
Organic Chemistry (6th Edition)
6th Edition
ISBN: 9781260119107
Author: Janice Gorzynski Smith
Publisher: McGraw Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6.3, Problem 5P
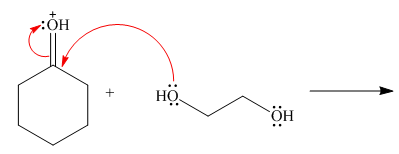
Follow the curved arrows and draw the products of the following reaction.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
From the given compound, choose the proton that best fits each given description.
a
CH2
CH 2
Cl
b
с
CH2
F
Most shielded:
(Choose one)
Least shielded:
(Choose one)
Highest chemical shift:
(Choose one)
Lowest chemical shift:
(Choose one)
×
Consider this molecule:
How many H atoms are in this molecule?
How many different signals could be found in its 1H NMR spectrum?
Note: A multiplet is considered one signal.
For each of the given mass spectrum data, identify whether the compound contains chlorine, bromine, or neither.
Compound
m/z of M* peak
m/z of M
+ 2 peak
ratio of M+ : M
+ 2 peak
Which element is present?
A
122
no M
+ 2 peak
not applicable
(Choose one)
B
78
80
3:1
(Choose one)
C
227
229
1:1
(Choose one)
Chapter 6 Solutions
Organic Chemistry (6th Edition)
Ch. 6.2 - Prob. 2PCh. 6.3 - Problem 6.3 By taking into account...Ch. 6.3 - Problem 6.4 Use curved arrows to show the movement...Ch. 6.3 - Problem 6.5 Follow the curved arrows and draw the...Ch. 6.4 - Prob. 6PCh. 6.4 - Problem 6.7 Use the values in Table 6.2 to...Ch. 6.4 - Prob. 8PCh. 6.5 - aWhich Keq corresponds to a negative value of G,...Ch. 6.5 - Given each of the following values, is the...Ch. 6.5 - Given each of the following values, is the...
Ch. 6.5 - The equilibrium constant for the conversion of the...Ch. 6.6 - Prob. 13PCh. 6.6 - For a reaction with H=40kJ/mol, decide which of...Ch. 6.6 - For a reaction with H=20kJ/mol, decide which of...Ch. 6.7 - Draw an energy diagram for a reaction in which the...Ch. 6.7 - Prob. 17PCh. 6.7 - Prob. 18PCh. 6.8 - Problem 6.19 Consider the following energy...Ch. 6.8 - Draw an energy diagram for a two-step reaction,...Ch. 6.9 - Which value if any corresponds to a faster...Ch. 6.9 - Prob. 22PCh. 6.9 - Problem 6.23 For each rate equation, what effect...Ch. 6.9 - Prob. 24PCh. 6.10 - Identify the catalyst in each equation. a....Ch. 6 - Draw the products of homolysis or heterolysis of...Ch. 6 - Explain why the bond dissociation energy for bond...Ch. 6 - Classify each transformation as substitution,...Ch. 6 - Prob. 29PCh. 6 - 6.31 (a) Add curved arrows for each step to show...Ch. 6 - Prob. 35PCh. 6 - 6.39. a. Which value corresponds to a negative...Ch. 6 - Prob. 40PCh. 6 - For which of the following reaction is S a...Ch. 6 - Prob. 42PCh. 6 - Prob. 43PCh. 6 - 6.44 Consider the following reaction: .
Use curved...Ch. 6 - Prob. 45PCh. 6 - 6.50 The conversion of acetyl chloride to methyl...Ch. 6 - Prob. 50PCh. 6 - Prob. 53P
Additional Science Textbook Solutions
Find more solutions based on key concepts
1. Rub your hands together vigorously. What happens? Discuss the energy transfers and transformations that take...
College Physics: A Strategic Approach (3rd Edition)
Define histology.
Fundamentals of Anatomy & Physiology (11th Edition)
Why are mutants used as test organisms in the Ames test?
Laboratory Experiments in Microbiology (12th Edition) (What's New in Microbiology)
2. Define equilibrium population. Outline the conditions that must be met for a population to stay in genetic e...
Biology: Life on Earth (11th Edition)
Gregor Mendel never saw a gene, yet he concluded that some inherited factors were responsible for the patterns ...
Campbell Essential Biology (7th Edition)
Choose the best answer to each of the following. Explain your reasoning. If Earth were twice as far as it actua...
Cosmic Perspective Fundamentals
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Don't used hand raiting and don't used Ai solutionarrow_forward2' P17E.6 The oxidation of NO to NO 2 2 NO(g) + O2(g) → 2NO2(g), proceeds by the following mechanism: NO + NO → N₂O₂ k₁ N2O2 NO NO K = N2O2 + O2 → NO2 + NO₂ Ко Verify that application of the steady-state approximation to the intermediate N2O2 results in the rate law d[NO₂] _ 2kk₁[NO][O₂] = dt k+k₁₂[O₂]arrow_forwardPLEASE ANSWER BOTH i) and ii) !!!!arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT
How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY