
Chemistry: The Central Science Plus Mastering Chemistry with Pearson eText -- Access Card Package (14th Edition)
14th Edition
ISBN: 9780134292816
Author: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Matthew E. Stoltzfus
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 6, Problem 53E
Classify the following statements as either true or false:
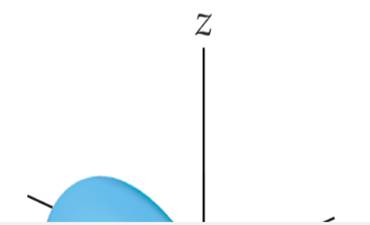
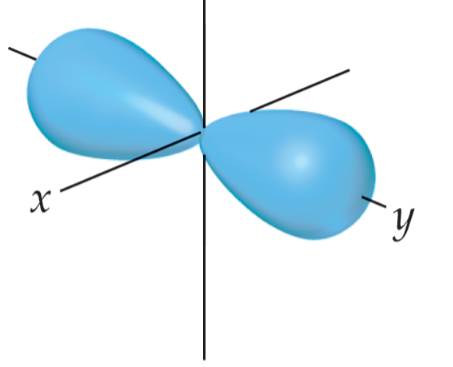
- In a contour representation of an orbital, such as the one shown here for a 2p orbital, the electron is confined to move about the nucleus on the outer surface of the shape.
- The probability density [0(r)]2 gives the probability of finding the electron at a specific distance from the nucleus.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The U. S. Environmental Protection Agency (EPA) sets limits on healthful levels of air pollutants. The maximum level that the EPA considers safe for lead air pollution is 1.5 μg/m3 . If your lungs were filled with air containing this level of lead, how many lead atoms would be in your lungs? (Assume a total lung volume of 5.40 L
During a(n) ________ process, energy is transferred from the system to the surroundings.
exothermic
endothermic
thermodynamic
thermochemical
physical
Use the following information to determine the enthalpy for the reaction shown below.
→
S(s) + O2(g) SO2(9)
ΔΗ Π
?
Reference reactions:
S(s) + O2(g)
SO3(9)
2SO2(g) + O2(g) → 2SO3(g)
AHxn
=
-395kJ
AHrxn
= ―
-198kJ
Chapter 6 Solutions
Chemistry: The Central Science Plus Mastering Chemistry with Pearson eText -- Access Card Package (14th Edition)
Ch. 6.1 - A source of electromagnetic radiation produces...Ch. 6.1 - Which type of visible light has a longer...Ch. 6.1 - Consider the following three statements: For any...Ch. 6.1 - A laser used in orthopedic spine surgery produces...Ch. 6.2 - Prob. 6.3.1PECh. 6.2 -
A laser emits light that has a frequency of 4.69...Ch. 6.3 - Prob. 6.4.1PECh. 6.3 -
For each of the following transitions, give the...Ch. 6.4 - Consider the following three moving objects: a...Ch. 6.4 - Calculate the velocity of a neutron whose de...
Ch. 6.5 - An orbital has n = 4 and ml = 0, 1, 2, 3 -3, - 2,...Ch. 6.5 -
What is the designation for the subshell with = 5...Ch. 6.8 - How many of the elements in the second row of the...Ch. 6.8 - Write the electron configuration for silicon,...Ch. 6.9 - A certain atom has an ns2np2electron configuration...Ch. 6.9 -
Which group of elements is characterized by an...Ch. 6.9 -
A certain atom has a [noble gas]5s24d105p4...Ch. 6.9 - Prob. 6.9.2PECh. 6 - In this chapter, we have learned about the...Ch. 6 - The speed of sound in dry air at 20°C is 343 m/s...Ch. 6 -
6.2 A popular kitchen appliance produces...Ch. 6 - 6.3 The following diagrams represent two...Ch. 6 -
6.4 Stars do not all have the same temperature....Ch. 6 - 6 5 The familiar phenomenon of a rainbow results...Ch. 6 -
6.7 A certain quantum mechanical system has the...Ch. 6 - Consider the three electronic transitions in a...Ch. 6 - Prob. 8ECh. 6 -
6.9 The contour representation of one of the...Ch. 6 -
6.10 The accompanying drawing shows a contour...Ch. 6 -
8.11 Four possible electron configurations for a...Ch. 6 -
6.12 State where in the periodic table these...Ch. 6 - Prob. 13ECh. 6 -
6.14
a What is the relationship between the...Ch. 6 - Label each of the following statements as true or...Ch. 6 - Determine which of the following statements are...Ch. 6 - Arrange the following kinds of electromagnetic...Ch. 6 - List the following types of electromagnetic...Ch. 6 - What is the frequency of radiation that has a...Ch. 6 - What is the frequency of radiation whose...Ch. 6 - A laser pointer used in a lecture hall emits light...Ch. 6 - It is possible to convert radiant energy into...Ch. 6 - If human height were quantized in 1-foot...Ch. 6 - Einstein's 1905 paper on the photoelectric effect...Ch. 6 - Calculate the energy of a photon of...Ch. 6 - Prob. 26ECh. 6 - Prob. 27ECh. 6 - An AM radio station broadcasts at 1010 kHz, and...Ch. 6 - One type of sunburn occurs on exposure to UV light...Ch. 6 - Prob. 30ECh. 6 - Prob. 31ECh. 6 - A stellar object is emitting radiation at 3.55 mm....Ch. 6 - Molybdenum metal must absorb radiation with a...Ch. 6 - Titanium metal requires a photon with a minimum...Ch. 6 - Prob. 35ECh. 6 - Classify each of the following statements as...Ch. 6 - Prob. 37ECh. 6 -
6 38 Indicate whether energy is emitted or...Ch. 6 - Using Equation 6.5. calculate the energy of an...Ch. 6 - Prob. 40ECh. 6 - The visible emission lines observed by Balmer all...Ch. 6 - Prob. 42ECh. 6 - Prob. 43ECh. 6 - The hydrogen atom can absorb light of wavelength...Ch. 6 - Prob. 45ECh. 6 - Prob. 46ECh. 6 - Use the de Brogue relationship to determine the...Ch. 6 - Prob. 48ECh. 6 - Neutron diffraction is an important technique for...Ch. 6 - The electron microscope has been widely used to...Ch. 6 - Using Heisenberg's uncertainty principle,...Ch. 6 - Prob. 52ECh. 6 - Classify the following statements as either true...Ch. 6 - 0 1 2 3 4 5 6 7 8 9 10 Distance from the nucleus,...Ch. 6 - Prob. 55ECh. 6 - Prob. 56ECh. 6 - Prob. 57ECh. 6 - Give the values for n, I,and mlfor each orbital in...Ch. 6 - Prob. 59ECh. 6 - Prob. 60ECh. 6 - Which of the following represent impossible...Ch. 6 - For the table that follows, write which orbital...Ch. 6 - Sketch the shape and orientation of the following...Ch. 6 - Prob. 64ECh. 6 - Prob. 65ECh. 6 - Prob. 66ECh. 6 - Prob. 67ECh. 6 - Prob. 68ECh. 6 - Two possible electron configurations for an Li...Ch. 6 -
6.70 An experiment called the Stern—Gerlach...Ch. 6 - Prob. 71ECh. 6 - Prob. 72ECh. 6 - What are "valence electrons"? What are "core...Ch. 6 - For each element, indicate the number of valence...Ch. 6 - Write the condensed electron configurations for...Ch. 6 - Write the condensed electron configurations for...Ch. 6 - Identify the specific element that corresponds to...Ch. 6 - Identify the group of elements that corresponds to...Ch. 6 - The following do not represent valid ground-state...Ch. 6 - Prob. 80ECh. 6 - Prob. 81AECh. 6 - Prob. 82AECh. 6 - Prob. 83AECh. 6 - Prob. 84AECh. 6 - Prob. 85AECh. 6 - Prob. 86AECh. 6 - Prob. 87AECh. 6 - In an experiment to study the photoelectric...Ch. 6 - Prob. 89AECh. 6 - Prob. 90AECh. 6 - Prob. 91AECh. 6 - Prob. 92AECh. 6 - Prob. 93AECh. 6 - Prob. 94AECh. 6 - Prob. 95AECh. 6 - Prob. 96AECh. 6 - Prob. 97AECh. 6 - Prob. 98AECh. 6 - Prob. 99AECh. 6 - [6.100] The Chemistry and Life box in Section 6.7...Ch. 6 - Prob. 101AECh. 6 - Using the periodic table as a guide, write the...Ch. 6 - Prob. 103AECh. 6 - [6.104] In the experiment shown schematically...Ch. 6 - Microwave ovens use microwave radiation to heat...Ch. 6 - Prob. 106IECh. 6 - The discovery of hafnium, element number 72,...Ch. 6 - Account for formation of the following series of...Ch. 6 - Prob. 109IECh. 6 - The two most common isotopes of uranium are 235U...