
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 8.7, Problem 8.48P
Interpretation Introduction
Interpretation:
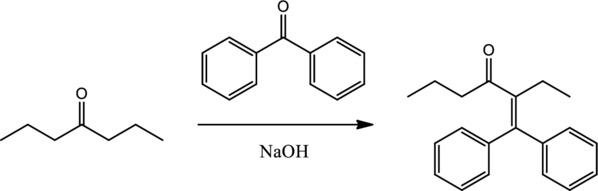
Mechanism has to be drawn for the given transformation.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
am os
Д
Br
Please use the information from the spectrum to answer these questions
Don't used hand raiting
Chapter 8 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.2 - Prob. 8.9PCh. 8.2 - Prob. 8.10PCh. 8.2 - Prob. 8.11PCh. 8.2 - Prob. 8.12P
Ch. 8.2 - Prob. 8.13PCh. 8.3 - Predict the products of each of the following...Ch. 8.3 - Predict the products of each of the following...Ch. 8.3 - Predict the products of each of the following...Ch. 8.3 - Predict the products of each of the following...Ch. 8.4 - Prob. 8.20PCh. 8.4 - Prob. 8.21PCh. 8.4 - Prob. 8.22PCh. 8.4 - Prob. 8.23PCh. 8.5 - Prob. 8.25PCh. 8.5 - Prob. 8.26PCh. 8.5 - On a separate piece of paper, draw a mechanism for...Ch. 8.6 - Prob. 8.29PCh. 8.6 - Predict the major product for each of the...Ch. 8.6 - Predict the major product for each of the...Ch. 8.6 - Predict the major product for each of the...Ch. 8.6 - Identify the reagents you would use to achieve...Ch. 8.6 - Identify the reagents you would use to achieve...Ch. 8.6 - Identify the reagents you would use to achieve...Ch. 8.7 - Predict the major product for each of the...Ch. 8.7 - Predict the major product for each of the...Ch. 8.7 - Predict the major product for each of the...Ch. 8.7 - Prob. 8.42PCh. 8.7 - Prob. 8.43PCh. 8.7 - Prob. 8.44PCh. 8.7 - Prob. 8.45PCh. 8.7 - Prob. 8.47PCh. 8.7 - Prob. 8.48PCh. 8.7 - Prob. 8.49PCh. 8.7 - Prob. 8.50PCh. 8.8 - Prob. 8.52PCh. 8.8 - Prob. 8.53PCh. 8.8 - Prob. 8.54PCh. 8.8 - Prob. 8.55PCh. 8.8 - Prob. 8.57PCh. 8.8 - Prob. 8.58PCh. 8.8 - Prob. 8.59PCh. 8.8 - Prob. 8.60PCh. 8.8 - Propose a mechanism for each of the following...Ch. 8.8 - Propose a mechanism for each of the following...Ch. 8.8 - Prob. 8.64PCh. 8.9 - Prob. 8.66PCh. 8.9 - Prob. 8.67PCh. 8.9 - Prob. 8.68PCh. 8.9 - Prob. 8.69PCh. 8.9 - Prob. 8.70PCh. 8.9 - Prob. 8.71PCh. 8.9 - Prob. 8.72PCh. 8.9 - Identify what reagents you would use to achieve...Ch. 8.9 - Identify what reagents you would use to achieve...Ch. 8.9 - Identify what reagents you would use to achieve...Ch. 8.10 - Prob. 8.78PCh. 8.10 - Prob. 8.79PCh. 8.10 - Prob. 8.80PCh. 8.10 - Propose a synthesis for each of the following...Ch. 8.10 - Propose a synthesis for each of the following...Ch. 8.10 - Propose a synthesis for each of the following...Ch. 8.10 - Propose a synthesis for each of the following...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- X Draw the major products of the elimination reaction below. If elimination would not occur at a significant rate, check the box under the drawing area instead. ది www. Cl + OH Elimination will not occur at a significant rate. Click and drag to start drawing a structure.arrow_forwardNonearrow_forward1A H 2A Li Be Use the References to access important values if needed for this question. 8A 3A 4A 5A 6A 7A He B C N O F Ne Na Mg 3B 4B 5B 6B 7B 8B-1B 2B Al Si P 1B 2B Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe * Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac Rf Ha ****** Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr Analyze the following reaction by looking at the electron configurations given below each box. Put a number and a symbol in each box to show the number and kind of the corresponding atom or ion. Use the smallest integers possible. cation anion + + Shell 1: 2 Shell 2: 8 Shell 3: 1 Shell 1 : 2 Shell 2 : 6 Shell 1 : 2 Shell 2: 8 Shell 1: 2 Shell 2: 8arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning