
(a)
Interpretation: The solubility of
Concept Introduction:A solution is a mixture of solute and solvent. The solubility of solute in solvent depends on the interaction between solute and solvent. It also depends on the polarity of molecules also. As the polar and ionic compounds are soluble in polar solvents whereas the non-polar compounds are soluble in non-polar solvents such as organic solvents. Therefore, it is said to be “Like dissolves Like”.
(a)
Answer to Problem 6E
Explanation of Solution
(b)
Interpretation: The solubility of naphthalene in organic solvent or water needs to be explained.
Concept Introduction:A solution is a mixture of solute and solvent. The solubility of solute in solvent depends on the interaction between solute and solvent. It also depends on the polarity of molecules also. As the polar and ionic compounds are soluble in polar solvents whereas the non-polar compounds are soluble in non-polar solvents such as organic solvents. Therefore, it is said to be “Like dissolves Like”.
(b)
Answer to Problem 6E
Naphthalene is soluble in organic solvent as it is a non-polar compound.
Explanation of Solution
Naphthalene is an aromatic hydrocarbon which is composed of C and H atoms. The structural formula of naphthalene can be shown as below:
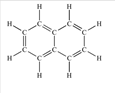
Since there is no other
(c)
Interpretation: The solubility of Anthracenein organic solvent or water needs to be explained.
Concept Introduction:A solution is a mixture of solute and solvent. The solubility of solute in solvent depends on the interaction between solute and solvent. It also depends on the polarity of molecules also. As the polar and ionic compounds are soluble in polar solvents whereas the non-polar compounds are soluble in non-polar solvents such as organic solvents. Therefore, it is said to be “Like dissolves Like”.
(c)
Answer to Problem 6E
Anthracene is soluble in organic solvent as it is a non-polar compound.
Explanation of Solution
Anthracene is an aromatic hydrocarbon which is composed of C and H atoms. The structural formula of Anthracene can be shown as below:
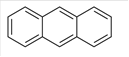
Since there is no other functional group in the molecule therefore it is a non-polar molecule and is soluble in a non-polar solvent.
(d)
Interpretation: The solubility of phenolin organic solvent or water needs to be explained.
Concept Introduction:A solution is a mixture of solute and solvent. The solubility of solute in solvent depends on the interaction between solute and solvent. It also depends on the polarity of molecules also. The polar and ionic compounds are soluble in polar solvents, whereas the non-polar compounds are soluble in non-polar solvents such as organic solvents. Therefore, it is said to be “Like dissolves Like”.
(d)
Answer to Problem 6E
Phenol is soluble water as it is a polar compound.
Explanation of Solution
Phenol is an aromatic hydrocarbon which is composed of C and H atoms with −OH group. The structural formula of phenol can be shown as below:
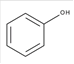
The −OH group can form hydrogen bond with water molecule therefore it is soluble in water.
(e)
Interpretation: The solubility of
Concept Introduction:A solution is a mixture of solute and solvent. The solubility of solute in solvent depends on the interaction between solute and solvent. It also depends on the polarity of molecules also. The polar and ionic compounds are soluble in polar solvents, whereas, the non-polar compounds are soluble in non-polar solvents such as organic solvents. Therefore, it is said to be “Like dissolves Like”.
(e)
Answer to Problem 6E
Explanation of Solution
can be shown as below:
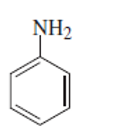
(f)
Interpretation: The solubility of
Concept Introduction:A solution is a mixture of solute and solvent. The solubility of solute in solvent depends on the interaction between solute and solvent. It also depends on the polarity of molecules also. The polar and ionic compounds are soluble in polar solvents, whereas, the non-polar compounds are soluble in non-polar solvents such as organic solvents. Therefore, it is said to be “Like dissolves Like”.
(f)
Answer to Problem 6E
Explanation of Solution
(h)
Interpretation: The solubility of
Concept Introduction:A solution is a mixture of solute and solvent. The solubility of solute in solvent depends on the interaction between solute and solvent. It also depends on the polarity of molecules also. The polar and ionic compounds are soluble in polar solvents, whereas, the non-polar compounds are soluble in non-polar solvents such as organic solvents. Therefore, it is said to be “Like dissolves Like”.
(h)
Answer to Problem 6E
Explanation of Solution
Want to see more full solutions like this?
Chapter 5 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- Please help me solve these problems. Thank you in advance.arrow_forwardPredict the products of this organic reaction: O N IN A N + H2O + HCI ? Specifically, in the drawing area below draw the skeletal ("line") structure of the product, or products, of this reaction. If there's more than one product, draw them in any arrangement you like, so long as they aren't touching. If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. 田 C + Explanation Check Click and drag to start drawing a structure. C © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Centerarrow_forward6. For each of the following, fill in the synthesis arrows with reagents and show the intermediates. You DO NOT need to use the same number of arrows that are shown (you may use more or less), but the product must be formed from the reactant. Then write the mechanism of one step in the synthesis (you can choose which step to write the mechanism for), including all reagents required, clearly labeling the nucleophile and electrophile for each step, and using curved arrows to show the steps in the mechanism. a. b. OHarrow_forward
- Draw the productsarrow_forwardDraw the correct productsarrow_forwardE Organic Chemistry Maxwell Draw the correct products, in either order, for the ozonolysis reaction: 1) O3, CH2Cl2, -78 °C Product 1 + Product 2 2) Zn, HOAc Draw product 1. Select Draw Templates More C H O presented by M Draw product 2. Erase Select Draw Templates M / # # carrow_forward
- ✓ edict the products of this organic reaction: ---- ။ A CH3–C−NH–CH2–C−CH3 + KOH ? Specifically, in the drawing area below draw the condensed structure of the product, or products, of this reaction. If there's more than one product, draw them in any arrangement you like, so long as they aren't touching. If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. Explanation Check Click anywhere to draw the first atom of your structure. C 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibiliarrow_forwardPredict the product of this organic reaction: A HO-C-CH3 + CH3NH2 P+ H2O Specifically, in the drawing area below draw the condensed structure of P. If there is no reasonable possibility for P, check the No answer box under the drawing area. Explanation Check Click anywhere to draw the first atom of your structure. marrow_forwardH 1) OsO4, pyridine 2) Na2SO3 or NaHSO3 in H₂O 2 productsarrow_forward
- ● Biological Macromolecules Naming and drawing cyclic monosaccharides Your answer is incorrect. • Row 1: Your answer is incorrect. Row 3: Your answer is incorrect. • Row 4: Your answer is incorrect. Try again... 0/5 Give the complete common name, including anomer and stereochemistry labels, of the following molecules. You will find helpful information in the ALEKS resource. CH2OH OH OH H H I H OH OH H] H CH2OH H OH ẞ-L-sorbose HOCH2 OH OH H HOCH2 H OH OH H OH H H CH2OH OH H H OH H I- H OH H OH Explanation Recheck W E R % 25 α B Y X & 5 D F G H McGraw Hill LLC. All Rights Reserved. Terms of Use | Pr Parrow_forwardWhat is the missing reactant in this organic reaction? + R -A HO IN + H₂O Specifically, in the drawing area below draw the skeletal ("line") structure of R. If there is more than one reasonable answer, you can draw any one of them. If there is no reasonable answer, check the No answer box under the drawing area. Note for advanced students: you may assume no products other than those shown above are formed. Explanation Check Click and drag to start drawing a structure. © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Centerarrow_forwardStuc X ctclix ALE X A ALE אן A ALEX Lab (195 X Nut x M Inb x NU X NUT X Unt x + → C www-awu.aleks.com/alekscgi/x/Isl.exe/10_u-lgNslkr7j8P3jH-IQ1g8NUi-mObKa_ZLx2twjEhK7mVG6PulJI006NcKTV37JxMpZuyrVCdQolLAKqp_7U3r1GUD3... New Chrome available: Naomi Question 26 of 39 (4 points) | Question Attempt: 1 of Unlimited Give the IUPAC name. 2,3-dimethylhexane Part: 1/2 Part 2 of 2 Draw the skeletal structure of a constitutional isomer of the alkane above that contains a different number of carbons in its longest chain. Skip Part Check Click and drag to start drawing a structure. 3 Finance headline Q Search mwa Harvard Intensifi... X Save For Later 00 dlo HB Submit Assignment 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility a 9:11 PM 4/22/2025arrow_forward
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning



