
(a)
Interpretation:
The major product of the given reaction and the structure of any intermediate are to be predicted.
Concept introduction:
Substitution reactions are the reactions in which there is a replacement of an atom or a
Elimination reactions are the reaction in which
(a)
Answer to Problem 8.46SP
The structure of an intermediate is shown in Figure 1.
The major product of the given reaction is shown in Figure 2.
Explanation of Solution
When
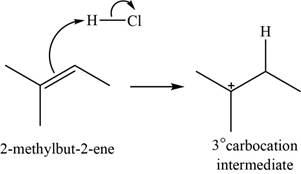
Figure 1
The chloride ion acts as a nucleophile that attacks on the electrophilic tertiary carbocation and results in the formation of
The major product of the given reaction is,
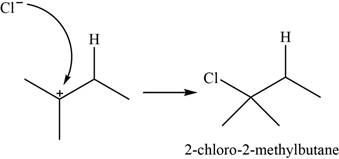
Figure 2
(b)
Interpretation:
The major product of the given reaction and the structure of any intermediate are to be predicted.
Concept introduction:
Substitution reactions are the reactions in which there is a replacement of an atom or a functional group by another atom or a functional group.
Elimination reactions are the reaction in which alkenes are prepared. In this two substituents are eliminated by either one step or two step mechanism.
(b)
Answer to Problem 8.46SP
The structure of an intermediate is shown in Figure 3.
The major product of the given reaction is shown in Figure 4.
Explanation of Solution
When
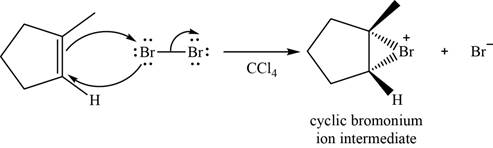
Figure 3
Bromide ion opens the bromonium ion intermediate at the more substituted carbon atom and forms
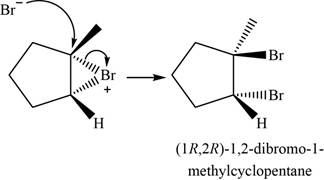
Figure 4
(c)
Interpretation:
The major product of the given reaction and the structure of any intermediate are to be predicted.
Concept introduction:
Substitution reactions are the reactions in which there is a replacement of an atom or a functional group by another atom or a functional group.
Elimination reactions are the reaction in which alkenes are prepared. In this two substituents are eliminated by either one step or two step mechanism.
(c)
Answer to Problem 8.46SP
The structure of an intermediate is shown in Figure 5.
The major product of the given reaction is shown in Figure 6.
Explanation of Solution
When
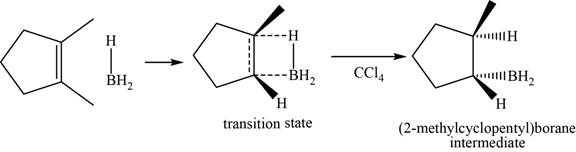
Figure 5
When
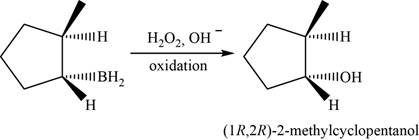
Figure 6
(d)
Interpretation:
The major product of the given reaction and the structure of any intermediate are to be predicted.
Concept introduction:
Substitution reactions are the reactions in which there is a replacement of an atom or a functional group by another atom or a functional group.
Elimination reactions are the reaction in which alkenes are prepared. In this two substituents are eliminated by either one step or two step mechanism.
(d)
Answer to Problem 8.46SP
The structure of an intermediate is shown in Figure 7.
The major product of the given reaction is shown in Figure 8.
Explanation of Solution
When
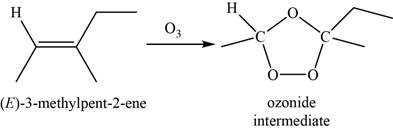
Figure 7
Now, the ozonide intermediate reacts with dimethyl sulfide and forms acetaldehyde and
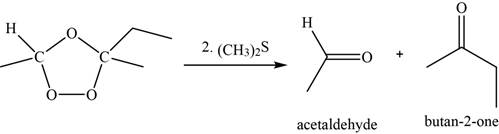
Figure 8
(e)
Interpretation:
The major product of the given reaction and the structure of any intermediate are to be predicted.
Concept introduction:
Substitution reactions are the reactions in which there is a replacement of an atom or a functional group by another atom or a functional group.
Elimination reactions are the reaction in which alkenes are prepared. In this two substituents are eliminated by either one step or two step mechanism.
(e)
Answer to Problem 8.46SP
The major product of the given reaction is shown in Figure 9.
Explanation of Solution
When alkene reacts with
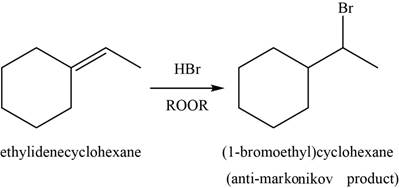
Figure 9
(f)
Interpretation:
The major product of the given reaction and the structure of any intermediate are to be predicted.
Concept introduction:
Substitution reactions are the reactions in which there is a replacement of an atom or a functional group by another atom or a functional group.
Elimination reactions are the reaction in which alkenes are prepared. In this two substituents are eliminated by either one step or two step mechanism.
(f)
Answer to Problem 8.46SP
The major product of the given reaction is shown in Figure 10.
Explanation of Solution
When alkene reacts with
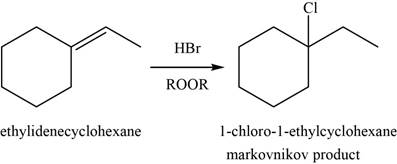
Figure 10
(g)
Interpretation:
The major product of the given reaction and the structure of any intermediate are to be predicted.
Concept introduction:
Substitution reactions are the reactions in which there is a replacement of an atom or a functional group by another atom or a functional group.
Elimination reactions are the reaction in which alkenes are prepared. In this two substituents are eliminated by either one step or two step mechanism.
(g)
Answer to Problem 8.46SP
The major product of the given reaction is shown in Figure 11.
Explanation of Solution
When an alkene reacts with peroxyacid, it forms an
The major product of the given reaction is,
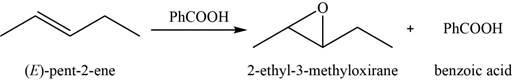
Figure 11
(h)
Interpretation:
The major product of the given reaction and the structure of any intermediate are to be predicted.
Concept introduction:
Substitution reactions are the reactions in which there is a replacement of an atom or a functional group by another atom or a functional group.
Elimination reactions are the reaction in which alkenes are prepared. In this two substituents are eliminated by either one step or two step mechanism.
(h)
Answer to Problem 8.46SP
The structure of an intermediate is shown in Figure 12.
The major product of the given reaction is shown in Figure 13.
Explanation of Solution
Alkene undergoes
The structure of an intermediate is given as,
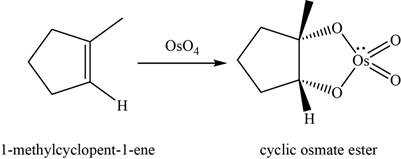
Figure 12
Now, the cyclic osmate undergoes oxidation in the presence of
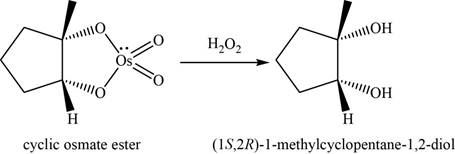
Figure 13
(i)
Interpretation:
The major product of the given reaction and the structure of any intermediate are to be predicted.
Concept introduction:
Substitution reactions are the reactions in which there is a replacement of an atom or a functional group by another atom or a functional group.
Elimination reactions are the reaction in which alkenes are prepared. In this two substituents are eliminated by either one step or two step mechanism.
(i)
Answer to Problem 8.46SP
The major product of the given reaction is shown in Figure 14.
Explanation of Solution
The major product of the given reaction is,
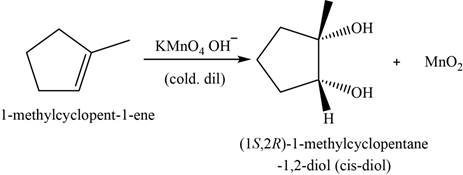
Figure 14
(j)
Interpretation:
The major product of the given reaction and the structure of any intermediate are to be predicted.
Concept introduction:
Substitution reactions are the reactions in which there is a replacement of an atom or a functional group by another atom or a functional group.
Elimination reactions are the reaction in which alkenes are prepared. In this two substituents are eliminated by either one step or two step mechanism.
(j)
Answer to Problem 8.46SP
The structure of an intermediate is shown in Figure 15.
The major product of the given reaction is shown in Figure 16.
Explanation of Solution
When an alkene reacts with peroxyacid, it forms an epoxide or oxirane. Here,
The structure of an intermediate is given as,
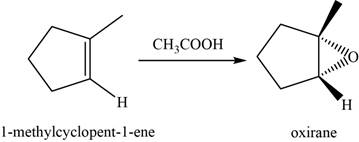
Figure 15
The oxirane is opened at more substituted carbon atom in the presence of an acid
The major product of the given reaction is,
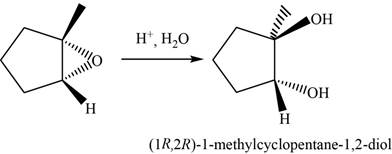
Figure 16
(k)
Interpretation:
The major product of the given reaction and the structure of any intermediate are to be predicted.
Concept introduction:
Substitution reactions are the reactions in which there is a replacement of an atom or a functional group by another atom or a functional group.
Elimination reactions are the reaction in which alkenes are prepared. In this two substituents are eliminated by either one step or two step mechanism.
(k)
Answer to Problem 8.46SP
The major product of the given reaction is shown in Figure 17.
Explanation of Solution
Alkene undergoes oxidative cleavage in the presence of hot concentrated
The major product of the given reaction is,
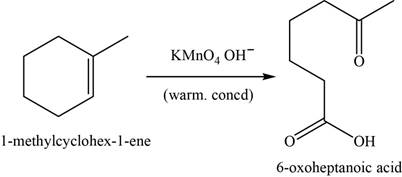
Figure 17
(l)
Interpretation:
The major product of the given reaction and the structure of any intermediate are to be predicted.
Concept introduction:
Substitution reactions are the reactions in which there is a replacement of an atom or a functional group by another atom or a functional group.
Elimination reactions are the reaction in which alkenes are prepared. In this two substituents are eliminated by either one step or two step mechanism.
(l)
Answer to Problem 8.46SP
The major product of the given reaction is shown in Figure 18.
Explanation of Solution
Alkene undergoes ozonolysis to form carbonyl compounds. Here,
The major product of the given reaction is,
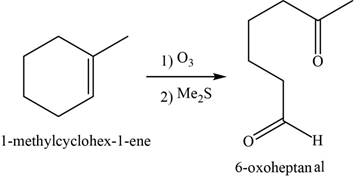
Figure 18
(m)
Interpretation:
The major product of the given reaction and the structure of any intermediate are to be predicted.
Concept introduction:
Substitution reactions are the reactions in which there is a replacement of an atom or a functional group by another atom or a functional group.
Elimination reactions are the reaction in which alkenes are prepared. In this two substituents are eliminated by either one step or two step mechanism.
(m)
Answer to Problem 8.46SP
The major product of the given reaction is shown in Figure 19.
Explanation of Solution
In this reaction,
The major product of the given reaction is,
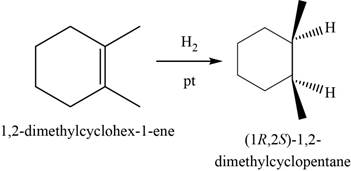
Figure 19
(n)
Interpretation:
The major product of the given reaction and the structure of any intermediate are to be predicted.
Concept introduction:
Substitution reactions are the reactions in which there is a replacement of an atom or a functional group by another atom or a functional group.
Elimination reactions are the reaction in which alkenes are prepared. In this two substituents are eliminated by either one step or two step mechanism.
(n)
Answer to Problem 8.46SP
The structure of an intermediate and the major product of the given reaction are shown in Figure 20.
Explanation of Solution
This reaction is an acid catalyzed hydration of alkene and forms
The structure of an intermediate and the major product of the given reaction is,
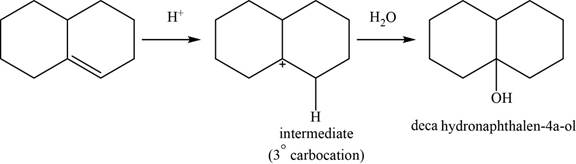
Figure 20
(o)
Interpretation:
The major product of the given reaction and the structure of any intermediate are to be predicted.
Concept introduction:
Substitution reactions are the reactions in which there is a replacement of an atom or a functional group by another atom or a functional group.
Elimination reactions are the reaction in which alkenes are prepared. In this two substituents are eliminated by either one step or two step mechanism.
(o)
Answer to Problem 8.46SP
The major product of the given reaction is shown in Figure 21.
Explanation of Solution
The given reaction is an olefin metathesis. The major product of the given reaction is,
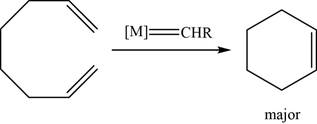
Figure 21
(p)
Interpretation:
The major product of the given reaction and the structure of any intermediate are to be predicted.
Concept introduction:
Substitution reactions are the reactions in which there is a replacement of an atom or a functional group by another atom or a functional group.
Elimination reactions are the reaction in which alkenes are prepared. In this two substituents are eliminated by either one step or two step mechanism.
(p)
Answer to Problem 8.46SP
The structure of an intermediate is shown in Figure 22.
The major product of the given reaction is shown in Figure 24.
Explanation of Solution
Alkene reacts with mercuric acetate and forms mercurinium ion intermediate.
The structure of an intermediate is given as,
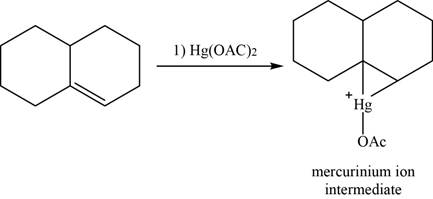
Figure 22
Now, water molecule acts as a nucleophile that opens the mercurinium ion at the most substituted carbon atom.
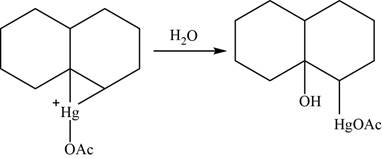
Figure 23
Now, organomercurial alcohol undergoes demercuration in the presence of
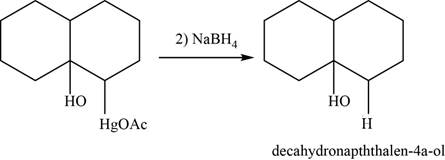
Figure 24
(q)
Interpretation:
The major product of the given reaction and the structure of any intermediate are to be predicted.
Concept introduction:
Substitution reactions are the reactions in which there is a replacement of an atom or a functional group by another atom or a functional group.
Elimination reactions are the reaction in which alkenes are prepared. In this two substituents are eliminated by either one step or two step mechanism.
(q)
Answer to Problem 8.46SP
The structure of an intermediate is shown in Figure 25.
The major product of the given reaction is shown in Figure 26.
Explanation of Solution
Alkene reacts with chlorine and forms chloronium ion intermediate.
The structure of an intermediate is given as,
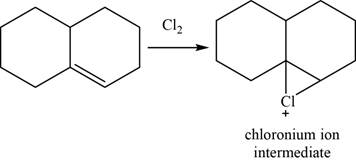
Figure 25
Now, the water molecule acts as a nucleophile that opens the chloronium ion intermediate at more substituted carbon atom. The major product of the given reaction is,
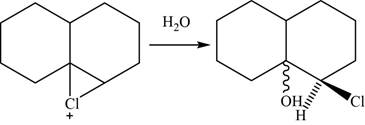
Figure 26
Want to see more full solutions like this?
Chapter 8 Solutions
Organic Chemistry Plus Masteringchemistry With Pearson Etext, Global Edition
- Determine whether each of the following molecules is a hemiacetal, acetal, or neither and select the appropriate box in the table. CH3O OH OH OH hemiacetal acetal neither hemiacetal acetal neither Xarrow_forwardWhat is the missing reactant R in this organic reaction? N N དལ་ད་་ + R • Draw the structure of R in the drawing area below. • Be sure to use wedge and dash bonds if it's necessary to draw one particular enantiomer. Click and drag to start drawing a structure. ㄖˋarrow_forwardDraw the condensed structure of 4-hydroxy-3-methylbutanal. Click anywhere to draw the first atom of your structure.arrow_forward
- Using the bond energy values, calculate the energy that must be supplied or is released upon the polymerization of 755 monomers. If energy must be supplied, provide a positive number; if energy is released, provide a negative number. Hint: Avogadro’s number is 6.02 × 1023.arrow_forward-AG|F=2E|V 3. Before proceeding with this problem you may want to glance at p. 466 of your textbook where various oxo-phosphorus derivatives and their oxidation states are summarized. Shown below are Latimer diagrams for phosphorus at pH values at 0 and 14: Acidic solution -0.93 +0.38 -0.51 -0.06 H3PO4 →H4P206 H3PO3 H3PO2 → P→ PH3 -0.28 -0.50 → -0.50 Basic solution 3-1.12 -1.57 -2.05 -0.89 PO HPO →→H2PO2 P PH3 -1.73 a) Under acidic conditions, H3PO4 can be reduced into H3PO3 directly (-0.28V), or via the formation and reduction of H4P2O6 (-0.93/+0.38V). Calculate the values of AG's for both processes; comment. (3 points) 0.5 PH, 0.0 -0.5- 2 3 9 3 -1.5 -2.0 Pa H,PO H,PO H,PO -3 -1 0 2 4 Oxidation state, N 2 b) Frost diagram for phosphorus under acidic conditions is shown. Identify possible disproportionation and comproportionation processes; write out chemical equations describing them. (2 points) c) Elemental phosphorus tends to disproportionate under basic conditions. Use data in…arrow_forwardThese two reactions appear to start with the same starting materials but result in different products. How do the chemicals know which product to form? Are both products formed, or is there some information missing that will direct them a particular way?arrow_forward
- What would be the best choices for the missing reagents 1 and 3 in this synthesis? 1. PPh3 3 1 2 2. n-BuLi • Draw the missing reagents in the drawing area below. You can draw them in any arrangement you like. • Do not draw the missing reagent 2. If you draw 1 correctly, we'll know what it is. • Note: if one of your reagents needs to contain a halogen, use bromine. Explanation Check Click and drag to start drawing a structure. 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Priva ×arrow_forwardPredict the products of this organic reaction: Explanation Check IN NaBH3CN H+ ? Click and drag to start drawing a structure. D 5 C +arrow_forwardPredict the products of this organic reaction: H3O+ + ? • Draw all the reasonable products in the drawing area below. If there are no products, because no reaction will occur, check the box under the drawing area. • Include both major and minor products, if some of the products will be more common than others. • Be sure to use wedge and dash bonds if you need to distinguish between enantiomers. No reaction. Click and drag to start drawing a structure. dmarrow_forward
- Iarrow_forwardDraw the anti-Markovnikov product of the hydration of this alkene. this problem. Note for advanced students: draw only one product, and don't worry about showing any stereochemistry. Drawing dash and wedge bonds has been disabled for esc esc ☐ Explanation Check F1 1 2 F2 # 3 F3 + $ 14 × 1. BH THE BH3 2. H O NaOH '2 2' Click and drag to start drawing a structure. F4 Q W E R A S D % 905 LL F5 F6 F7 © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility < & 6 7 27 8 T Y U G H I F8 F9 F10 F11 F12 9 0 J K L P + // command option Z X C V B N M H H rol option commandarrow_forwardAG/F-2° V 3. Before proceeding with this problem you may want to glance at p. 466 of your textbook where various oxo-phosphorus derivatives and their oxidation states are summarized. Shown below are Latimer diagrams for phosphorus at pH values at 0 and 14: -0.93 +0.38 -0.50 -0.51 -0.06 H3PO4 →H4P206 →H3PO3 →→H3PO₂ → P → PH3 Acidic solution Basic solution -0.28 -0.50 3--1.12 -1.57 -2.05 -0.89 PO HPO H₂PO₂ →P → PH3 -1.73 a) Under acidic conditions, H3PO4 can be reduced into H3PO3 directly (-0.28V), or via the formation and reduction of H4P206 (-0.93/+0.38V). Calculate the values of AG's for both processes; comment. (3 points) 0.5 PH P 0.0 -0.5 -1.0- -1.5- -2.0 H.PO, -2.3+ -3 -2 -1 1 2 3 2 H,PO, b) Frost diagram for phosphorus under acidic conditions is shown. Identify possible disproportionation and comproportionation processes; write out chemical equations describing them. (2 points) H,PO 4 S Oxidation stale, Narrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


