
Concept explainers
(a)
Interpretation: The effect on the rate of the given second-order reaction is to be interpreted if the concentration of chlorocyclopentane is tripled and the concentration of sodium hydroxide remains the same.
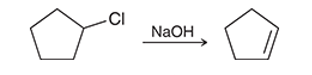
Concept introduction:
The change in reactant concentration over the course of the specified period can be viewed as the pace of a
(b)
Interpretation: The effect on the rate of the given second-order reaction is to be interpreted if the concentration of chlorocyclopentane remains the same and the concentration of sodium hydroxide is doubled.
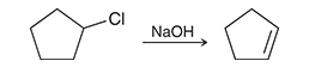
Concept introduction:
The change in reactant concentration over the course of the specified period can be viewed as the pace of a chemical reaction. According to the rate rule, the active concentration of the reactant molecules directly affects how quickly a chemical reaction proceeds. The rate constant is the name for the proportionality constant.
(c)
Interpretation: The effect on the rate of the given second-order reaction is to be interpreted if the concentration of chlorocyclopentane is doubled and the concentration of sodium hydroxide is tripled.
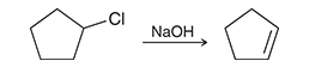
Concept introduction:
The change in reactant concentration over the course of the specified period can be viewed as the pace of a chemical reaction. According to the rate rule, the active concentration of the reactant molecules directly affects how quickly a chemical reaction proceeds. The rate constant is the name for the proportionality constant.
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
ORGANIC CHEM PRINT STUDY GDE & SSM
- € CH3-CH-C-O-CH2-CH2-CH3 + NaOH A? Specifically, in the drawing area below draw the condensed structure of the product, or products, of this reaction. (If there's more than one product, draw them in any arrangement you like, so long as they aren't touching.) If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. Predict the products of this organic reaction: CH3 O Click anywhere to draw the first atom of your structure. No reaction ✓ Garrow_forwardA molecule can have a temporary or permanent depending on the structure and the way the electrons can move. True Falsearrow_forwardedict the products of this organic reaction: CH3 O A CH3-CH-C-NH2 + H2O + HCI ? Specifically, in the drawing area below draw the condensed structure of the product, or products, of this reaction. If there's more than one product, draw them in any arrangement you like, so long as they aren't touching. If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. Click anywhere to draw the first atom of your structure. No Reaction planation Check 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center +arrow_forward
- Draw the mechanism for the following reaction: OH A few notes: CI O • You may assume that each reagent is present in whatever amount you need to draw your mechanism. • To save you some time, one of the starting materials has been copied into the first step of the drawing area. AP Add/Remove step Cl Click and drag to start drawing a structure.arrow_forwardWhat is the missing reactant in this organic reaction? CH3 O [0] R CH3-CH-C-OH Specifically, in the drawing area below draw the condensed structure of R. If there is more than one reasonable answer, you can draw any one of them. If there is no reasonable answer, check the No answer box under the drawing area. Note: the organic equation above only shows the important organic reactant and product. Minor small-molecule reactants or products (like H₂O) are not shown. No Answer Click anywhere to draw the first atom of your structure. ×arrow_forwardPredict the product of this organic reaction: O CH3 A NH3 + HO–C—CH—CH, P+H₂O Specifically, in the drawing area below draw the condensed structure of P. If there is no reasonable possibility for P, check the No answer box under the drawing area. No Answer Click anywhere to draw the first atom of your structure. A 5arrow_forward
- For a reaction to be spontaneous, the Gibbs Free Energy must be less than zero. True or Falsearrow_forwardWhich of the following reactions will be exothermic? a) Reaction has enthalpy less than zero. b) Reaction has enthalpy greater than zero. c) Reaction has entropy less than zero. d) Reaction has entropy equal to zero.arrow_forwardHow many valence electrons are used in predicting the Lewis Dot structure of CH4? a) 8 b) 6 c) 10 d) 2arrow_forward
- How many valence electrons are used in predicting the Lewis Dot structure of CO2? a) 32 b) 26 c) 24 d) 16arrow_forwardHow many valence electrons are used in predicting the Lewis Dot structure of NO2? a) 17 b) 12 c) 5 d) 10arrow_forwardA compound that is bonded with an ionic bond is unlikely to dissolve in water. True or Falsearrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





