
Concept explainers
(a)
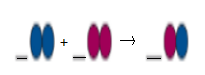
Interpretation: To balance the following reaction by adding coefficients and identify the type of reaction.
Concept Introduction:
Mass can neither be created nor be destroyed.
A
Types of chemical reaction:
- Combination reaction: Here two or more reactants combines to form a single product
- Decomposition product: In this type of reaction, one reactant is breakdown into two or more products
- Displacement reaction: This is a type of reaction where more reactive element displaces the less reactive element.
- Double-displacement reaction: In this type of reaction, two compounds react in such a way that cations and anions of two reactants interchange their place.
Balanced equation will be:
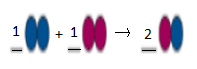
This is a type of combination reaction.
In the given reaction, the reactant contains two blue atoms and two pink atoms.
So,
Product should contain 2 blue atoms and 2 pink atoms.
But in product side, we have a molecule having only one blue atom and 1 pink atom. Therefore, we need to multiply the product with 2 to make it balance.
In this chemical reaction,
2 reactants are combined to form a single product. Hence, it is a type of combination reaction.
(a)
Answer to Problem 7.101UTC
Balanced equation will be:
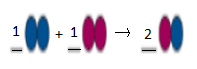
This is a type of combination reaction.
Explanation of Solution
In the given reaction, the reactant contains two blue atoms and two pink atoms.
So,
Product should contain 2 blue atoms and 2 pink atoms.
But in product side, we have a molecule having only one blue atom and 1 pink atom. Therefore, we need to multiply the product with 2 to make it balance.
In this chemical reaction,
2 reactants are combined to form a single product. Hence, it is a type of combination reaction.
(b)
Interpretation: To balance the following reaction by adding coefficients and identify the type of reaction.
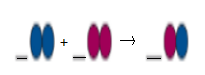
Concept Introduction:
Mass can neither be created nor be destroyed.
A chemical reaction should be well balance i.e. the number of atoms of each element in reactant side must be equal to number of atoms in product side.
Types of chemical reaction:
- Combination reaction: Here two or more reactants combines to form a single product
- Decomposition product: In this type of reaction, one reactant is breakdown into two or more products
- Displacement reaction: This is a type of reaction where more reactive element displaces the less reactive element.
- Double-displacement reaction: In this type of reaction, two compounds react in such a way that cations and anions of two reactants interchange their place.
(b)
Answer to Problem 7.101UTC
Balanced equation will be:
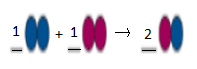
This is a type of decomposition reaction.
Explanation of Solution
In the given reaction, the reactant side contains 2 blue atoms and 1 pink atom.
So, product side should contain 2 blue atoms and 1 pink atom.
But in product side, we have two different products as:
First product has 2 blue atoms and second molecule has 2 pink atoms.
If we multiply the reactant and first product by 2, then reaction will be balance as now both reactants and product have 4 blue atoms and 2 pink atoms respectively.
Here one reactant breaks down into two products. Thus, it is a type of decomposition reaction.
Want to see more full solutions like this?
Chapter 7 Solutions
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
- Nonearrow_forwardTransmitance 3. Which one of the following compounds corresponds to this IR spectrum? Point out the absorption band(s) that helped you decide. OH H3C OH H₂C CH3 H3C CH3 H3C INFRARED SPECTRUM 0.8- 0.6 0.4- 0.2 3000 2000 1000 Wavenumber (cm-1) 4. Consider this compound: H3C On the structure above, label the different types of H's as A, B, C, etc. In table form, list the labeled signals, and for each one state the number of hydrogens, their shifts, and the splitting you would observe for these hydrogens in the ¹H NMR spectrum. Label # of hydrogens splitting Shift (2)arrow_forwardNonearrow_forward
- Draw the Lewis structure of C2H4Oarrow_forwarda) 5. Circle all acidic (and anticoplanar to the Leaving group) protons in the following molecules, Solve these elimination reactions, and identify the major and minor products where appropriate: 20 points + NaOCH3 Br (2 productarrow_forwardNonearrow_forward
- Dr. Mendel asked his BIOL 260 class what their height was and what their parent's heights were. He plotted that data in the graph below to determine if height was a heritable trait. A. Is height a heritable trait? If yes, what is the heritability value? (2 pts) B. If the phenotypic variation is 30, what is the variation due to additive alleles? (2 pts) Offspring Height (Inches) 75 67.5 60 52.5 y = 0.9264x + 4.8519 55 60 65 MidParent Height (Inches) 70 75 12pt v V Paragraph B IUA > AT2 v Varrow_forwardExperiment: Each team will be provided with 5g of a mixture of acetanilide and salicylic acid. You will divide it into three 1.5 g portions in separate 125 mL Erlenmeyer flasks savıng some for melting point analysis. Dissolve the mixture in each flask in ~60mL of DI water by heating to boiling on a hotplate. Take the flasks off the hotplate once you have a clear solution and let them stand on the bench top for 5 mins and then allow them to cool as described below. Sample A-Let the first sample cool slowly to room temperature by letting it stand on your lab bench, with occasional stirring to promote crystallization. Sample B-Cool the second sample 1n a tap-water bath to 10-15 °C Sample C-Cool the third sample in an ice-bath to 0-2 °C Results: weight after recrystalization and melting point temp. A=0.624g,102-115° B=0.765g, 80-105° C=1.135g, 77-108 What is the percent yield of A,B, and C.arrow_forwardRel. Intensity Q 1. Which one of the following is true of the compound whose mass spectrum is shown here? Explain how you decided. 100 a) It contains chlorine. b) It contains bromine. c) It contains neither chlorine nor bromine. 80- 60- 40- 20- 0.0 0.0 TT 40 80 120 160 m/z 2. Using the Table of IR Absorptions how could you distinguish between these two compounds in the IR? What absorbance would one compound have that the other compound does not? HO CIarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





