
Chemistry: An Atoms First Approach
2nd Edition
ISBN: 9781305079243
Author: Steven S. Zumdahl, Susan A. Zumdahl
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 81E
Give the empirical formula for each of the compounds represented below.
a. 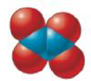
b. 
c. 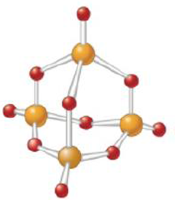
d. 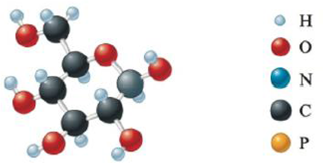
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Nail polish remover containing acetone was spilled in a room 5.23 m × 3.28 m × 2.76 m.
Measurements indicated that 2,250 mg of acetone evaporated. Calculate the acetone concentration in micrograms per cubic meter.
Please help me answer number 1. 1. If your graphs revealed a mathematical relationship between specific heat and atomic mass, write down an equation for the relationship.
I also don't understand, is the equation from the line regression the one that I'm suppose use to show the relationship? If so could you work it all the way out?
Describe the principle of resonance and give a set of Lewis Structures to illustrate your explanation.
Chapter 5 Solutions
Chemistry: An Atoms First Approach
Ch. 5 - Prob. 1RQCh. 5 - Atomic masses are relative masses. What does this...Ch. 5 - The atomic mass of boron (B) is given in the...Ch. 5 - What three conversion factors and in what order...Ch. 5 - Fig. 5-5 illustrates a schematic diagram of a...Ch. 5 - What is the difference between the empirical and...Ch. 5 - Consider the hypothetical reaction between A2 and...Ch. 5 - Prob. 8RQCh. 5 - Consider the following mixture of SO2(g) and...Ch. 5 - Why is the actual yield of a reaction often less...
Ch. 5 - The following are actual student responses to the...Ch. 5 - What information do we get from a chemical...Ch. 5 - You are making cookies and are missing a key...Ch. 5 - Nitrogen gas (N2) and hydrogen gas (H2) react to...Ch. 5 - For the preceding question, which of the following...Ch. 5 - Prob. 6ALQCh. 5 - Prob. 7ALQCh. 5 - Consider an iron bar on a balance as shown. As the...Ch. 5 - You may have noticed that water sometimes drips...Ch. 5 - Prob. 10ALQCh. 5 - What is true about the chemical properties of the...Ch. 5 - Is there a difference between a homogeneous...Ch. 5 - Prob. 13ALQCh. 5 - The average mass of a carbon atom is 12.011....Ch. 5 - Can the subscripts in a chemical formula be...Ch. 5 - Consider the equation 2A + B . A2B. If you mix 1.0...Ch. 5 - According to the law of conservation of mass, mass...Ch. 5 - Which of the following pairs of compounds have the...Ch. 5 - Atoms of three different elements are represented...Ch. 5 - In chemistry, what is meant by the term mole? What...Ch. 5 - Which (if any) of the following is (are) true...Ch. 5 - Consider the equation 3A + B C + D. You react 4...Ch. 5 - Reference Section 5-2 to find the atomic masses of...Ch. 5 - Avogadros number, molar mass, and the chemical...Ch. 5 - If you had a mole of U.S. dollar bills and equally...Ch. 5 - Prob. 26QCh. 5 - Which of the following compounds have the same...Ch. 5 - Prob. 28QCh. 5 - How is the mass percent of elements in a compound...Ch. 5 - A balanced chemical equation contains a large...Ch. 5 - Prob. 31QCh. 5 - Hydrogen gas and oxygen gas react to form water,...Ch. 5 - What is the theoretical yield for a reaction, and...Ch. 5 - What does it mean to say a reactant is present in...Ch. 5 - Consider the following generic reaction: A2B2 + 2C...Ch. 5 - Consider the following generic reaction:...Ch. 5 - An element consists of 1.40% of an isotope with...Ch. 5 - An element X bas five major isotopes, which are...Ch. 5 - The element rhenium (Re) bas two naturally...Ch. 5 - Assume silicon has three major isotopes in nature...Ch. 5 - The element europium exists in nature as two...Ch. 5 - The element silver (Ag) has two naturally...Ch. 5 - The mass spectrum of bromine (Br2) consists of...Ch. 5 - The stable isotopes of iron arc 54Fe, 56Fe, 57Fe,...Ch. 5 - Calculate the mass of 500. atoms of iron (Fe).Ch. 5 - What number of Fe atoms and what amount (moles) of...Ch. 5 - Diamond is a natural form of pure carbon. What...Ch. 5 - A diamond contains 5.0 1021 atoms of carbon. What...Ch. 5 - Aluminum metal is produced by passing an electric...Ch. 5 - The Freons are a class of compounds containing...Ch. 5 - Calculate the molar mass of the following...Ch. 5 - Calculate the molar mass of the following...Ch. 5 - What amount (moles) of compound is present in 1.00...Ch. 5 - What amount (moles) of compound is present in 1.00...Ch. 5 - What mass of compound is present in 5.00 moles of...Ch. 5 - What mass of compound is present in 5.00 moles of...Ch. 5 - Prob. 57ECh. 5 - Prob. 58ECh. 5 - Prob. 59ECh. 5 - What number of molecules (or formula units) are...Ch. 5 - What number of atoms of nitrogen are present in...Ch. 5 - Prob. 62ECh. 5 - Freon- 12 (CCI2F2) is used as a refrigerant in air...Ch. 5 - Bauxite, the principal ore used in the production...Ch. 5 - What amount (moles) is represented by each of...Ch. 5 - What amount (moles) is represented by each of...Ch. 5 - What number of atoms of nitrogen are present in...Ch. 5 - Complete the following table.Ch. 5 - Ascorbic acid, or vitamin C (C6H8O6), is an...Ch. 5 - The molecular formula of acetylsalicylic acid...Ch. 5 - Chloral hydrate (C2H3Cl3O2) is a drug formerly...Ch. 5 - Dimethylnitrosamine, (CH3)2N2O , is a carcinogenic...Ch. 5 - Calculate the percent composition by mass of the...Ch. 5 - In 1987 the first substance to act as a...Ch. 5 - Prob. 75ECh. 5 - Arrange the following substances in order of...Ch. 5 - Fungal laccase, a blue protein found in...Ch. 5 - Hemoglobin is the protein that transports oxygen...Ch. 5 - Express the composition of each of the following...Ch. 5 - Considering your answer to Exercise 79, which type...Ch. 5 - Give the empirical formula for each of the...Ch. 5 - Determine the molecular formulas to which the...Ch. 5 - A compound that contains only carbon, hydrogen,...Ch. 5 - The most common form of nylon (nylon-6) is 63.68%...Ch. 5 - There are two binary compounds of mercury and...Ch. 5 - A sample of urea contains 1.121 g N, 0.161 g H,...Ch. 5 - Prob. 87ECh. 5 - Determine the molecular formula of a compound that...Ch. 5 - A compound contains 47.08% carbon, 6.59% hydrogen,...Ch. 5 - Maleic acid is an organic compound composed of...Ch. 5 - One of the components that make up common table...Ch. 5 - A compound contains only C, H, and N. Combustion...Ch. 5 - Prob. 93ECh. 5 - A compound contains only carbon, hydrogen, and...Ch. 5 - Give the balanced equation for each of the...Ch. 5 - Give the balanced equation for each of the...Ch. 5 - Prob. 97ECh. 5 - Iron oxide ores, commonly a mixture of FeO and...Ch. 5 - Balance the following equations: a. Ca(OH)2(aq) +...Ch. 5 - Balance each of the following chemical equations....Ch. 5 - Prob. 101ECh. 5 - Balance the following equations: a. Cr(s) + S8(s) ...Ch. 5 - Silicon is produced for the chemical and...Ch. 5 - Prob. 104ECh. 5 - Over the years, the thermite reaction has been...Ch. 5 - The reaction between potassium chlorate and red...Ch. 5 - The reusable booster rockets of the U.S. space...Ch. 5 - One of relatively few reactions that takes place...Ch. 5 - Elixirs such as Atka-Seltzer use the reaction of...Ch. 5 - Aspirin (C9H8O4) is synthesized by reacting...Ch. 5 - Bacterial digestion is an economical method of...Ch. 5 - Phosphorus can be prepared from calcium phosphate...Ch. 5 - Coke is an impure form of carbon that is often...Ch. 5 - The space shuttle environmental control system...Ch. 5 - Consider the reaction between NO(g) and O2(g)...Ch. 5 - Consider the following reaction:...Ch. 5 - Prob. 117ECh. 5 - Consider the following unbalanced equation:...Ch. 5 - Hydrogen peroxide is used as a cleansing agent in...Ch. 5 - Silver sulfadiazine bum-treating cream creates a...Ch. 5 - Hydrogen cyanide is produced industrially from the...Ch. 5 - Acrylonitrile C3H3N) is the starting material for...Ch. 5 - Prob. 123ECh. 5 - DDT, an insecticide harmful to fish, birds, and...Ch. 5 - Bornite (Cu3FeS3) is a copper ore used in the...Ch. 5 - Consider the following unbalanced reaction:...Ch. 5 - In using a mass spectrometer, a chemist sees a...Ch. 5 - Boron consists of two isotopes, 10B and 11B....Ch. 5 - A given sample of a xenon fluoride compound...Ch. 5 - Aspartame is an artificial sweetener that is 160...Ch. 5 - Anabolic steroids are performance enhancement...Ch. 5 - Many cereals are made with high moisture content...Ch. 5 - The compound adrenaline contains 56.79% C, 6.56%...Ch. 5 - Adipic acid is an organic compound composed of...Ch. 5 - Prob. 135AECh. 5 - Some bismuth tablets, a medication used to treat...Ch. 5 - The empirical formula of styrene is CH; the molar...Ch. 5 - Terephthalic acid is an important chemical used in...Ch. 5 - A sample of a hydrocarbon (a compound consisting...Ch. 5 - A binary compound between an unknown element E and...Ch. 5 - A 0.755-g sample of hydrated copper(II) sulfate...Ch. 5 - ABS plastic is a tough, hard plastic used in...Ch. 5 - Prob. 143AECh. 5 - Methane (CH4) is the main component of marsh gas....Ch. 5 - A potential fuel for rockets is a combination of...Ch. 5 - A 0.4230-g sample of impure sodium nitrate was...Ch. 5 - Prob. 147AECh. 5 - Commercial brass, an alloy of Zn and Cu, reacts...Ch. 5 - Prob. 149AECh. 5 - You have seven closed containers, each with equal...Ch. 5 - A substance X2Z has the composition (by mass) of...Ch. 5 - Consider samples of phosphine (PH3), water (H2O),...Ch. 5 - Calculate the number of moles for each compound in...Ch. 5 - Arrange the following substances in order of...Ch. 5 - Para-cresol, a substance used as a disinfectant...Ch. 5 - A compound with molar mass 180.1 g/mol has the...Ch. 5 - Prob. 157CWPCh. 5 - Consider the following unbalanced chemical...Ch. 5 - Sulfur dioxide gas reacts with sodium hydroxide to...Ch. 5 - Gallium arsenide, GaAs, has gained widespread use...Ch. 5 - Consider the following data for three binary...Ch. 5 - Natural rubidium has the average mass of 85.4678 u...Ch. 5 - A compound contains only carbon, hydrogen,...Ch. 5 - Nitric acid is produced commercially by the...Ch. 5 - When the supply of oxygen is limited, iron metal...Ch. 5 - A 9.780-g gaseous mixture contains ethane (C2H6)...Ch. 5 - Zinc and magnesium metal each reacts with...Ch. 5 - Prob. 168CPCh. 5 - Consider a gaseous binary compound with a molar...Ch. 5 - Prob. 170CPCh. 5 - Prob. 171CPCh. 5 - The aspirin substitute, acetaminophen (C8H9O2N),...Ch. 5 - An element X forms both a dichloride (XCl2) and a...Ch. 5 - Prob. 174CPCh. 5 - When aluminum metal is heated with an element from...Ch. 5 - Consider a mixture of potassium chloride and...Ch. 5 - Ammonia reacts with O2 to form either NO(g) or...Ch. 5 - You take 1.00 g of an aspirin tablet (a compound...Ch. 5 - With the advent of techniques such as scanning...Ch. 5 - Tetrodotoxin is a toxic chemical found in fugu...Ch. 5 - Prob. 181IPCh. 5 - Prob. 182IPCh. 5 - Prob. 183IPCh. 5 - A 2.077-g sample of an element, which has an...Ch. 5 - Consider the following balanced chemical equation:...