
Organic Chemistry (6th Edition)
6th Edition
ISBN: 9781260119107
Author: Janice Gorzynski Smith
Publisher: McGraw Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7, Problem 68P
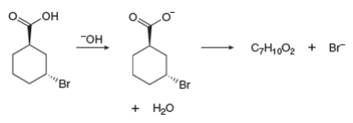
When a single compound contains both a nucleophile and a leaving group, an intramolecular reaction may occur. With this in mind, draw the product of the following reaction.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Explain the importance of having a sampling plan with respect to food analysis.
Explain the importance of having a sampling plan with respect to food analysis. Provide examples.
Please predict the products for each of the
following reactions.
Clearly show the regiochemistry (Markovnikov
vs anti-Markovnikov) and stereochemistry
(syn- vs anti- or both).
If a mixture of enantiomers is formed, please
draw all the enantiomers.
cold
KMnO4, NaOH
2. DMS
1. 03
CH3OH
Br2
1.
03
2. (CH3)2S
H₂
Pd or Pt (catalyst)
HBr
18
19
20 1
HBr
ROOR (peroxide)
H₂O
H₂SO4
HCI
HI
17
16
6
15
MCPBA
1. BH3 THF
2. H₂O2, NaOH
1. OsO4
2. H₂O₂
110
CH3CO₂H
(peroxyacid)
1. MCPBA
2. H₂O*
Br2
H₂O
BH3 THF
B12
EtOH
Pd or Ni (catalyst)
D₂ (deuterium)
Bra
A
B
C
D
H
OH
H
OH
OH
H
OH
α α α
OH
H
OH
OH
фон
d
H
"H
Briefly indicate the models that describe the structure of the interface: Helmholtz-Perrin, Gouy-Chapman, Stern and Grahame models.
Chapter 7 Solutions
Organic Chemistry (6th Edition)
Ch. 7.1 - Problem 7.1 Telfairine, a naturally occurring...Ch. 7.2 - Give the IUPAC name for each compound. a. b. c. d.Ch. 7.2 - Prob. 3PCh. 7.3 - An sp3 hybridized CCl bond is more polar than an...Ch. 7.4 - Prob. 5PCh. 7.6 - Prob. 6PCh. 7.7 - Prob. 10PCh. 7.8 - Prob. 15PCh. 7.8 - Prob. 16PCh. 7.8 - Prob. 17P
Ch. 7.11 - Prob. 18PCh. 7.11 - Prob. 19PCh. 7.11 - Draw the product of each SN2 reaction and indicate...Ch. 7.11 - Prob. 21PCh. 7.11 - Prob. 22PCh. 7.12 - What happens to the rate of an SN1 reaction under...Ch. 7.12 - Draw the products of each SN1 reaction and...Ch. 7.13 - Classify each carbocation as 1,2, or 3. a. b. c....Ch. 7.15 - Problem 7.30 For each alkyl halide and...Ch. 7.15 - Prob. 30PCh. 7.15 - Prob. 31PCh. 7.15 - Prob. 32PCh. 7.15 - Prob. 33PCh. 7.15 - Prob. 34PCh. 7 - Give the IUPAC name for each compound, including...Ch. 7 - Draw the products of each nucleophilic...Ch. 7 - Prob. 50PCh. 7 - Prob. 51PCh. 7 - 7.53 Consider the following reaction.
Draw a...Ch. 7 - Prob. 57PCh. 7 - Prob. 58PCh. 7 - Consider the following SN1 reaction. a.Draw a...Ch. 7 - 7.59 Pick the reactant or solvent in each part...Ch. 7 - Draw the products of each SN1 reaction and...Ch. 7 - Prob. 62PCh. 7 - Prob. 63PCh. 7 - Draw a stepwise, detailed mechanism for the...Ch. 7 - When a single compound contains both a nucleophile...Ch. 7 - Prob. 69PCh. 7 - Prob. 70PCh. 7 - Draw a stepwise, detailed mechanism f or the...Ch. 7 - Prob. 72PCh. 7 - Prob. 78P
Additional Science Textbook Solutions
Find more solutions based on key concepts
The validity of a scientific law.
Physical Universe
Why is it unlikely that two neighboring water molecules would be arranged like this?
Campbell Biology (11th Edition)
What were the major microbiological interests of Martinus Beijerinck and Sergei Winogradsky? It can be said tha...
Brock Biology of Microorganisms (15th Edition)
Separate the list P,F,V,,T,a,m,L,t, and V into intensive properties, extensive properties, and nonproperties.
Fundamentals Of Thermodynamics
Why are mutants used as test organisms in the Ames test?
Laboratory Experiments in Microbiology (12th Edition) (What's New in Microbiology)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Using Benzene as starting materid show how each of the Following molecules Contel Ve syntheswed CHI 9. b -50311 с CHY 503H Ночто d. อ •NOV e 11-0-650 NO2arrow_forwardThe molecule PYRIDINE, 6th electrons and is therefore aromatre and is Assigned the Following structure contering Since aromatk moleculoy undergo electrophilic anomatic substitution, Pyridine shodd undergo The Following reaction + HNO3 12504 a. write all of the possible Mononitration Products that could Result From this reaction 18. Bared upon the reaction mechanison determime which of these producty would be the major Product of the hegetionarrow_forwarda. Explain Why electron withdrawing groups tend to be meta-Directors. Your answer Should lyclude all apropriate. Resonance contributing Structures fo. Explain why -ll is an outho -tura drccton even though chlorine has a very High Electronegativityarrow_forward
- 9. Write Me product as well as the reaction Mechanism For each of the Following Vanctions +H₂504 4.50+ T C. +212 Fellz 237 b. Praw the potential energy Diagrams For each OF Mese Rauctions and account For any differences that appear in the two potential Puergy Diagrams which of here two reactions 19 Found to be Reversable, Rationalice your answer based upon the venation mechanisms and the potential energy diagrams.arrow_forward9. Write Me product as well as the reaction Mechanism For each of the Following Veritious +H2504 4.50+ + 1/₂ Felly ◎+ 7 b. Praw he potential energy Diagrams For each OF Mese Ronctions and account for any differences that appeak in the two potential Puergy Diagramsarrow_forwardDraw the major product of this reaction. Ignore inorganic byproducts. Incorrect, 3 attempts remaining 1. excess Br2, NaOH 2. neutralizing workup Qarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY