
Concept explainers
(a)
Interpretation:
Lewis dot symbol for the given atoms/ions must be shown
Concept Introduction:
Lewis dot symbol is used to represent the valence electrons of an atom or ion using dots surrounding the element symbol along four sides of the element symbol without maintaining exact order for the placement of dots.
In Lewis dot symbol representation, the
The valence electron is the number of electrons present in the outermost shell of the atom. The number of valence electrons will be same for the same group elements which is represented by Lewis dot symbol.
(a)
Answer to Problem 5.7QP
Lewis dot representation for
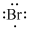
Explanation of Solution
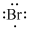
The electronic configuration of Bromine is
(b)
Interpretation:
Lewis dot symbol for the given atoms/ions must be shown
Concept Introduction:
Lewis dot symbol is used to represent the valence electrons of an atom or ion using dots surrounding the element symbol along four sides of the element symbol without maintaining exact order for the placement of dots.
In Lewis dot symbol representation, the symbol of element is surrounded by "dots" indicating the number of valence electrons available for the element. The dots can be placed one at a time on all the four sides, further electron can be placed by pairing up with the first placed dots. According to the number of electrons added or removed, charge must be placed on the Lewis dot symbol for cations and anions.
The valence electron is the number of electrons present in the outermost shell of the atom. The number of valence electrons will be same for the same group elements which is represented by Lewis dot symbol.
(b)
Answer to Problem 5.7QP
Lewis dot representation for
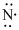
Explanation of Solution
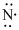
The electronic configuration of Nitrogen is
Lewis dot symbol denotes the valence electron of an atom. The valence electron of
(c)
Interpretation:
Lewis dot symbol for the given atoms/ions must be shown
Concept Introduction:
Lewis dot symbol is used to represent the valence electrons of an atom or ion using dots surrounding the element symbol along four sides of the element symbol without maintaining exact order for the placement of dots.
In Lewis dot symbol representation, the symbol of element is surrounded by "dots" indicating the number of valence electrons available for the element. The dots can be placed one at a time on all the four sides, further electron can be placed by pairing up with the first placed dots. According to the number of electrons added or removed, charge must be placed on the Lewis dot symbol for cations and anions.
The valence electron is the number of electrons present in the outermost shell of the atom. The number of valence electrons will be same for the same group elements which is represented by Lewis dot symbol.
(c)
Answer to Problem 5.7QP
Lewis dot representation for
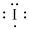
Explanation of Solution
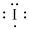
The electronic configuration of Iodine is
(d)
Interpretation:
Lewis dot symbol for the given atoms/ions must be shown
Concept Introduction:
Lewis dot symbol is used to represent the valence electrons of an atom or ion using dots surrounding the element symbol along four sides of the element symbol without maintaining exact order for the placement of dots.
In Lewis dot symbol representation, the symbol of element is surrounded by "dots" indicating the number of valence electrons available for the element. The dots can be placed one at a time on all the four sides, further electron can be placed by pairing up with the first placed dots. According to the number of electrons added or removed, charge must be placed on the Lewis dot symbol for cations and anions.
The valence electron is the number of electrons present in the outermost shell of the atom. The number of valence electrons will be same for the same group elements which is represented by Lewis dot symbol.
(d)
Answer to Problem 5.7QP
Lewis dot representation for
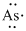
Explanation of Solution
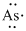
The electronic configuration of Arsenic is
(e)
Interpretation:
Lewis dot symbol for the given atoms/ions must be shown
Concept Introduction:
Lewis dot symbol is used to represent the valence electrons of an atom or ion using dots surrounding the element symbol along four sides of the element symbol without maintaining exact order for the placement of dots.
In Lewis dot symbol representation, the symbol of element is surrounded by "dots" indicating the number of valence electrons available for the element. The dots can be placed one at a time on all the four sides, further electron can be placed by pairing up with the first placed dots. According to the number of electrons added or removed, charge must be placed on the Lewis dot symbol for cations and anions.
The valence electron is the number of electrons present in the outermost shell of the atom. The number of valence electrons will be same for the same group elements which is represented by Lewis dot symbol.
(e)
Answer to Problem 5.7QP
Lewis dot representation for
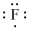
Explanation of Solution
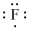
The electronic configuration of Fluorine is
(f)
Interpretation:
Lewis dot symbol for the given atoms/ions must be shown
Concept Introduction:
Lewis dot symbol is used to represent the valence electrons of an atom or ion using dots surrounding the element symbol along four sides of the element symbol without maintaining exact order for the placement of dots.
In Lewis dot symbol representation, the symbol of element is surrounded by "dots" indicating the number of valence electrons available for the element. The dots can be placed one at a time on all the four sides, further electron can be placed by pairing up with the first placed dots. According to the number of electrons added or removed, charge must be placed on the Lewis dot symbol for cations and anions.
The valence electron is the number of electrons present in the outermost shell of the atom. The number of valence electrons will be same for the same group elements which is represented by Lewis dot symbol.
(f)
Answer to Problem 5.7QP
Lewis dot representation for
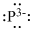
Explanation of Solution
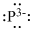
The electronic configuration of Phosphorus is
(g)
Interpretation:
Lewis dot symbol for the given atoms/ions must be shown
Concept Introduction:
Lewis dot symbol is used to represent the valence electrons of an atom or ion using dots surrounding the element symbol along four sides of the element symbol without maintaining exact order for the placement of dots.
In Lewis dot symbol representation, the symbol of element is surrounded by "dots" indicating the number of valence electrons available for the element. The dots can be placed one at a time on all the four sides, further electron can be placed by pairing up with the first placed dots. According to the number of electrons added or removed, charge must be placed on the Lewis dot symbol for cations and anions.
The valence electron is the number of electrons present in the outermost shell of the atom. The number of valence electrons will be same for the same group elements which is represented by Lewis dot symbol.
(g)
Answer to Problem 5.7QP
Lewis dot representation for
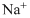
Explanation of Solution
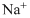
The electronic configuration of Sodium is
(h)
Interpretation:
Lewis dot symbol for the given atoms/ions must be shown
Concept Introduction:
Lewis dot symbol is used to represent the valence electrons of an atom or ion using dots surrounding the element symbol along four sides of the element symbol without maintaining exact order for the placement of dots.
In Lewis dot symbol representation, the symbol of element is surrounded by "dots" indicating the number of valence electrons available for the element. The dots can be placed one at a time on all the four sides, further electron can be placed by pairing up with the first placed dots. According to the number of electrons added or removed, charge must be placed on the Lewis dot symbol for cations and anions.
The valence electron is the number of electrons present in the outermost shell of the atom. The number of valence electrons will be same for the same group elements which is represented by Lewis dot symbol.
(h)
Answer to Problem 5.7QP
Lewis dot representation for
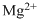
Explanation of Solution
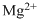
The electronic configuration of Magnesium is
(i)
Interpretation:
Lewis dot symbol for the given atoms/ions must be shown
Concept Introduction:
Lewis dot symbol is used to represent the valence electrons of an atom or ion using dots surrounding the element symbol along four sides of the element symbol without maintaining exact order for the placement of dots.
In Lewis dot symbol representation, the symbol of element is surrounded by "dots" indicating the number of valence electrons available for the element. The dots can be placed one at a time on all the four sides, further electron can be placed by pairing up with the first placed dots. According to the number of electrons added or removed, charge must be placed on the Lewis dot symbol for cations and anions.
The valence electron is the number of electrons present in the outermost shell of the atom. The number of valence electrons will be same for the same group elements which is represented by Lewis dot symbol.
(i)
Answer to Problem 5.7QP
Lewis dot representation for
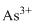
Explanation of Solution
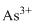
The electronic configuration of Arsenic is
(j)
Interpretation:
Lewis dot symbol for the given atoms/ions must be shown
Concept Introduction:
Lewis dot symbol is used to represent the valence electrons of an atom or ion using dots surrounding the element symbol along four sides of the element symbol without maintaining exact order for the placement of dots.
In Lewis dot symbol representation, the symbol of element is surrounded by "dots" indicating the number of valence electrons available for the element. The dots can be placed one at a time on all the four sides, further electron can be placed by pairing up with the first placed dots. According to the number of electrons added or removed, charge must be placed on the Lewis dot symbol for cations and anions.
The valence electron is the number of electrons present in the outermost shell of the atom. The number of valence electrons will be same for the same group elements which is represented by Lewis dot symbol.
(j)
Answer to Problem 5.7QP
Lewis dot representation for
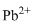
Explanation of Solution
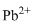
The electronic configuration of Lead is
Want to see more full solutions like this?
Chapter 5 Solutions
CHEMISTRY: ATOMS FIRST VOL 1 W/CONNECT
- A mixture of C7H12O2, C9H9OCl, biphenyl and acetone was put together in a gas chromatography tube. Please decide from the GC resutls which correspond to the peak for C7,C9 and biphenyl and explain the reasoning based on GC results. Eliminate unnecessary peaks from Gas Chromatography results.arrow_forwardIs the molecule chiral, meso, or achiral? CI .CH3 H₂C CIarrow_forwardPLEASE HELP ! URGENT!arrow_forward
- Identify priority of the substituents: CH3arrow_forwardHow many chiral carbons are in the molecule? OH F CI Brarrow_forwardA mixture of three compounds Phen-A, Acet-B and Rin-C was analyzed using TLC with 1:9 ethanol: hexane as the mobile phase. The TLC plate showed three spots of R, 0.1 and 0.2 and 0.3. Which of the three compounds (Phen-A; Acet-B or Rin-C) would have the highest (Blank 1), middle (Blank 2) and lowest (Blank 3) spot respectively? 0 CH: 0 CH, 0 H.C OH H.CN OH Acet-B Rin-C phen-A A A <arrow_forward
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





