
(a)
Interpretation: The electron sublevel starts to fill after the completion of 5s sublevel should be determined.
Concept Introduction:
The rules for the allowed quantum numbers combinations are as follows:
- All the three quantum numbers ( n, l and m ) describes the orbital of an atom and they are integers.
- The principal quantum number, n value cannot be zero. Thus, the values allowed for the principal quantum number are 1, 2, 3, 4, and so on.
- The value of angular quantum number, l can be between 0 to n-1. Thus, if value of n is equal to 3 the value of l can be 0, 1 or 2.
- The value of magnetic quantum number, m can be between − l to +l . Thus, if value of l is equal to 2, m can be wither -2, -1, 0, +1, or +2
For same number of principal quantum number, an orbital form a shell. The first character denotes the shell and the second identifies the sub-shell.
Here, for s orbital value of
The relative energy of orbitals is represented as follows:
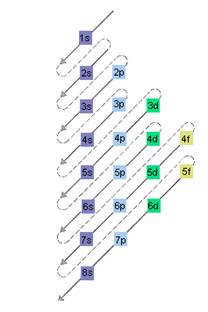
(b)
Interpretation: The electron sublevel starts to fill after the completion of 4d sublevel should be determined.
Concept Introduction:
The rules for the allowed quantum numbers combinations are as follows:
- All the three quantum numbers ( n, l and m ) describes the orbital of an atom and they are integers.
- The principal quantum number, n value cannot be zero. Thus, the values allowed for the principal quantum number are 1, 2, 3, 4, and so on.
- The value of angular quantum number, l can be between 0 to n-1. Thus, if value of n is equal to 3 the value of l can be 0, 1 or 2.
- The value of magnetic quantum number, m can be between − l to +l . Thus, if value of l is equal to 2, m can be wither -2, -1, 0, +1, or +2
For same number of principal quantum number, an orbital form a shell. The first character denotes the shell and the second identifies the sub-shell.
Here, for s orbital value of
The relative energy of orbitals is represented as follows:
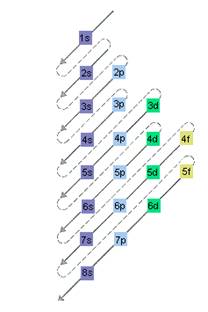
(c)
Interpretation: The electron sublevel starts to fill after the completion of 4f sublevel should be determined.
Concept Introduction:
The rules for the allowed quantum numbers combinations are as follows:
- All the three quantum numbers ( n, l and m ) describes the orbital of an atom and they are integers.
- The principal quantum number, n value cannot be zero. Thus, the values allowed for the principal quantum number are 1, 2, 3, 4, and so on.
- The value of angular quantum number, l can be between 0 to n-1. Thus, if value of n is equal to 3 the value of l can be 0, 1 or 2.
- The value of magnetic quantum number, m can be between − l to +l . Thus, if value of l is equal to 2, m can be wither -2, -1, 0, +1, or +2
For same number of principal quantum number, an orbital form a shell. The first character denotes the shell and the second identifies the sub-shell.
Here, for s orbital value of
The relative energy of orbitals is represented as follows:
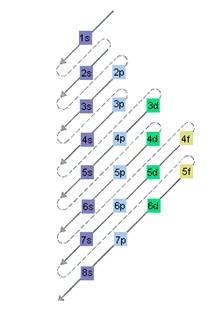
(d)
Interpretation: The electron sublevel starts to fill after the completion of 5p sublevel should be determined.
Concept Introduction:
The rules for the allowed quantum numbers combinations are as follows:
- All the three quantum numbers ( n, l and m ) describes the orbital of an atom and they are integers.
- The principal quantum number, n value cannot be zero. Thus, the values allowed for the principal quantum number are 1, 2, 3, 4, and so on.
- The value of angular quantum number, l can be between 0 to n-1. Thus, if value of n is equal to 3 the value of l can be 0, 1 or 2.
- The value of magnetic quantum number, m can be between − l to +l . Thus, if value of l is equal to 2, m can be wither -2, -1, 0, +1, or +2
lFor same number of principal quantum number, an orbital form a shell. The first character denotes the shell and the second identifies the sub-shell.
Here, for s orbital value of
The relative energy of orbitals is represented as follows:
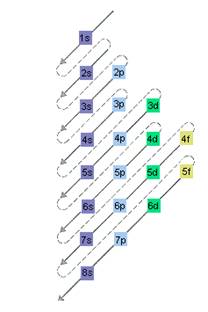
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
EP BASIC CHEMISTRY-MODIFIED MASTERING
- Can you please help mne with this problem. Im a visual person, so can you redraw it, potentislly color code and then as well explain it. I know im given CO2 use that to explain to me, as well as maybe give me a second example just to clarify even more with drawings (visuals) and explanations.arrow_forwardPart 1. Aqueous 0.010M AgNO 3 is slowly added to a 50-ml solution containing both carbonate [co32-] = 0.105 M and sulfate [soy] = 0.164 M anions. Given the ksp of Ag2CO3 and Ag₂ soy below. Answer the ff: Ag₂ CO3 = 2 Ag+ caq) + co} (aq) ksp = 8.10 × 10-12 Ag₂SO4 = 2Ag+(aq) + soy² (aq) ksp = 1.20 × 10-5 a) which salt will precipitate first? (b) What % of the first anion precipitated will remain in the solution. by the time the second anion starts to precipitate? (c) What is the effect of low pH (more acidic) condition on the separate of the carbonate and sulfate anions via silver precipitation? What is the effect of high pH (more basic)? Provide appropriate explanation per answerarrow_forwardPart 4. Butanoic acid (ka= 1.52× 10-5) has a partition coefficient of 3.0 (favors benzene) when distributed bet. water and benzene. What is the formal concentration of butanoic acid in each phase when 0.10M aqueous butanoic acid is extracted w❘ 25 mL of benzene 100 mL of a) at pit 5.00 b) at pH 9.00arrow_forward
- Calculate activation energy (Ea) from the following kinetic data: Temp (oC) Time (s) 23.0 180. 32.1 131 40.0 101 51.8 86.0 Group of answer choices 0.0269 kJ/mole 2610 kJ/mole 27.6 kJ/mole 0.215 kJ/mole 20.8 kJ/molearrow_forwardCalculate activation energy (Ea) from the following kinetic data: Temp (oC) Time (s) 23.0 180. 32.1 131 40.0 101 51.8 86.0 choices: 0.0269 kJ/mole 2610 kJ/mole 27.6 kJ/mole 0.215 kJ/mole 20.8 kJ/molearrow_forwardCalculate activation energy (Ea) from the following kinetic data: Temp (oC) Time (s) 23.0 180. 32.1 131 40.0 101 51.8 86.0arrow_forward
- Please solvearrow_forwardRank the compounds in each group below according to their reactivity toward electrophilic aromatic substitution (most reactive = 1; least reactive = 3). Place the number corresponding to the compounds' relative reactivity in the blank below the compound. a. CH₂F CH3 F b. At what position, and on what ring, is bromination of phenyl benzoate expected to occur? Explain your answer. :0: C-O phenyl benzoate 6.Consider the reaction below to answer the following questions. A B C NO₂ FeBr3 + Br₂ D a. The nucleophile in the reaction is: BODADES b. The Lewis acid catalyst in the reaction is: C. This reaction proceeds d. Draw the structure of product D. (faster or slower) than benzene.arrow_forwardPart 2. A solution of 6.00g of substance B in 100.0mL of aqueous solution is in equilibrium, at room temperature, wl a solution of B in diethyl ether (ethoxyethane) containing 25.0 g of B in 50.0 mL 9) what is the distribution coefficient of substance B b) what is the mass of B extracted by shaking 200 ml of an aqueous solution containing 10g of B with call at room temp): i) 100 mL of diethyl ether ii) 50ml of diethyl ether twice iii) 25ml of diethyl ether four timesarrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning