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Don't used hand raitingarrow_forwardIt is not unexpected that the methoxyl substituent on a cyclohexane ring prefers to adopt the equatorial conformation. OMe H A G₂ = +0.6 kcal/mol OMe What is unexpected is that the closely related 2-methoxytetrahydropyran prefers the axial conformation: H H OMe OMe A Gp=-0.6 kcal/mol Methoxy: CH3O group Please be specific and clearly write the reason why this is observed. This effect that provides stabilization of the axial OCH 3 group in this molecule is called the anomeric effect. [Recall in the way of example, the staggered conformer of ethane is more stable than eclipsed owing to bonding MO interacting with anti-bonding MO...]arrow_forward206 Pb 82 Express your answers as integers. Enter your answers separated by a comma. ▸ View Available Hint(s) VAΣ ΜΕ ΑΣΦ Np, N₁ = 82,126 Submit Previous Answers ? protons, neutronsarrow_forward
- Please draw the inverted chair forms of the products for the two equilibrium reactions shown below. Circle the equilibrium reaction that would have a AG = 0, i.e., the relative energy of the reactant (to the left of the equilibrium arrows) equals the relative energy of the product? [No requirement to show or do calculations.] CH3 CH3 HH CH3 1 -CH3arrow_forward5. Please consider the Newman projection of tartaric acid drawn below as an eclipsed conformer (1). Please draw the most stable conformer and two intermediate energy conformers noting that staggered conformers are lower in energy than eclipsed forms even if the staggered conformers have gauche relationships between groups. [Draw the substituents H and OH on the front carbons and H, OH and CO₂H on the back carbons based on staggered forms. -CO₂H is larger than -OH.] OH COH ICOOH COOH COOH 1 2 COOH COOH 3 4 Staggered Staggered Staggered (most stable) Indicate the number of each conformer above (1, 2, 3 and 4) that corresponds to the relative energies below. Ref=0 Rotation 6. (60 points) a. Are compounds 1 and 2 below enantiomers, diastereomers or identical? OH OH HO HO LOH HO HO OH 2 OH OH b. Please complete the zig-zag conformation of the compound (3R,4S)-3,4-dichloro-2,5-dimethylhexane by writing the respective atoms in the boxes. 3.arrow_forwardThe plutonium isotope with 144 neutrons Enter the chemical symbol of the isotope.arrow_forward
- The mass ratio of sodium to fluorine in sodium fluoride is 1.21:1. A sample of sodium fluoride produced 26.1 gg of sodium upon decomposition. How much fluorine was formed?arrow_forward32S 16 Enter your answers numerically separated by a comma. Np. Nn = 跖 ΟΙ ΑΣΦ Submit Request Answer ? protons, neutronsarrow_forward2. Which dimethylcyclohexane compounds shown below exhibit symmetry and therefore are not chiral and would not rotate plane polarized light. 1 CH3 CH CH3 CH3 2 3 CH3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning 
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning
Step by Step Stoichiometry Practice Problems | How to Pass ChemistryMole Conversions Made Easy: How to Convert Between Grams and Moles; Author: Ketzbook;https://www.youtube.com/watch?v=b2raanVWU6c;License: Standard YouTube License, CC-BY