
Concept explainers
(a)
Interpretation:
The specific type of relationship between the molecules in the given pair is to be determined.
Concept introduction:
If two molecules have different molecular formulas, they are unrelated. If they have the same molecular formula, they may be the same molecule or isomers. If the two can be interconverted by rotation about a bond, they are conformers. If they have different connectivity of atoms, they are constitutional isomers. If they have the same connectivity, but are not conformers, they must be stereoisomers. Stereoisomers may be enantiomers or diastereomers depending on whether they have nonsuperimposable mirror images or not.
Answer to Problem 5.71P
The two molecules are unrelated.
Explanation of Solution
Structures of the two molecules are:
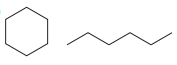
Both molecules contain the same number of carbon atoms. However, their formulas are different. The first molecule contains a ring with all single bonds. Therefore it has an IHD of 1, and its molecular formula must be
Thus, these are different, unrelated molecules.
Two molecules are unrelated if they have different molecular formulas.
(b)
Interpretation:
The specific type of relationship between the molecules in the given pair is to be determined.
Concept introduction:
If two molecules have different molecular formulas, they are unrelated. If they have the same molecular formula, they may be the same molecule or isomers. If the two can be interconverted by rotation about a bond, they are conformers. If they have different connectivity of atoms, they are constitutional isomers. If they have the same connectivity, but are not conformers, they must be stereoisomers. Stereoisomers may be enantiomers or diastereomers depending on whether they have nonsuperimposable mirror images or not.
Answer to Problem 5.71P
The two molecules are constitutional isomers.
Explanation of Solution
The structures of the two molecules are:
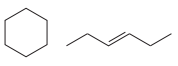
Both molecules contain six carbon atoms. The first one contains a ring, while the second contains one double bond. They both have the same IHD of 1 and the same molecular formula
Therefore, they must be constitutional isomers.
Molecules with the same molecular formula but different connectivities are constitutional isomers.
(c)
Interpretation:
The specific type of relationship between the molecules in the given pair is to be determined.
Concept introduction:
If two molecules have different molecular formulas, they are unrelated. If they have the same molecular formula, they may be the same molecule or isomers. If the two can be interconverted by rotation about a bond, they are conformers. If they have different connectivity of atoms, they are constitutional isomers. If they have the same connectivity, but are not conformers, they must be stereoisomers. Stereoisomers may be enantiomers or diastereomers depending on whether they have nonsuperimpoable mirror images or not.
Answer to Problem 5.71P
The two molecules are unrelated.
Explanation of Solution
The structures of the molecules are:
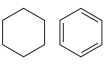
Both have the same number of carbon atoms and also contain the same ring. However, they have different IHDs, and therefore, different molecular formulas. The first molecule contains only one ring, but no multiple bonds. Therefore, it has an IHD of 1 and a molecular formula of
Therefore, they are unrelated molecules.
Two molecules are unrelated if they have different molecular formulas.
(d)
Interpretation:
The specific type of relationship between the molecules in the given pair is to be determined.
Concept introduction:
If two molecules have different molecular formulas, they are unrelated. If they have the same molecular formula, they may be the same molecule or isomers. If the two can be interconverted by rotation about a bond, they are conformers. If they have different connectivity of atoms, they are constitutional isomers. If they have the same connectivity, but are not conformers, they must be stereoisomers. Stereoisomers may be enantiomers or diastereomers depending on whether they have nonsuperimposable mirror images or not.
Answer to Problem 5.71P
The molecules are unrelated.
Explanation of Solution
The structures of the molecules are:
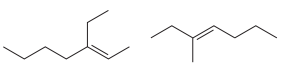
The molecules have different number of carbon atoms, nine and eight, respectively.
Therefore, they are unrelated molecules.
Molecules with different molecular formulas are unrelated.
(e)
Interpretation:
The specific type of relationship between the molecules in the given pair is to be determined.
Concept introduction:
If two molecules have different molecular formulas, they are unrelated. If they have the same molecular formula, they may be the same molecule or isomers. If the two can be interconverted by rotation about a bond, they are conformers. If they have different connectivity of atoms, they are constitutional isomers. If they have the same connectivity, but are not conformers, they must be stereoisomers. Stereoisomers may be enantiomers or diastereomers depending on whether they have nonsuperimposable mirror images or not.
Answer to Problem 5.71P
The two molecules are enantiomers.
Explanation of Solution
The structures of the two molecules are:
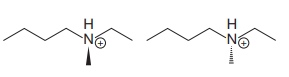
The chemical formulas of both molecules are the same,
The nitrogen atom in each molecule is bonded to four different groups;
The specific relation between the two isomers can be determined from the configuration at the nitrogen atom.
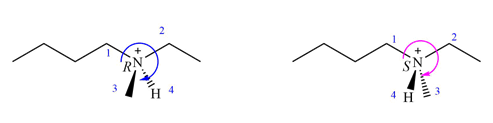
The priorities of
In the first molecule, the 1 to 3 priority groups are arranged clockwise with the lowest priority H at the back. Therefore, the configuration at this nitrogen is R.
In the second molecule also, they are arranged clockwise, but with the lowest priority H in front. Therefore, the configuration at the nitrogen in second molecule is S.
Thus, they are enantiomer.
The molecules are enantiomers because they have different configurations and are the only chiral center.
(f)
Interpretation:
The specific type of relationship between the molecules in the given pair is to be determined.
Concept introduction:
If two molecules have different molecular formulas, they are unrelated. If they have the same molecular formula, they may be the same molecule or isomers. If the two can be interconverted by rotation about a bond, they are conformers. If they have different connectivity of atoms, they are constitutional isomers. If they have the same connectivity, but are not conformers, they must be stereoisomers. Stereoisomers may be enantiomers or diastereomers depending on whether they have nonsuperimposable mirror images or not.
Answer to Problem 5.71P
The two molecules are conformers.
Explanation of Solution
The structures of the molecules are:
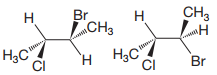
The molecules have the same molecular formula and same connectivity. Therefore, they are either conformers or configurational isomers.
If rotation of one group in the first molecule about a single bond converts it to the other structure, then they must be conformers. If not, they would be configurational isomers.
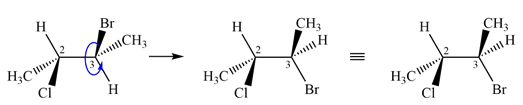
Rotating C3 of first molecule about
Therefore, these two molecules are two conformers of the same molecule.
The two molecules are conformers as they differ from each other only by rotation about a single bond.
(g)
Interpretation:
The specific type of relationship between the molecules in the given pair is to be determined.
Concept introduction:
If two molecules have different molecular formulas, they are unrelated. If they have the same molecular formula, they may be the same molecule or isomers. If the two can be interconverted by rotation about a bond, they are conformers. If they have different connectivity of atoms, they are constitutional isomers. If they have the same connectivity, but are not conformers, they must be stereoisomers. Stereoisomers may be enantiomers or diastereomers depending on whether they have nonsuperimposable mirror images or not.
Answer to Problem 5.71P
The two molecules are enantiomers.
Explanation of Solution
Structures of the molecules are:
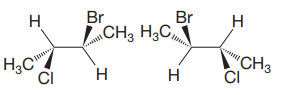
The molecules have the same formula and same connectivity. Therefore, they must be either conformers or configurational isomers. The exact relation between the two molecules can be determined from the configurations about the chiral centers in the two. Each molecule contains two chiral centers, C2 and C3.
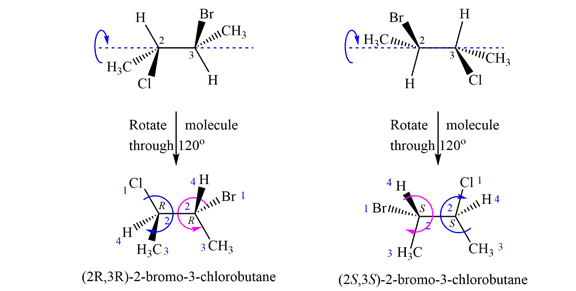
The structures of both are shown with the lowest priority H atoms in the plane of the paper. Therefore, the molecules are rotated about the
The priorities of the four groups attached to C2 and C3 are then determined.
In the first molecule, on C2, chlorine is assigned the highest priority as it is the heaviest (highest
For C2, 1 to 3 priority groups are arranged clockwise with H at the back. On C3, 1 to 3 priority groups are arranged counterclockwise, but with H in the front. Therefore, the configurations are
A similar analysis shows that the configurations of the two chiral centers in second molecule are
Therefore, the two molecules are enantiomers.
Two molecules with multiple chiral centers are enantiomers if they have different configuration at each chiral center.
(h)
Interpretation:
The specific type of relationship between the molecules in the given pair is to be determined.
Concept introduction:
If two molecules have different molecular formulas, they are unrelated. If they have the same molecular formula, they may be the same molecule or isomers. If the two can be interconverted by rotation about a bond, they are conformers. If they have different connectivity of atoms, they are constitutional isomers. If they have the same connectivity, but are not conformers, they must be stereoisomers. Stereoisomers may be enantiomers or diastereomers depending on whether they have nonsuperimpoable mirror images or not. Molecules with multiple chiral centers are diastereomers if the configuration at some but not all centers is different.
Answer to Problem 5.71P
The two molecules are diastereomers.
Explanation of Solution
The structures of the molecules are:
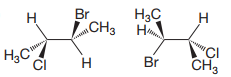
The molecules have the same formulas and same connectivities. Therefore, they are stereoisomers. They are not interconverted by rotation about a single bond, therefore, they are not conformers.
The exact relation can then be determined from the configurations at the two chiral centers they contain.
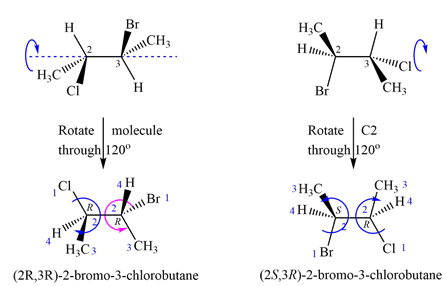
The priorities assigned according to Cahn-Ingold-Prelog rules to the four groups on the chiral centers are as shown above.
In the first molecule, 1 to 3 priority groups on C2 are arranged clockwise with the lowest priority H at the back. 1 to 3 priority groups on C3 are arranged counterclockwise with the lowest priority H in the front. Therefore, the configurations of the chiral centers in the first molecule are
A similar analysis shows that the configurations of the chiral centers in the second molecule are
Therefore, these molecules are diastereomers.
Molecules containing multiple chiral centers are diastereomers if they differ in configuration at only some of the chiral centers.
Want to see more full solutions like this?
Chapter 5 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- Problem 6-29 Identify the functional groups in the following molecules, and show the polarity of each: (a) CH3CH2C=N CH, CH, COCH (c) CH3CCH2COCH3 NH2 (e) OCH3 (b) (d) O Problem 6-30 Identify the following reactions as additions, eliminations, substitutions, or rearrangements: (a) CH3CH2Br + NaCN CH3CH2CN ( + NaBr) Acid -OH (+ H2O) catalyst (b) + (c) Heat NO2 Light + 02N-NO2 (+ HNO2) (d)arrow_forwardPredict the organic product of Y that is formed in the reaction below, and draw the skeletal ("line") structures of the missing organic product. Please include all steps & drawings & explanations.arrow_forwardPlease choose the best reagents to complete the following reactionarrow_forward
- Problem 6-17 Look at the following energy diagram: Energy Reaction progress (a) Is AG for the reaction positive or negative? Label it on the diagram. (b) How many steps are involved in the reaction? (c) How many transition states are there? Label them on the diagram. Problem 6-19 What is the difference between a transition state and an intermediate? Problem 6-21 Draw an energy diagram for a two-step reaction with Keq > 1. Label the overall AG°, transition states, and intermediate. Is AG° positive or negative? Problem 6-23 Draw an energy diagram for a reaction with Keq = 1. What is the value of AG° in this reaction?arrow_forwardProblem 6-37 Draw the different monochlorinated constitutional isomers you would obtain by the radical chlorination of the following compounds. (b) (c) Problem 6-39 Show the structure of the carbocation that would result when each of the following alkenes reacts with an acid, H+. (a) (b) (c)arrow_forwardPlease draw the major product of this reaction. Ignore inorganic byproducts and the carboxylic side productarrow_forward
- predict the product formed by the reaction of one mole each of cyclohex-2-en-1-one and lithium diethylcuprate. Assume a hydrolysis step follows the additionarrow_forwardPlease handwriting for questions 1 and 3arrow_forwardIs (CH3)3NHBr an acidic or basic salt? What happens when dissolved in aqueous solution? Doesn't it lose a Br-? Does it interact with the water? Please advise.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

