
Pearson eText Microbiology: An Introduction -- Instant Access (Pearson+)
13th Edition
ISBN: 9780135789377
Author: Gerard Tortora, Berdell Funke
Publisher: PEARSON+
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 2A
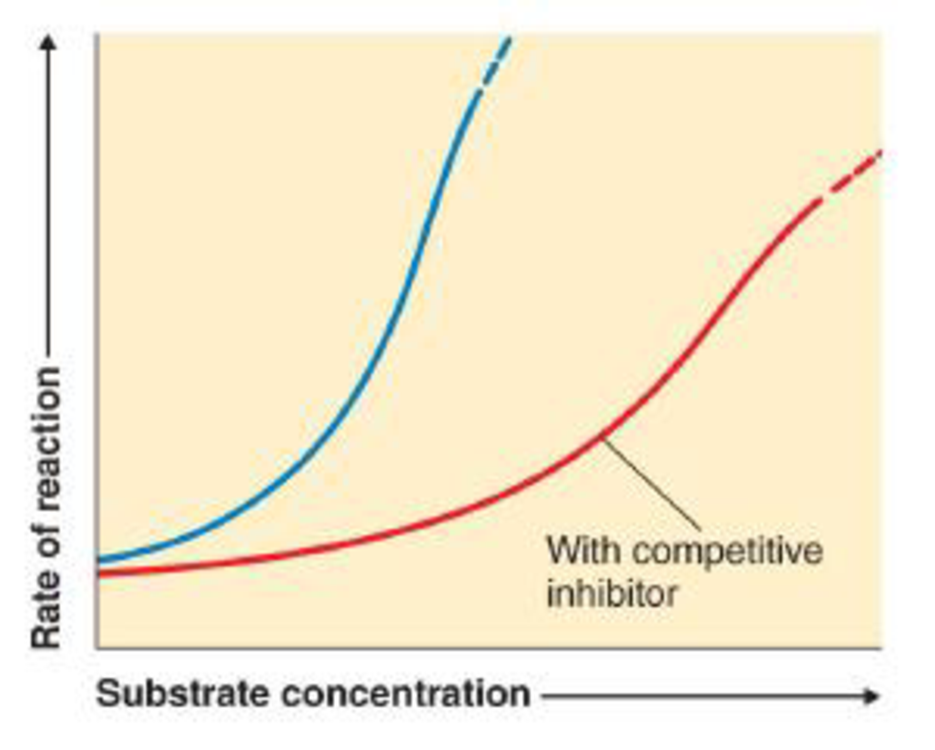
The following graph shows the normal
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
(This is a 2-part work, Part 1 is done, only Part 2 is to be worked on at the bottom)Part 1 (Done): Describe the levels of structural hierarchy for the human body, starting with the organismal level and ending with the chemical level. In addition, you should make sure you link each level to the previous level, emphasizing the structural relationships.
The human body is organized into a structural hierarchy that progresses from the macroscopic organismal level down to the microscopic chemical level. Each level builds upon the previous one, creating increasingly complex structures. Below is the hierarchy, from largest to smallest, with explanations linking each level to its predecessor:
1. Organismal Level
The entire living human body.Relationship: All lower levels work together to sustain life at this highest level.
2. Organ System Level
Groups of organs working together to perform major functions.Examples: circulatory, nervous, digestive systems.Relationship: The organismal…
Describe the levels of structural hierarchy for the human body, starting with the organismal level and ending with the chemical level. In addition, you should make sure you link each level to the previous level, emphasizing the structural relationships.
9
S
es
Read the section "Investigating Life: In (Extremely) Cold Blood." Then, drag and drop the terms on the left to complete the concept
map.
Red blood cells
Genes
Icefishes
-have mutated
have colorless
Oxygen
have few
lack
encode
Blood
Cellular
respiration
consists of-
contain
carries
is a
Platelets
White blood
cells
carries
low
amounts
of
Hemoglobin
is necessary for
Plasma
Protein
Reset.
Chapter 5 Solutions
Pearson eText Microbiology: An Introduction -- Instant Access (Pearson+)
Ch. 5 - Prob. 1RCh. 5 - DRAW ITUsing the diagrams below, show each of the...Ch. 5 - DRAW IT An enzyme and substrate are combined. The...Ch. 5 - Define oxidation-reduction, and differentiate the...Ch. 5 - There are three mechanisms for the phosphorylation...Ch. 5 - All of the energy-producing biochemical reactions...Ch. 5 - Fill in the following table with the carbon source...Ch. 5 - Write your own definition of the chemiosmotic...Ch. 5 - Why must NADH be reoxidized? How does this happen...Ch. 5 - NAME IT What nutritional type is a colorless...
Ch. 5 - Which substance in the following reaction is being...Ch. 5 - Which of the following reactions produces the most...Ch. 5 - Prob. 3MCQCh. 5 - Which of the following compounds has the greatest...Ch. 5 - Prob. 5MCQCh. 5 - Prob. 6MCQCh. 5 - Which culture produces the most lactic acid? Use...Ch. 5 - Which culture produces the most ATP? Use the...Ch. 5 - Which culture uses NAD+? Use the following choices...Ch. 5 - Which culture uses the most glucose? Use the...Ch. 5 - Explain why, even under ideal conditions,...Ch. 5 - The following graph shows the normal rate of...Ch. 5 - Compare and contrast carbohydrate catabolism and...Ch. 5 - How much ATP could be obtained from the complete...Ch. 5 - The chemoautotroph Acidithiobacillus can obtain...Ch. 5 - Haemophilus influenzae requires hemin (X factor)...Ch. 5 - The drug Hivid, also called ddC, inhibits DNA...Ch. 5 - The bacterial enzyme streptokinase is used to...
Additional Science Textbook Solutions
Find more solutions based on key concepts
1. Suppose a chloride ion and a sodium ion are separated by a center—center distance of 5 Å. Is
the interactio...
Biochemistry: Concepts and Connections (2nd Edition)
Why do scientists think that all forms of life on earth have a common origin?
Genetics: From Genes to Genomes
Gregor Mendel never saw a gene, yet he concluded that some inherited factors were responsible for the patterns ...
Campbell Essential Biology (7th Edition)
Describe the role and impact of microbes on the earth.
Microbiology Fundamentals: A Clinical Approach
On what molecule does the anticodon appear? Explain the role of this molecule in protein synthesis.
Human Physiology: An Integrated Approach (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Plating 50 microliters of a sample diluted by a factor of 10-6 produced 91 colonies. What was the originalcell density (CFU/ml) in the sample?arrow_forwardEvery tutor here has got this wrong, don't copy off them.arrow_forwardSuppose that the population from question #1 (data is in table below) is experiencing inbreeding depression (F=.25) (and no longer experiencing natural selection). Calculate the new expected genotype frequencies (f) in this population after one round of inbreeding. Please round to 3 decimal places. Genotype Adh Adh Number of Flies 595 Adh Adh 310 Adhs Adhs 95 Total 1000 fladh Adh- flAdn Adh fAdhs Adharrow_forward
- Which of the following best describes why it is difficult to develop antiviral drugs? Explain why. A. antiviral drugs are very difficult to develop andhave no side effects B. viruses are difficult to target because they usethe host cell’s enzymes and ribosomes tometabolize and replicate C. viruses are too small to be targeted by drugs D. viral infections usually clear up on their ownwith no problemsarrow_forwardThis question has 3 parts (A, B, & C), and is under the subject of Nutrition. Thank you!arrow_forwardThey got this question wrong the 2 previous times I uploaded it here, please make sure it's correvct this time.arrow_forward
- This question has multiple parts (A, B & C), and under the subject of Nutrition. Thank you!arrow_forwardCalculate the CFU/ml of a urine sample if 138 E. coli colonies were counted on a Nutrient Agar Plate when0.5 mls were plated on the NA plate from a 10-9 dilution tube. You must highlight and express your answerin scientific notatioarrow_forwardDon't copy off the other answer if there is anyarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning


Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Principles Of Radiographic Imaging: An Art And A ...
Health & Nutrition
ISBN:9781337711067
Author:Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:Cengage Learning

Biology (MindTap Course List)
Biology
ISBN:9781337392938
Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:Cengage Learning


Enzyme Kinetics; Author: MIT OpenCourseWare;https://www.youtube.com/watch?v=FXWZr3mscUo;License: Standard Youtube License