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Q1. (a) Draw equations for homolytic and heterolytic cleavages of the N-H bond in NH3. Use curved arrows to show the electron movement. (b) Draw equations for homolytic and heterolytic cleavages of the N-H bond in NH4*. Use curved arrows to show the electron movement.arrow_forwardIndicate which of the following is not an element in its standard state at 25oC and 1 atm. Group of answer choices O2(g) H2(g) Ne(g) N(g) C(s, graphite)arrow_forward6. Show how you would accomplish the following transformations. (Show the steps and reagents/solvents needed) 2-methylpropene →2,2-dimethyloxiran Iarrow_forward
- 4) Answer the following exercise with curved arrows indicating who is a nucleophile or Who is the electrophile? 2.44 Predict the structure of the product formed in the reaction of the organic base pyridine with the organic acid acetic acid, and use curved arrows to indicate the direction of electron flow. 7 H3C OH N Pyridine Acetic acidarrow_forwardUsing the data provided please help me answer this question. Determine the concentration of the iron(Ill) salicylate in the unknown directly from to graph and from the best fit trend-line (least squares analysis) of the graph that yielded a straight line.arrow_forwardPlease help me figure out what the slope is and how to calculate the half life Using the data provided.arrow_forward
- Curved arrows are used to illustrate the flow of electrons. Follow the curved arrows and draw the structure of the missing reactants, intermediates, or products in the following mechanism. Include all lone pairs. Ignore stereochemistry. Ignore inorganic byproducts. H Br2 (1 equiv) H- Select to Draw Starting Alkene Draw Major Product I I H2O 四: ⑦.. Q Draw Major Charged Intermediate Iarrow_forwardNH (aq)+CNO (aq) → CO(NH2)2(s) Experiment [NH4] (M) [CNO] (M) Initial rate (M/s) 1 0.014 0.02 0.002 23 0.028 0.02 0.008 0.014 0.01 0.001 Calculate the rate contant for this reaction using the data provided in the table.arrow_forward2CIO2 + 20H-1 CIO31 + CIO2 + H2O Experiment [CIO2], M [OH-1], M 1 0.0500 0.100 23 2 0.100 0.100 3 0.100 0.0500 Initial Rate, M/s 0.0575 0.230 0.115 ... Given this date, calculate the overall order of this reaction.arrow_forward
- 2 3 .(be)_[Ɔ+(be)_OI ← (b²)_IƆO+ (be)_I Experiment [1-] M 0.005 [OCI-] 0.005 Initial Rate M/min 0.000275 0.0025 0.005 0.000138 0.0025 0.0025 0.000069 4 0.0025 0.0025 0.000140 Calculate the rate constant of this reaction using the table data.arrow_forward1 2 3 4 I(aq) +OCl(aq) → IO¯¯(aq) + Cl¯(aq) Experiment [I-] M 0.005 [OCI-] 0.005 Initial Rate M/min 0.000275 0.0025 0.005 0.000138 0.0025 0.0025 Calculate the overall order of this reaction using the table data. 0.0025 0.000069 0.0025 0.000140arrow_forwardH2O2(aq) +3 I¯(aq) +2 H+(aq) → 13(aq) +2 H₂O(l)· ••• Experiment [H2 O2]o (M) [I]o (M) [H+]。 (M) Initial rate (M/s) 1 0.15 0.15 0.05 0.00012 234 0.15 0.3 0.05 0.00024 0.3 0.15 0.05 0.00024 0.15 0.15 0.1 0.00048 Calculate the overall order of this reaction using the table data.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning
Quantum Numbers, Atomic Orbitals, and Electron Configurations; Author: Professor Dave Explains;https://www.youtube.com/watch?v=Aoi4j8es4gQ;License: Standard YouTube License, CC-BY
QUANTUM MECHANICAL MODEL/Atomic Structure-21E; Author: H to O Chemistry;https://www.youtube.com/watch?v=mYHNUy5hPQE;License: Standard YouTube License, CC-BY