
(a)
Interpretation:
Drawing the molecular-level pictures of strong electrolytes when its breaks up into component ions upon dissolving in water.
Concept Introduction:
Strong electrolyte totally dissociates in a solution. These ions are good conductors of emotional current in the solution.
(a)
Answer to Problem 25E
The dissociation of the given strong electrolyte is,
Explanation of Solution

Figure 1
Let us consider the above molecular-level pictures of
number of
(b)
Interpretation:
Drawing the molecular-level pictures of strong electrolytes when its breaks up into component ions upon dissolving in water.
Concept Introduction:
Strong electrolyte totally dissociates in a solution. These ions are good conductors of emotional current in the solution.
(b)
Answer to Problem 25E
The dissociation of the given strong electrolyte is,.
Explanation of Solution
To draw the molecular-level pictures of
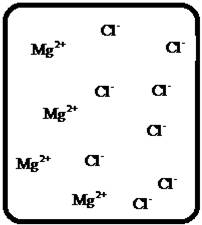
Figure 2
Let us consider the above molecular-level pictures of
number of
(c)
Interpretation:
Drawing the molecular-level pictures of strong electrolytes when its breaks up into component ions upon dissolving in water.
Concept Introduction:
Strong electrolyte totally dissociates in a solution. These ions are good conductors of emotional current in the solution.
(c)
Answer to Problem 25E
The dissociation of the given strong electrolyte is,
Explanation of Solution
To draw the molecular-level pictures of

Figure 3
Let us consider the above molecular-level pictures of
number of
should show equal
number of
(d)
Interpretation:
Drawing the molecular-level pictures of strong electrolytes when its breaks up into component ions upon dissolving in water.
Concept Introduction:
Strong electrolyte totally dissociates in a solution. These ions are good conductors of emotional current in the solution.
(d)
Answer to Problem 25E
The dissociation of the given strong electrolyte is,
Explanation of Solution
To draw the molecular-level pictures of
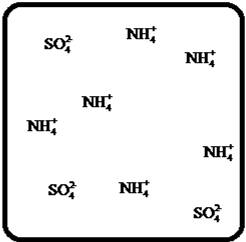
Figure 4
Let us consider the above molecular-level pictures of
number of
(e)
Interpretation:
Drawing the molecular-level pictures of strong electrolytes when its breaks up into component ions upon dissolving in water.
Concept Introduction:
Strong electrolyte totally dissociates in a solution. These ions are good conductors of emotional current in the solution.
(e)
Answer to Problem 25E
The dissociation of the given strong electrolyte is,
Explanation of Solution
To draw the molecular-level pictures of
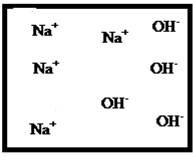
Figure 5
Let us consider the above molecular-level pictures of
Number of
(f)
Interpretation:
Drawing the molecular-level pictures of strong electrolytes when its breaks up into component ions upon dissolving in water.
Concept Introduction:
Strong electrolyte totally dissociates in a solution. These ions are good conductors of emotional current in the solution.
(f)
Answer to Problem 25E
The dissociation of the given strong electrolyte is,
Explanation of Solution
To draw the molecular-level pictures of
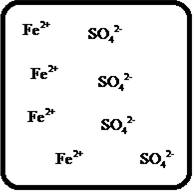
Figure 6
Let us consider the above molecular-level pictures of
number of
(g)
Interpretation:
Drawing the molecular-level pictures of strong electrolytes when its breaks up into component ions upon dissolving in water.
Concept Introduction:
Strong electrolyte totally dissociates in a solution. These ions are good conductors of emotional current in the solution.
(g)
Answer to Problem 25E
The dissociation of the given strong electrolyte is,
Explanation of Solution
To draw the molecular-level pictures of
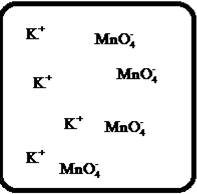
Figure 7
Let us consider the above molecular-level pictures of
number of
(h)
Interpretation:
Drawing the molecular-level pictures of strong electrolytes when its breaks up into component ions upon dissolving in water.
Concept Introduction:
Strong electrolyte totally dissociates in a solution. These ions are good conductors of emotional current in the solution.
(h)
Answer to Problem 25E
The dissociation of the given strong electrolyte is,
Explanation of Solution
To draw the molecular-level pictures of
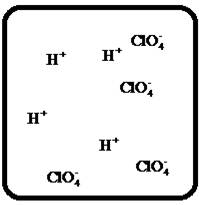
Figure 8
Let us consider the above molecular-level pictures of
number of
(i)
Interpretation:
Drawing the molecular-level pictures of strong electrolytes when its breaks up into component ions upon dissolving in water.
Concept Introduction:
Strong electrolyte totally dissociates in a solution. These ions are good conductors of emotional current in the solution.
(i)
Answer to Problem 25E
The dissociation of the given strong electrolyte is,
Explanation of Solution
To draw the molecular-level pictures of
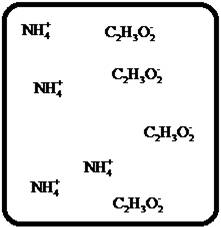
Figure 9
Let us consider the above molecular-level pictures of
Number of
Want to see more full solutions like this?
Chapter 6 Solutions
EBK CHEMISTRY: AN ATOMS FIRST APPROACH
- Provide the drawing of the unknown structure that corresponds with this data.arrow_forward20.44 The Diels-Alder reaction is not limited to making six-membered rings with only car- bon atoms. Predict the products of the following reactions that produce rings with atoms other than carbon in them. OCCH OCCH H (b) CH C(CH₂)s COOCH མ་ནས་བ (c) N=C H -0.X- (e) H C=N COOCHS + CH2=CHCH₂ →→arrow_forwardGiven the attached data, provide the drawing for the corresponding structure.arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





