
Concept explainers
(a)
Interpretation: The relation between the compounds A and B is to be determined.
Concept introduction: Newman projections are drawn to visualize the different conformations of a compound around
The stereochemistry of the compound is determined by prioritizing the groups attached to its stereogenic center. The groups are prioritized on the basis of
Answer to Problem 5.60P
Compounds A and B are identical.
Explanation of Solution
The given Newman projection A is,
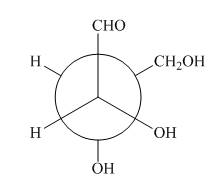
Figure 1
In Newman projection, the circle represents the back carbon and the dot represents the front carbon. The back carbon is substituted by one hydroxyl, one hydrogen atom and one hydroxymethyl group and the front carbon is substituted by one hydrogen atom, one hydroxyl group and one formyl group. The skeletal structure for the given projection is,
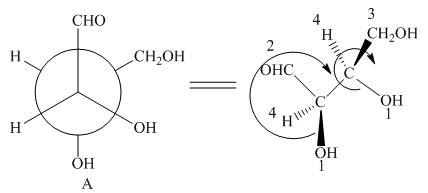
Figure 2
In the given compound,
The given Newman projection B is,
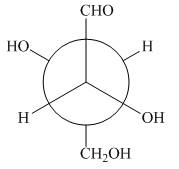
Figure 3
In Newman projection the circle represents the back carbon and the dot represents the front carbon. The back carbon is substituted by one hydroxyl, one hydrogen atom and one hydroxymethyl group and the front carbon is substituted by one hydrogen atom, one hydroxyl group and one formyl group. The skeletal structure for the given projection is,
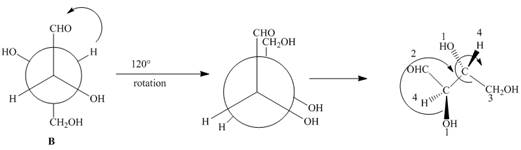
Figure 4
In the given compound,
Both A and B have same configuration around the stereogenic centers. Hence, compounds A and B are identical.
Compounds A and B are identical.
(b)
Interpretation: The relation between the compounds A and C is to be determined.
Concept introduction: Newman projections are drawn to visualize the different conformations of a compound around
The stereochemistry of the compound is determined by prioritizing the groups attached to its stereogenic center. The groups are prioritized on the basis of atomic number of their atoms. The group that contain atom with higher atomic number is given higher priority. Complete the circle in decreasing order of priority from
Answer to Problem 5.60P
Compounds A and C are enantiomers.
Explanation of Solution
The given Newman projection A is,

Figure 1
In Newman projection the circle represents the back carbon and the dot represents the front carbon. The back carbon is substituted by one hydroxyl, one hydrogen atom and one hydroxymethyl group and the front carbon is substituted by one hydrogen atom, one hydroxyl group and one formyl group. The skeletal structure for the given projection is,

Figure 2
In the given compound,
The given Newman projection C is,
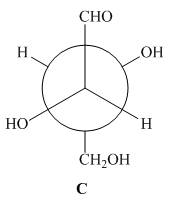
Figure 5
In Newman projection the circle represents the back carbon and the dot represents the front carbon. The back carbon is substituted by one hydroxyl, one hydrogen atom and one hydroxymethyl group and the front carbon is substituted by one hydrogen atom, one hydroxyl group and one formyl group. The skeletal structure for the given projection is,
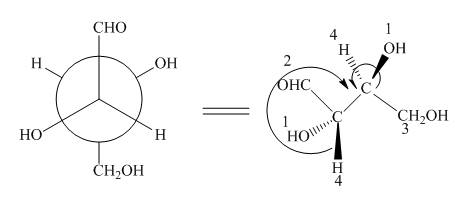
Figure 6
In the given compound,
Both A and C have opposite configuration around the stereogenic centers. Hence, compounds A and C are enantiomers.
Compounds A and C are enantiomers.
(c)
Interpretation: The relation between the compounds A and D is to be determined.
Concept introduction: Newman projections are drawn to visualize the different conformations of a compound around
Enantiomer of a compound has opposite configuration at stereogenic centers. Diastereomers of a compound have at least one stereogenic centre with same configuration and at least one with opposite configuration.
Answer to Problem 5.60P
Compounds A and D are diastereomers.
Explanation of Solution
The given Newman projection A is,

Figure 1
In Newman projection, the circle represents the back carbon and the dot represents the front carbon. The back carbon is substituted by one hydroxyl, one hydrogen atom and one hydroxymethyl group and the front carbon is substituted by one hydrogen atom, one hydroxyl group and one formyl group. The skeletal structure for the given projection is,

Figure 2
In the given compound,
The given Newman projection D is,
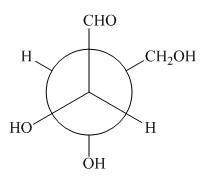
Figure 7
In Newman projection the circle represents the back carbon and the dot represents the front carbon. The back carbon is substituted by one hydroxyl, one hydrogen atom and one hydroxymethyl group and the front carbon is substituted by one hydrogen atom, one hydroxyl group and one formyl group. The skeletal structure for the given projection is,
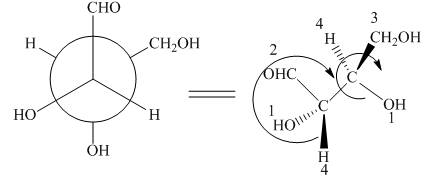
Figure 8
In the given compound,
Both A and D have one stereogenic centre with same configuration and one with opposite configuration. Hence, A and D are diastereomers.
Compounds A and D are diastereomers.
(d)
Interpretation: The relation between the compounds C and D is to be determined.
Concept introduction: Newman projections are drawn to visualize the different conformations of a compound around
Enantiomer of a compound has opposite configuration at stereogenic centers. Diastereomers of a compound have at least one stereogenic centre with same configuration and at least one with opposite configuration.
Answer to Problem 5.60P
Compounds C and D are diastereomers.
Explanation of Solution
The given Newman projection C is,

Figure 5
In Newman projection the circle represents the back carbon and the dot represents the front carbon. The back carbon is substituted by one hydroxyl, one hydrogen atom and one hydroxymethyl group and the front carbon is substituted by one hydrogen atom, one hydroxyl group and one formyl group. The skeletal structure for the given projection is,

Figure 6
In the given compound,
The given Newman projection D is,

Figure 7
In Newman projection the circle represents the back carbon and the dot represents the front carbon. The back carbon is substituted by one hydroxyl, one hydrogen atom and one hydroxymethyl group and the front carbon is substituted by one hydrogen atom, one hydroxyl group and one formyl group. The skeletal structure for the given projection is,

Figure 8
In the given compound,
Both C and D have one stereogenic centre with same configuration and one with opposite configuration. Hence, compounds C and D are diastereomers.
Compounds C and D are diastereomers.
Want to see more full solutions like this?
Chapter 5 Solutions
Organic Chemistry
- Can I get helpp drawing my arrowsarrow_forwardWhich of the m/z values corresponds to the base peak in the mass spectrum shown? 100 80 A. 45 B. 44 C. 29 D. 15 Intensity 20 0 10 20 30 40 B- m/z -8 50 E. 30 Which of the m/z values correspond to the molecular ion for the compound shown? A. 18 B. 82 OH C. 100 D. 102 E. 103arrow_forwardCan someone help me with drawing my arrows.arrow_forward
- I'm having trouble with converting lewis diagrams into VSEPR diagrams. I currently have this example of C2BrCl3 which I want to turn into a lewis structure, but I'm not sure what steps I need to do in order to do so. I have the table written down, however, there's two central atoms so what would I do? There seems to be 4 electron domains on the carbon atom and no lone pairs so it would seem like this shape would be tetrahedral. Here's what I have now. Thanks!arrow_forwardWe discussed the solid phase resin using in peptide synthesis. Provide a mechanism, for its formation. DRAW THE MECHANISM.arrow_forwardPlease help. Every time I've asked an expert in the past, it's been wrong :(arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning




