
Interpretation: The principle behind the working of barometer and manometer has to be explained.
Concept Introduction:
The force acting per unit area is called as Pressure. Differential pressure is indicated by measuring devices i.e. in relative with the atmosphere pressure. This is known as gauge pressure. The calculated pressure can either be positive or negative with respect to the atmospheric pressure. Vacuum is generally known as a negative gauge pressure.
Atmospheric pressure is measured by using an instruments called as barometer and manometer.
Explanation of Solution
Explanation
A barometer consists of Mercury column that is tipped inverted and positioned in a dish containing Mercury. The Mercury in the column moves to and fro. The measure of atmospheric pressure is done by the height of the column. There is a downward force produced by the weight of the Mercury, this downward force pushes the mercury to fall out of the column. But, there is repulsive force that keeps Mercury in the column. This repulsive force is due to the atmospheric gas particles that collide with the surface of the Mercury in the dish; this makes the mercury to get pushed up in the column. The level of Mercury in the column stays constant, when the two repulsive forces are same in strength to each other. The Mercury’s constant height is supported by the atmosphere is a measure of pressure of the atmosphere.
Manometer: A manometer also has two repulsive forces against each other. A force is exerted by the gas molecules in the flask. The repulsive force is seen on the further region of the Mercury filled U tube; that is force exerted by atmospheric gases. The measure of the variation in pressure between the gas in the flask and the atmosphere gives the difference in height of the Mercury in the U tube. The greater or lesser the gas in the flask to the atmosphere pressure can be determined by measuring the difference in the height of the Mercury.
Explain the working of Barometer.
A barometer consists of Mercury column that is tipped inverted and positioned in a dish containing Mercury. The Mercury in the column moves to and fro. The measure of atmospheric pressure is done by the height of the column. There is a downward force produced by the weight of the Mercury, this downward force pushes the mercury to fall out of the column. But, there is repulsive force that keeps Mercury in the column. This repulsive force is due to the atmospheric gas particles that collide with the surface of the Mercury in the dish; this makes the mercury to get pushed up in the column. The level of Mercury in the column stays constant, when the two repulsive forces are same in strength to each other. The Mercury’s constant height is supported by the atmosphere is a measure of pressure of the atmosphere.
A simple barometer is illustrated in the figure 1,
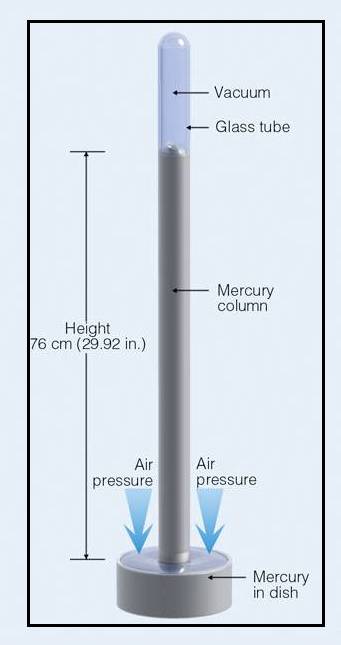
Figure 1: Simple Barometer
Explain the working of Manometer
A manometer also has two repulsive forces against each other. A force is exerted by the gas molecules in the flask. The repulsive force is seen on the further region of the Mercury filled U tube; that is force exerted by atmospheric gases. The measure of the variation in pressure between the gas in the flask and the atmosphere gives the difference in height of the Mercury in the U tube. The greater or lesser the gas in the flask to the atmosphere pressure can be determined by measuring the difference in the height of the Mercury.
Illustration of simple manometer is shown in figure 2,
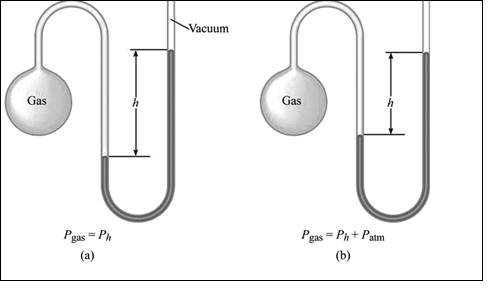
Figure 2: A simple manometer
Conclusion
The principle and working of barometer and manometer are explained.
Want to see more full solutions like this?
Chapter 8 Solutions
Bundle: Chemistry: An Atoms First Approach, Loose-leaf Version, 2nd + OWLv2 with Student Solutions Manual, 4 terms (24 months) Printed Access Card
- Q5: Label each chiral carbon in the following molecules as R or S. Make sure the stereocenter to which each of your R/S assignments belong is perfectly clear to the grader. (8pts) R OCH 3 CI H S 2pts for each R/S HO R H !!! I OH CI HN CI R Harrow_forwardCalculate the proton and carbon chemical shifts for this structurearrow_forwardA. B. b. Now consider the two bicyclic molecules A. and B. Note that A. is a dianion and B. is a neutral molecule. One of these molecules is a highly reactive compound first characterized in frozen noble gas matrices, that self-reacts rapidly at temperatures above liquid nitrogen temperature. The other compound was isolated at room temperature in the early 1960s, and is a stable ligand used in organometallic chemistry. Which molecule is the more stable molecule, and why?arrow_forward
- A mixture of C7H12O2, C9H9OCl, biphenyl and acetone was put together in a gas chromatography tube. Please decide from the GC resutls which correspond to the peak for C7,C9 and biphenyl and explain the reasoning based on GC results. Eliminate unnecessary peaks from Gas Chromatography results.arrow_forwardIs the molecule chiral, meso, or achiral? CI .CH3 H₂C CIarrow_forwardPLEASE HELP ! URGENT!arrow_forward
- Identify priority of the substituents: CH3arrow_forwardHow many chiral carbons are in the molecule? OH F CI Brarrow_forwardA mixture of three compounds Phen-A, Acet-B and Rin-C was analyzed using TLC with 1:9 ethanol: hexane as the mobile phase. The TLC plate showed three spots of R, 0.1 and 0.2 and 0.3. Which of the three compounds (Phen-A; Acet-B or Rin-C) would have the highest (Blank 1), middle (Blank 2) and lowest (Blank 3) spot respectively? 0 CH: 0 CH, 0 H.C OH H.CN OH Acet-B Rin-C phen-A A A <arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning





