
Concept explainers
Interpretation:
The electron-poor H atom in methanol is to be identified, and the mechanism by which methanol acts as an acid in a proton transfer reaction with
Concept introduction:
An electron poor atom or a molecule is an atom or a site in a molecule that has fewer electrons than the number required for stability. In a proton transfer reaction, a proton is transferred from a Bronsted-Lowry acid (proton donor) to a Bronsted–Lowry base (proton acceptor). In an elementary step, electrons tend to flow from an electron-rich site to an electron-poor site. The curved arrow notation shows the movement of valence electrons.
Answer to Problem 7.1P
The electron-poor H atom in methanol is:
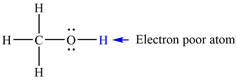
The mechanism by which methanol acts as an acid in a proton transfer reaction with
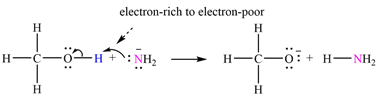
Explanation of Solution
Atoms in
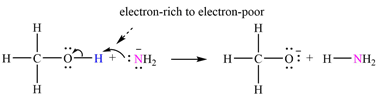
The curved arrows are drawn from the electron-rich site
Want to see more full solutions like this?
Chapter 7 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- Show how you would accomplish the following transformations. More than one step may be required. ow all reagents and all intermediate structures [one ONLY] A. H Br H CH3 NHz CH3 CH3 B. CH3CH2C-Br CH3CH2C-CN CH3 CH3.arrow_forwardShow how you would accomplish the following transformations. More than one step may be required. now all reagents and all intermediate structures [one ONLY] A. H Br H CH3 NHz CH3 CH3 B. CH3CH2C-Br CH3 CH3CH2C-CN CH3arrow_forwardCan I please get help with this?arrow_forward
- C. I, II, III Consider the reaction sequence below to answer the following questions: 0 0 1. NaOEt, EtOH ΕΙΟ OEt 2 Compound X CO₂Et NaOEt, EtOH CO₂Et Br Compound Y A Compound Z A. Compound X, diethyl propanedioate, is more commonly known as a. ethyl acetoacetate acetoacetic ester b. C. oxalic ester d. malonic ester B. Write the complete stepwise mechanism for the conversion of Compound X into Compound Y. Show all electron flow with arrows and draw all intermediate structures.arrow_forwardDiethyl malonate can be prepared by the following reaction sequence. Draw the structures of each of the missing intermediates in the boxes provided EtO 0 H3C 11 C 1. Br₂ PBr OH 2 H₂O 010 0 CH3CH₂OH C CH2 OEt Ha CH3CH2OH на NaCN H₂SO4 NC H₂O, heat CH2 OCH2CH3arrow_forwardShow how you would accomplish each of the following transformations. More than one step may be quired. Show all reagents and all intermediate structures. [three only] A. 0 CH3 B. C. D. H 0 0 OCH 3 CH₂CO₂CH2CH3 H3C ➤ HN C NO₂ Clarrow_forward
- Choose the BEST reagent for carrying out each of the following conversions. A. CO₂CH3 CO₂CH3 0 CO₂H a. LiAlH4, ether C. CrO3, pyridine B. 0 H a. C. NaBH4, ethanol NaOH, H2O CO₂H OH HD b. NaBH4, ethanol d. H₂/Pd CH₂OH b. CH₂PPh3 d. All of the abovearrow_forwardWrite the complete stepwise mechanism for the acid-catalyzed hydrolysis of the following amide to yield mandelic acid. Show all electron flow with arrows and draw the structures of all intermediate species. OH H-OH₂ CnH2 :0: OH C OH + NH4 10: The purpose of the acid catalyst in the hydrolysis of an amide is: to enhance the electrophilicity of the amide carbonyl carbon a. to enhance the nucleophilicity of the water molecule b. C. to enhance the electrophilicity of the water molecule d. to shift the equilibrium of the reactionarrow_forward1.arrow_forward
- Can I please get help with this?arrow_forward. Provide IUPAC names for each of the following structures OR draw structures corresponding to each of the following names: [Three only]kk a. H₂N- 0 COCH2CH3 benzocaine b. What is the correct structure for phenylbenzoate? C a. 0 C-O O b. H3C-C-O 0 0 C-O-CH3 d. CH₂O C-CHZ c. Acetyl chloride d. 3,4,5-trimethoxybenzoyl chloridearrow_forward. Draw structures corresponding to each of the following names or Provide IUPAC names for each of the ollowing structures [for 4 ONLY]. A. 2-propylpentanoic acid. B. m-chlorobenzoic acid. C. 0 0 HOC(CH2) COH glutaricadd D. E. F. 0 OH HO OH HO INCO salicylicadd H3C CH3 C=C tgicadd H COOH CH₂C=N 4arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning



