic dyes that are used for many...Ch. 9 - a. Using only the valence atomic orbitals of a...Ch. 9 - Carbon monoxide, CO, is isoelectronic to N2. a....Ch. 9 - The energy-level diagram in Figure 9.36 shows that...Ch. 9 - A compound composed of 2.1 29.8%N, and 68.1%O has...Ch. 9 -
9.114 Sulfur tetrafluoride (SR4) reacts slowly...Ch. 9 - Prob. 115IECh. 9 - The molecule 2-butene, C4Hs, can undergo a...Ch. 9 - Prob. 117IECh. 9 - Use average bond enthalpies (Table 8.3 ) to...Ch. 9 - Prob. 119IECh. 9 - Prob. 120IECh. 9 - Prob. 121IECh. 9 - Prob. 122IE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Show the mechanism steps to obtain the lowerenergy intermediate: *see imagearrow_forwardSoap is made by the previous reaction *see image. The main difference between one soap and another soap isthe length (number of carbons) of the carboxylic acid. However, if a soap irritates your skin, they mostlikely used too much lye.Detergents have the same chemical structure as soaps except for the functional group. Detergentshave sulfate (R-SO4H) and phosphate (R-PO4H2) functional groups. Draw the above carboxylic acidcarbon chain but as the two variants of detergents. *see imagearrow_forwardWhat are the reactions or reagents used? *see imagearrow_forward
- The two pKa values of oxalic acid are 1.25 and 3.81. Why are they not the same value? Show the protontransfer as part of your explanation. *see imagearrow_forwardасть Identify all the bonds that gauche interact with C-OMe in the most stable conformation of the above compound.arrow_forwardPredict the reactants used in the formation of the following compounds using Acid-Catalyzed dehydration reactionarrow_forward
- Can I please get help with this?arrow_forward.. Give the major organic product(s) for each of the following reactions or sequences of reactions. Show ll relevant stereochemistry [3 ONLY]. A H Br 1. NaCN 2 NaOH, H₂O, heat 3. H3O+ B. CH₂COOH 19000 1. LiAlH4 THF, heat 2 H₂O* C. CH Br 1. NaCN, acetone 2 H3O+, heat D. Br 1. Mg. ether 3. H₂O+ 2 CO₂ E. CN 1. (CH) CHMgBr, ether 2 H₂O+arrow_forwardAssign this COSY spectrumarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY