
(a)
Interpretation:
The hybridization of the central atom of the molecules with the following molecular geometries has to be predicted.
- (a) Tetrahedral (b) trigonal planar (c) trigonal bipyramidal (d) linear (e) octahedral
Concept Introduction:
Hybridization is a hypothetical concept. It refers to overlapping of atomic orbitals and the resultant orbitals formed are known as hybrid orbitals. An orbital that doesn’t involve in hybridization is termed as unhybridized orbital. After hybridization, the orbitals cannot be distinguished individually. The orientation of the orbitals while overlapping impacts the nature of the bond forms. By knowing the hybridization of central atom in the molecule its geometry can be predicted and vice-versa.
(a)
Answer to Problem 7.52QP
| Molecular Geometry | Hybridization of the central atom | |
| (a) | Tetrahedral |
|
Explanation of Solution
Tetrahedral
A molecule having tetrahedral geometry has the empirical formula
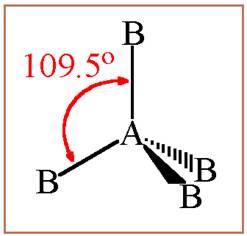
Figure 1
The bond angle between two atoms in a tetrahedral molecule is
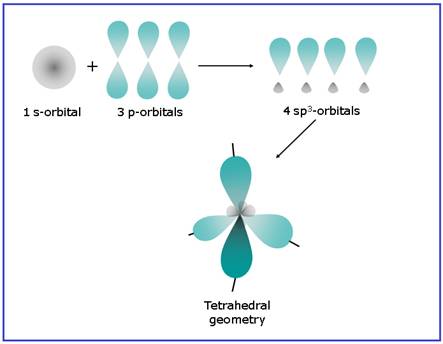
Figure 2
Thus a molecule having tetrahedral geometry has central atom with
(b)
Interpretation:
The hybridization of the central atom of the molecules with the following molecular geometries has to be predicted.
- (a) Tetrahedral (b) trigonal planar (c) trigonal bipyramidal (d) linear (e) octahedral
Concept Introduction:
Hybridization is a hypothetical concept. It refers to overlapping of atomic orbitals and the resultant orbitals formed are known as hybrid orbitals. An orbital that doesn’t involve in hybridization is termed as unhybridized orbital. After hybridization, the orbitals cannot be distinguished individually. The orientation of the orbitals while overlapping impacts the nature of the bond forms. By knowing the hybridization of central atom in the molecule its geometry can be predicted and vice-versa.
(b)
Answer to Problem 7.52QP
| Molecular Geometry | Hybridization of the central atom | |
| (b) | Trigonal planar |
|
Explanation of Solution
Trigonal planar
A molecule having trigonal planar geometry has the empirical formula
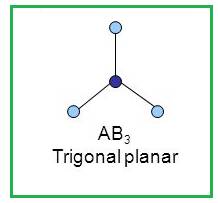
Figure 3
The bond angle between two atoms in a trigonal planar molecule is
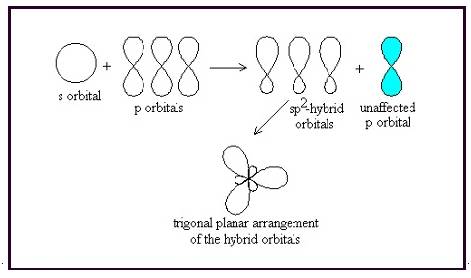
Figure 4
Thus a molecule having trigonal planar geometry has central atom with
(c)
Interpretation:
The hybridization of the central atom of the molecules with the following molecular geometries has to be predicted.
- (a) Tetrahedral (b) trigonal planar (c) trigonal bipyramidal (d) linear (e) octahedral
Concept Introduction:
Hybridization is a hypothetical concept. It refers to overlapping of atomic orbitals and the resultant orbitals formed are known as hybrid orbitals. An orbital that doesn’t involve in hybridization is termed as unhybridized orbital. After hybridization, the orbitals cannot be distinguished individually. The orientation of the orbitals while overlapping impacts the nature of the bond forms. By knowing the hybridization of central atom in the molecule its geometry can be predicted and vice-versa.
(c)
Answer to Problem 7.52QP
| S.No | Molecular Geometry | Hybridization of the central atom |
| (c) | Trigonal bipyramidal |
|
Explanation of Solution
Trigonal bipyramidal
A molecule having trigonal bipyramidal geometry has the empirical formula
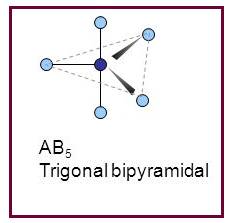
Figure 5
Trigonal bipyramidal molecule has two set of bonds – two axial bonds and three equatorial bonds. The two axial bonds are
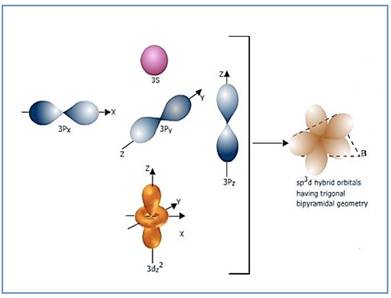
Figure 6
If the d-orbital of the
(d)
Interpretation:
The hybridization of the central atom of the molecules with the following molecular geometries has to be predicted.
- (a) Tetrahedral (b) trigonal planar (c) trigonal bipyramidal (d) linear (e) octahedral
Concept Introduction:
Hybridization is a hypothetical concept. It refers to overlapping of atomic orbitals and the resultant orbitals formed are known as hybrid orbitals. An orbital that doesn’t involve in hybridization is termed as unhybridized orbital. After hybridization, the orbitals cannot be distinguished individually. The orientation of the orbitals while overlapping impacts the nature of the bond forms. By knowing the hybridization of central atom in the molecule its geometry can be predicted and vice-versa.
(d)
Answer to Problem 7.52QP
| S.No | Molecular Geometry | Hybridization of the central atom |
| (d) | Linear |
|
Explanation of Solution
Linear
A molecule having linear geometry has the empirical formula
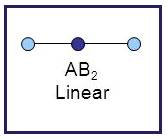
Figure 7
The bond angle between two atoms in linear molecule is
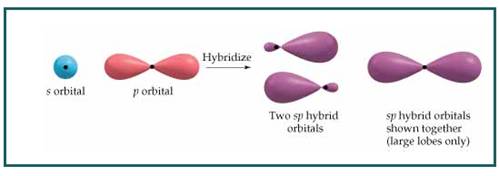
Figure 8
Thus a molecule having linear geometry has central atom with
(e)
Interpretation:
The hybridization of the central atom of the molecules with the following molecular geometries has to be predicted.
- (a) Tetrahedral (b) trigonal planar (c) trigonal bipyramidal (d) linear (e) octahedral
Concept Introduction:
Hybridization is a hypothetical concept. It refers to overlapping of atomic orbitals and the resultant orbitals formed are known as hybrid orbitals. An orbital that doesn’t involve in hybridization is termed as unhybridized orbital. After hybridization, the orbitals cannot be distinguished individually. The orientation of the orbitals while overlapping impacts the nature of the bond forms. By knowing the hybridization of central atom in the molecule its geometry can be predicted and vice-versa.
(e)
Answer to Problem 7.52QP
| S.No | Molecular Geometry | Hybridization of the central atom |
| (e) | Octahedral |
|
Explanation of Solution
Octahedral
A molecule having octahedral geometry has the empirical formula
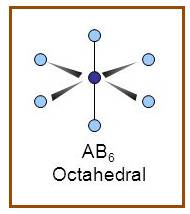
Figure 9
The bond angle between two atoms in octahedral molecule is
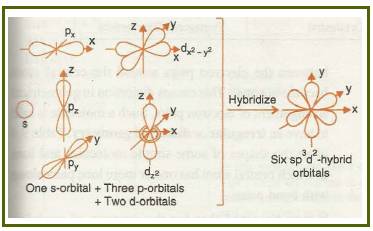
Figure 10
If the d-orbital of the
The hybridization of the central atom of the molecules with the given molecular geometries has been predicted.
Want to see more full solutions like this?
Chapter 7 Solutions
CHEMISTRY: ATOMS FIRST VOL 1 W/CONNECT
- Provide an IUPAC name for each of the compounds shown. (Specify (E)/(Z) stereochemistry, if relevant, for straight chain alkenes only. Pay attention to commas, dashes, etc.) H₁₂C C(CH3)3 C=C H3C CH3 CH3CH2CH CI CH3 Submit Answer Retry Entire Group 2 more group attempts remaining Previous Nextarrow_forwardArrange the following compounds / ions in increasing nucleophilicity (least to most nucleophilic) CH3NH2 CH3C=C: CH3COO 1 2 3 5 Multiple Choice 1 point 1, 2, 3 2, 1, 3 3, 1, 2 2, 3, 1 The other answers are not correct 0000arrow_forwardcurved arrows are used to illustrate the flow of electrons. using the provided starting and product structures, draw the cured electron-pushing arrows for thw following reaction or mechanistic steps. be sure to account for all bond-breaking and bond making stepsarrow_forward
- Using the graphs could you help me explain the answers. I assumed that both graphs are proportional to the inverse of time, I think. Could you please help me.arrow_forwardSynthesis of Dibenzalacetone [References] Draw structures for the carbonyl electrophile and enolate nucleophile that react to give the enone below. Question 1 1 pt Question 2 1 pt Question 3 1 pt H Question 4 1 pt Question 5 1 pt Question 6 1 pt Question 7 1pt Question 8 1 pt Progress: 7/8 items Que Feb 24 at You do not have to consider stereochemistry. . Draw the enolate ion in its carbanion form. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. ⚫ Separate multiple reactants using the + sign from the drop-down menu. ? 4arrow_forwardShown below is the mechanism presented for the formation of biasplatin in reference 1 from the Background and Experiment document. The amounts used of each reactant are shown. Either draw or describe a better alternative to this mechanism. (Note that the first step represents two steps combined and the proton loss is not even shown; fixing these is not the desired improvement.) (Hints: The first step is correct, the second step is not; and the amount of the anhydride is in large excess to serve a purpose.)arrow_forward
- Hi I need help on the question provided in the image.arrow_forwardDraw a reasonable mechanism for the following reaction:arrow_forwardDraw the mechanism for the following reaction: CH3 CH3 Et-OH Et Edit the reaction by drawing all steps in the appropriate boxes and connecting them with reaction arrows. Add charges where needed. Electron-flow arrows should start on the electron(s) of an atom or a bond and should end on an atom, bond, or location where a new bond should be created. H± EXP. L CONT. י Α [1] осн CH3 а CH3 :Ö Et H 0 N о S 0 Br Et-ÖH | P LL Farrow_forward
- 20.00 mL of 0.150 M NaOH is titrated with 37.75 mL of HCl. What is the molarity of the HCl?arrow_forward20.00 mL of 0.025 M HCl is titrated with 0.035 M KOH. What volume of KOH is needed?arrow_forward20.00 mL of 0.150 M NaOH is titrated with 37.75 mL of HCl. What is the molarity of the HCl?arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





