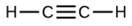Chemistry Atoms First2e
2nd Edition
ISBN: 9781947172647
Author: OpenStax
Publisher: OpenStax College
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6, Problem 6E
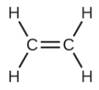
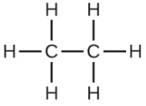
Which molecule has a molecular mass of 28.05 amu?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Michael Reactions
19.52 Draw the products from the following Michael addition reactions.
1.
H&C CH
(a)
i
2. H₂O*
(b)
OEt
(c)
EtO
H₂NEt
(d)
ΕΙΟ
+
1. NaOEt
2. H₂O'
H
H
1. NaOEt
2. H₂O*
Rank the labeled protons (Ha-Hd) in order of increasing acidity, starting with the least acidic.
НОН НЬ
OHd
Онс
Can the target compound at right be efficiently synthesized in good yield from the unsubstituted benzene at left?
?
starting
material
target
If so, draw a synthesis below. If no synthesis using reagents ALEKS recognizes is possible, check the box under the drawing area.
Be sure you follow the standard ALEKS rules for submitting syntheses.
+ More...
Note for advanced students: you may assume that you are using a large excess of benzene as your starting material.
C
:0
T
Add/Remove step
G
Chapter 6 Solutions
Chemistry Atoms First2e
Ch. 6 - What is the total mass (amu) of carbon in each of...Ch. 6 - What is the total mass of hydrogen in each of the...Ch. 6 - Calculate the molecular or formula mass of each of...Ch. 6 - Determine the molecular mass of the following...Ch. 6 - Determine the molecular mass of the following...Ch. 6 - Which molecule has a molecular mass of 28.05 amu?Ch. 6 - What information do we need to determine the...Ch. 6 - Calculate the following to four significant...Ch. 6 - Determine the following to four significant...Ch. 6 - Determine the percent ammonia, NH3, in Co(...
Ch. 6 - Determine the percent water in CuSO45H2O to three...Ch. 6 - Determine the empirical formulas for compounds...Ch. 6 - Determine the empirical formulas for compounds...Ch. 6 - A compound of carbon and hydrogen contains 92.3% C...Ch. 6 - Dichioroethane, a compound that is often used for...Ch. 6 - Determine the empirical and molecular formula for...Ch. 6 - Polymers are large molecules composed of simple...Ch. 6 - A major textile dye manufacturer developed a new...Ch. 6 - Explain what changes and what stays the same when...Ch. 6 - What information do we need to calculate the...Ch. 6 - A 200-mL sample and a 400-mL sample of a solution...Ch. 6 - Determine the molarity for each of the following...Ch. 6 - Determine the molarity of each of the following...Ch. 6 - Consider this question: What is the mass of the...Ch. 6 - Consider this question: What is the mass of solute...Ch. 6 - Calculate the number of moles and the mass of the...Ch. 6 - Calculate the number of moles and the mass of the...Ch. 6 - Consider this question: What is the molarity of...Ch. 6 - Consider this question: What is the molarity of...Ch. 6 - Calculate the molarity of each of the following...Ch. 6 - Calculate the molarity of each of the following...Ch. 6 - There is about 1.0 g of calcium, as Ca2+, in LO L...Ch. 6 - What volume of a 1.00MFe( NO3)3 solution can be...Ch. 6 - If 0.1718 L of a 0.3556-M C3H7OH solution is...Ch. 6 - If 4.12 L of a 0.850 MH3PO4 solution is be diluted...Ch. 6 - What volume of a 0.33MC12H22O11 solution can be...Ch. 6 - What is the concentration of the NaCl solution...Ch. 6 - What is the molarity of the diluted solution when...Ch. 6 - What is the final concentration of the solution...Ch. 6 - A 2.00-L bottle of a solution of concentrated HCl...Ch. 6 - An experiment in a general chemistry laboratory...Ch. 6 - What volume of a 0.20MK2SO4 solution contains 57 g...Ch. 6 - The US Environmental Protection Agency (EPA)...Ch. 6 - Consider this question: What mass of a...Ch. 6 - What mass of a 4.00% NaOH solution by mass...Ch. 6 - What mass of solid NaOH (97.0% NaOH by mass) is...Ch. 6 - What mass of HCl is contained in 45.0 mL of an...Ch. 6 - The hardness of water (hardness count) is usually...Ch. 6 - The hardness of water (hardness count) is usually...Ch. 6 - In Canada and the United Kingdom, devices that...Ch. 6 - A throat spray is 1.40% by mass phenol, C6H5OH, in...Ch. 6 - Copper(I) iodide (CuI) is often added to table...Ch. 6 - A cough syrup contains 5.0% ethyl alcohol, C2H5OH,...Ch. 6 - D5W is a solution used as an intravenous fluid. It...Ch. 6 - Find the molarity of a 40.0% by mass aqueous...
Additional Science Textbook Solutions
Find more solutions based on key concepts
CAUTION Why does the presence of extinct forms and transitional features in the fossil record support the patte...
Biological Science (6th Edition)
Fibrous connective tissue consists of ground substance and fibers that provide strength, support, and flexibili...
Human Biology: Concepts and Current Issues (8th Edition)
Why is an endospore called a resting structure? Of what advantage is an endospore to a bacterial cell?
Microbiology: An Introduction
Fill in the blanks: a. The wrist is also known as the _________ region. b. The arm is also known as the _______...
Human Anatomy & Physiology (2nd Edition)
4. Three groups of nonvascular plants are _______, ______, and _______. Three groups of seedless vascular plant...
Biology: Life on Earth (11th Edition)
Define histology.
Fundamentals of Anatomy & Physiology (11th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The following equations represent the formation of compound MX. What is the AH for the electron affinity of X (g)? X₂ (g) → 2X (g) M (s) → M (g) M (g) M (g) + e- AH = 60 kJ/mol AH = 22 kJ/mol X (g) + e-X (g) M* (g) +X (g) → MX (s) AH = 118 kJ/mol AH = ? AH = -190 kJ/mol AH = -100 kJ/mol a) -80 kJ b) -30 kJ c) -20 kJ d) 20 kJ e) 156 kJarrow_forwardA covalent bond is the result of the a) b) c) d) e) overlap of two half-filled s orbitals overlap of a half-filled s orbital and a half-filled p orbital overlap of two half-filled p orbitals along their axes parallel overlap of two half-filled parallel p orbitals all of the abovearrow_forwardCan the target compound at right be efficiently synthesized in good yield from the unsubstituted benzene at left? starting material target If so, draw a synthesis below. If no synthesis using reagents ALEKS recognizes is possible, check the box under the drawing area. Be sure you follow the standard ALEKS rules for submitting syntheses. + More... Note for advanced students: you may assume that you are using a large excess of benzene as your starting material. C T Add/Remove step X ноarrow_forward
- Which one of the following atoms should have the largest electron affinity? a) b) c) d) 으으 e) 1s² 2s² 2p6 3s¹ 1s² 2s² 2p5 1s² 2s² 2p 3s² 3p² 1s² 2s 2p 3s² 3p6 4s2 3ds 1s² 2s² 2p6arrow_forwardAll of the following are allowed energy levels except _. a) 3f b) 1s c) 3d d) 5p e) 6sarrow_forwardA student wants to make the following product in good yield from a single transformation step, starting from benzene. Add any organic reagents the student is missing on the left-hand side of the arrow, and any addition reagents that are necessary above or below the arrow. If this product can't be made in good yield with a single transformation step, check the box below the drawing area. Note for advanced students: you may assume that an excess of benzene is used as part of the reaction conditions. : ☐ + I X This product can't be made in a single transformation step.arrow_forward
- Ppplllleeeaaasssseeee helllppp wiithhh thisss Organic chemistryyyyyy I talked like this because AI is very annoyingarrow_forwardName the family to which each organic compound belongs. The first answer has been filled in for you. compound CH₂ || CH3-C-NH2 0 ။ CH3-C-CH₂ CH=O–CH=CH, CH₂ HO CH2-CH2-CH-CH3 family amine Darrow_forward1b. Br LOHarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Step by Step Stoichiometry Practice Problems | How to Pass ChemistryMole Conversions Made Easy: How to Convert Between Grams and Moles; Author: Ketzbook;https://www.youtube.com/watch?v=b2raanVWU6c;License: Standard YouTube License, CC-BY