
THERMODYNAMICS LLF W/ CONNECT ACCESS
9th Edition
ISBN: 9781264446889
Author: CENGEL
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4.5, Problem 129RP
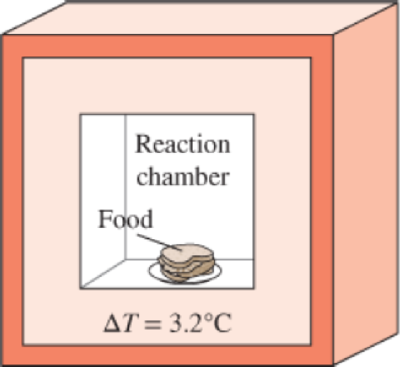
The energy content of a certain food is to be determined in a bomb calorimeter that contains 3 kg of water by burning a 2-g sample of it in the presence of 100 g of air in the reaction chamber. If the water temperature rises by 3.2°C when equilibrium is established, determine the energy content of the food, in kJ/kg, by neglecting the thermal energy stored in the reaction chamber and the energy supplied by the mixer. What is a rough estimate of the error involved in neglecting the thermal energy stored in the reaction chamber?
FIGURE P4–129
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Assume multiple single degree of freedom systems with natural periods T ∈ [0.05, 2.00] seconds with in-crement of period dT = 0.05 seconds. Assume three cases of damping ratio: Case (A) ξ = 0%; Case (B)ξ = 2%; Case (C) ξ = 5%. The systems are initially at rest. Thus, the initial conditions are u(t = 0) = 0 anḋu(t = 0) = 0. The systems are subjected to the base acceleration that was provided in the ElCentro.txt file(i.e., first column). For the systems in Case (A), Case (B), and Case (C) and for each natural period computethe peak acceleration, peak velocity, and peak displacement responses to the given base excitation. Please,use the Newmark method for β = 1/4 (average acceleration) to compute the responses. Create threeplots with three lines in each plot. The first plot will have the peak accelerations in y-axis and the naturalperiod of the system in x-axis. The second plot will have the peak velocities in y-axis and the natural periodof the system in x-axis. The third plot will have…
Both portions of the rod ABC are made of an aluminum for which E = 70 GPa.
Based on the given information find:
1- deformation at A
2- stress in BC
3- Total strain
4- If v (Poisson ratio is 0.25, find the
lateral deformation of AB
Last 3 student ID+ 300 mm=L2
724
A
P=Last 2 student ID+ 300 KN
24
24
Diameter Last 2 student ID+ 15 mm
Last 3 student ID+ 500 mm=L1
724
C
B
24
Q=Last 2 student ID+ 100 KN
24
Diameter Last 2 student ID+ 40 mm
Q2Two wooden members of uniform cross section are joined by the simple scarf splice shown. Knowing that the
maximum allowable tensile stress in the glued splice is 75 psi, determine (a) the largest load P that can be safely
supported, (b) the corresponding shearing stress in the splice.
น
Last 1 student ID+5 inch=W
=9
4
L=Last 1 student ID+8 inch
=12
60°
P'
Chapter 4 Solutions
THERMODYNAMICS LLF W/ CONNECT ACCESS
Ch. 4.5 - Is the boundary work associated with...Ch. 4.5 - On a P-V diagram, what does the area under the...Ch. 4.5 - An ideal gas at a given state expands to a fixed...Ch. 4.5 - Calculate the total work, in kJ, for process 13...Ch. 4.5 - Calculate the total work, in Btu, produced by the...Ch. 4.5 - Nitrogen at an initial state of 300 K, 150 kPa,...Ch. 4.5 - The volume of 1 kg of helium in a pistoncylinder...Ch. 4.5 - A pistoncylinder device with a set of stops...Ch. 4.5 - A mass of 5 kg of saturated water vapor at 150 kPa...Ch. 4.5 - A frictionless pistoncylinder device contains 16...
Ch. 4.5 - 1 m3 of saturated liquid water at 200C is expanded...Ch. 4.5 - Argon is compressed in a polytropic process with n...Ch. 4.5 - A gas is compressed from an initial volume of 0.42...Ch. 4.5 - A mass of 1.5 kg of air at 120 kPa and 24C is...Ch. 4.5 - During some actual expansion and compression...Ch. 4.5 - A frictionless pistoncylinder device contains 5 kg...Ch. 4.5 - During an expansion process, the pressure of a gas...Ch. 4.5 - A pistoncylinder device initially contains 0.4 kg...Ch. 4.5 - A pistoncylinder device contains 0.15 kg of air...Ch. 4.5 - Determine the boundary work done by a gas during...Ch. 4.5 - 1 kg of water that is initially at 90C with a...Ch. 4.5 - An ideal gas undergoes two processes in a...Ch. 4.5 - A pistoncylinder device contains 50 kg of water at...Ch. 4.5 - Prob. 26PCh. 4.5 - A closed system like that shown in Fig. P427E is...Ch. 4.5 - A rigid container equipped with a stirring device...Ch. 4.5 - Complete each line of the following table on the...Ch. 4.5 - A substance is contained in a well-insulated rigid...Ch. 4.5 - A 0.5-m3rigid tank contains refrigerant-134a...Ch. 4.5 - A 20-ft3 rigid tank initially contains saturated...Ch. 4.5 - A rigid 10-L vessel initially contains a mixture...Ch. 4.5 - A rigid 1-ft3 vessel contains R-134a originally at...Ch. 4.5 - A pistoncylinder device contains 5 kg of...Ch. 4.5 - A pistoncylinder device contains 0.5 lbm of water...Ch. 4.5 - 2 kg of saturated liquid water at 150C is heated...Ch. 4.5 - An insulated pistoncylinder device contains 5 L of...Ch. 4.5 - A 40-L electrical radiator containing heating oil...Ch. 4.5 - Steam at 75 kPa and 8 percent quality is contained...Ch. 4.5 - A pistoncylinder device initially contains 0.6 m3...Ch. 4.5 - An insulated tank is divided into two parts by a...Ch. 4.5 - Two tanks (Tank A and Tank B) are separated by a...Ch. 4.5 - Is the energy required to heat air from 295 to 305...Ch. 4.5 - A fixed mass of an ideal gas is heated from 50 to...Ch. 4.5 - A fixed mass of an ideal gas is heated from 50 to...Ch. 4.5 - A fixed mass of an ideal gas is heated from 50 to...Ch. 4.5 - Is the relation u = mcv,avgT restricted to...Ch. 4.5 - Is the relation h = mcp,avgT restricted to...Ch. 4.5 - What is the change in the internal energy, in...Ch. 4.5 - Neon is compressed from 100 kPa and 20C to 500 kPa...Ch. 4.5 - What is the change in the enthalpy, in kJ/kg, of...Ch. 4.5 - A mass of 10 g of nitrogen is contained in the...Ch. 4.5 - Determine the internal energy change u of...Ch. 4.5 - Determine the enthalpy change h of oxygen, in...Ch. 4.5 - Is it possible to compress an ideal gas...Ch. 4.5 - Nitrogen in a rigid vessel is cooled by rejecting...Ch. 4.5 - Nitrogen at 100 psia and 300F in a rigid container...Ch. 4.5 - A pistoncylinder device containing carbon-dioxide...Ch. 4.5 - A 3-m3 rigid tank contains hydrogen at 250 kPa and...Ch. 4.5 - 1 kg of oxygen is heated from 20 to 120C....Ch. 4.5 - A 10-ft3 tank contains oxygen initially at 14.7...Ch. 4.5 - A 4-m 5-m 7-m room is heated by the radiator of...Ch. 4.5 - An insulated rigid tank is divided into two equal...Ch. 4.5 - An ideal gas contained in a pistoncylinder device...Ch. 4.5 - A 4-m 5-m 6-m room is to be heated by a...Ch. 4.5 - An insulated pistoncylinder device initially...Ch. 4.5 - Argon is compressed in a polytropic process with n...Ch. 4.5 - An insulated pistoncylinder device contains 100 L...Ch. 4.5 - Air is contained in a variable-load pistoncylinder...Ch. 4.5 - A mass of 15 kg of air in a pistoncylinder device...Ch. 4.5 - Prob. 73PCh. 4.5 - A pistoncylinder device contains 2.2 kg of...Ch. 4.5 - A pistoncylinder device contains 4 kg of argon at...Ch. 4.5 - A spring-loaded pistoncylinder device contains 5...Ch. 4.5 - Prob. 78PCh. 4.5 - Prob. 79PCh. 4.5 - A 1-kg block of iron is heated from 25 to 75C....Ch. 4.5 - The state of liquid water is changed from 50 psia...Ch. 4.5 - During a picnic on a hot summer day, all the cold...Ch. 4.5 - An ordinary egg can be approximated as a...Ch. 4.5 - Consider a 1000-W iron whose base plate is made of...Ch. 4.5 - Stainless steel ball bearings ( = 8085 kg/m3 and...Ch. 4.5 - In a production facility, 1.6-in-thick 2-ft 2-ft...Ch. 4.5 - Long cylindrical steel rods ( = 7833 kg/m3 and cp...Ch. 4.5 - An electronic device dissipating 25 W has a mass...Ch. 4.5 - Prob. 90PCh. 4.5 - Prob. 91PCh. 4.5 - Is the metabolizable energy content of a food the...Ch. 4.5 - Is the number of prospective occupants an...Ch. 4.5 - Prob. 94PCh. 4.5 - Prob. 95PCh. 4.5 - Prob. 96PCh. 4.5 - Consider two identical 80-kg men who are eating...Ch. 4.5 - A 68-kg woman is planning to bicycle for an hour....Ch. 4.5 - A 90-kg man gives in to temptation and eats an...Ch. 4.5 - A 60-kg man used to have an apple every day after...Ch. 4.5 - Consider a man who has 20 kg of body fat when he...Ch. 4.5 - Consider two identical 50-kg women, Candy and...Ch. 4.5 - Prob. 103PCh. 4.5 - Prob. 104PCh. 4.5 - Prob. 105PCh. 4.5 - Prob. 106PCh. 4.5 - Prob. 107PCh. 4.5 - Prob. 108PCh. 4.5 - Prob. 109RPCh. 4.5 - Prob. 110RPCh. 4.5 - Prob. 111RPCh. 4.5 - Prob. 112RPCh. 4.5 - Prob. 113RPCh. 4.5 - Consider a pistoncylinder device that contains 0.5...Ch. 4.5 - Prob. 115RPCh. 4.5 - Air in the amount of 2 lbm is contained in a...Ch. 4.5 - Air is expanded in a polytropic process with n =...Ch. 4.5 - Nitrogen at 100 kPa and 25C in a rigid vessel is...Ch. 4.5 - Prob. 119RPCh. 4.5 - A mass of 3 kg of saturated liquidvapor mixture of...Ch. 4.5 - A mass of 12 kg of saturated refrigerant-134a...Ch. 4.5 - Prob. 122RPCh. 4.5 - A pistoncylinder device contains helium gas...Ch. 4.5 - Prob. 124RPCh. 4.5 - Prob. 125RPCh. 4.5 - Prob. 126RPCh. 4.5 - Prob. 127RPCh. 4.5 - Water is boiled at sea level in a coffeemaker...Ch. 4.5 - The energy content of a certain food is to be...Ch. 4.5 - Prob. 130RPCh. 4.5 - An insulated pistoncylinder device initially...Ch. 4.5 - An insulated rigid tank initially contains 1.4 kg...Ch. 4.5 - In order to cool 1 ton of water at 20C in an...Ch. 4.5 - A 0.3-L glass of water at 20C is to be cooled with...Ch. 4.5 - A well-insulated 3-m 4m 6-m room initially at 7C...Ch. 4.5 - Prob. 137RPCh. 4.5 - Prob. 138RPCh. 4.5 - Prob. 140RPCh. 4.5 - A pistoncylinder device initially contains 0.35 kg...Ch. 4.5 - Two 10-ft3 adiabatic tanks are connected by a...Ch. 4.5 - Prob. 143RPCh. 4.5 - Prob. 144RPCh. 4.5 - A 3-m3 rigid tank contains nitrogen gas at 500 kPa...Ch. 4.5 - A 0.5-m3 rigid tank contains nitrogen gas at 600...Ch. 4.5 - A well-sealed room contains 60 kg of air at 200...Ch. 4.5 - A room contains 75 kg of air at 100 kPa and 15C....Ch. 4.5 - Prob. 149FEPCh. 4.5 - A pistoncylinder device contains 5 kg of air at...Ch. 4.5 - Prob. 151FEPCh. 4.5 - A 2-kW electric resistance heater submerged in 5...Ch. 4.5 - Prob. 153FEPCh. 4.5 - 1.5 kg of liquid water initially at 12C is to be...Ch. 4.5 - Prob. 155FEPCh. 4.5 - An ordinary egg with a mass of 0.1 kg and a...Ch. 4.5 - Prob. 157FEPCh. 4.5 - A 6-pack of canned drinks is to be cooled from 18C...Ch. 4.5 - Prob. 159FEPCh. 4.5 - An ideal gas has a gas constant R = 0.3 kJ/kgK and...Ch. 4.5 - A pistoncylinder device contains an ideal gas. The...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Similar questions
- Q4 The two solid shafts are connected by gears as shown and are made of a steel for which the allowable shearing stress is 7000 psi. Knowing the diameters of the two shafts are, respectively, dBC determine the largest torque Tc that can be applied at C. 4 and dEF dBC=Last 1 student ID+3 inch dEF=Last 1 student ID+1 inch 7 R=Last 1 Student ID+5 inch 9 R B Tc 2.5 in. E TF Harrow_forwardExperiment تكنولوجيا السيارات - Internal Forced convenction Heat transfer Air Flow through Rectangular Duct. objective: Study the convection heat transfer of air flow through rectangular duct. Valve Th Top Dead Centre Exhaust Valve Class CP. N; ~ RIVavg Ti K 2.11 Te To 18.8 21.3 45.8 Nath Ne Pre Calculations:. Q = m cp (Te-Ti) m: Varg Ac Acca*b Q=hexp As (Ts-Tm) 2 2.61 18.5 20.846.3 Tm = Te-Ti = 25 AS-PL = (a+b)*2*L Nu exp= Re-Vavy D heep Dh k 2ab a+b Nu Dh the- (TS-Tm) Ts. Tmy Name / Nu exp Naxe بب ارتدان العشريarrow_forwardProcedure:1- Cartesian system, 2D3D,type of support2- Free body diagram3 - Find the support reactions4- If you find a negativenumber then flip the force5- Find the internal force3D∑Fx=0∑Fy=0∑Fz=0∑Mx=0∑My=0\Sigma Mz=02D\Sigma Fx=0\Sigma Fy=0\Sigma Mz=05- Use method of sectionand cut the elementwhere you want to findarrow_forward
- Procedure:1- Cartesian system, 2D3D,type of support2- Free body diagram3 - Find the support reactions4- If you find a negativenumber then flip the force5- Find the internal force3D∑Fx=0∑Fy=0∑Fz=0∑Mx=0∑My=0\Sigma Mz=02D\Sigma Fx=0\Sigma Fy=0\Sigma Mz=05- Use method of sectionand cut the elementwhere you want to findthe internal force andkeep either side of thearrow_forwardProcedure: 1- Cartesian system, 2D3D, type of support 2- Free body diagram 3 - Find the support reactions 4- If you find a negative number then flip the force 5- Find the internal force 3D ∑Fx=0 ∑Fy=0 ∑Fz=0 ∑Mx=0 ∑My=0 ΣMz=0 2D ΣFx=0 ΣFy=0 ΣMz=0 5- Use method of section and cut the element where you want to find the internal force and keep either side of thearrow_forwardProcedure:1- Cartesian system, 2D3D,type of support2- Free body diagram3 - Find the support reactions4- If you find a negativenumber then flip the force5- Find the internal force3D∑Fx=0∑Fy=0∑Fz=0∑Mx=0∑My=0\Sigma Mz=02D\Sigma Fx=0\Sigma Fy=0\Sigma Mz=05- Use method of sectionand cut the elementwhere you want to findthe internal force andkeep either side of thearrow_forward
- Procedure: 1- Cartesian system, 2(D)/(3)D, type of support 2- Free body diagram 3 - Find the support reactions 4- If you find a negative number then flip the force 5- Find the internal force 3D \sum Fx=0 \sum Fy=0 \sum Fz=0 \sum Mx=0 \sum My=0 \Sigma Mz=0 2D \Sigma Fx=0 \Sigma Fy=0 \Sigma Mz=0 5- Use method of section and cut the element where you want to find the internal force and keep either side of the sectionarrow_forwardProcedure: 1- Cartesian system, 2(D)/(3)D, type of support 2- Free body diagram 3 - Find the support reactions 4- If you find a negative number then flip the force 5- Find the internal force 3D \sum Fx=0 \sum Fy=0 \sum Fz=0 \sum Mx=0 \sum My=0 \Sigma Mz=0 2D \Sigma Fx=0 \Sigma Fy=0 \Sigma Mz=0 5- Use method of section and cut the element where you want to find the internal force and keep either side of the sectionarrow_forwardFor each system below with transfer function G(s), plot the pole(s) on the s-plane. and indicate whether the system is: (a) "stable" (i.e., a bounded input will always result in a bounded output), (b) "marginally stable," or (c) "unstable" Sketch a rough graph of the time response to a step input. 8 a) G(s) = 5-5 8 b) G(s) = c) G(s) = = s+5 3s + 8 s² - 2s +2 3s +8 d) G(s): = s²+2s+2 3s+8 e) G(s): = s² +9 f) G(s): 8 00 == Sarrow_forward
- Please answer the following question. Include all work and plase explain. Graphs are provided below. "Consider the Mg (Magnesium) - Ni (Nickel) phase diagram shown below. This phase diagram contains two eutectic reactions and two intermediate phases (Mg2Ni and MgNi2). At a temperature of 505oC, determine what the composition of an alloy would need to be to contain a mass fraction of 0.20 Mg and 0.80 Mg2Ni."arrow_forwardThe triangular plate, having a 90∘∘ angle at AA, supports the load PP = 370 lblb as shown in (Figure 1).arrow_forwardDesign a 4-bar linkage to carry the body in Figure 1 through the two positions P1 and P2 at the angles shown in the figure. Use analytical synthesis with the free choice values z = 1.075, q= 210°, ß2 = −27° for left side and s = 1.24, y= 74°, ½ = − 40° for right side. φ 1.236 P2 147.5° 210° 2.138 P1 Figure 1 Xarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY

Elements Of Electromagnetics
Mechanical Engineering
ISBN:9780190698614
Author:Sadiku, Matthew N. O.
Publisher:Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:9780134319650
Author:Russell C. Hibbeler
Publisher:PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:9781259822674
Author:Yunus A. Cengel Dr., Michael A. Boles
Publisher:McGraw-Hill Education

Control Systems Engineering
Mechanical Engineering
ISBN:9781118170519
Author:Norman S. Nise
Publisher:WILEY

Mechanics of Materials (MindTap Course List)
Mechanical Engineering
ISBN:9781337093347
Author:Barry J. Goodno, James M. Gere
Publisher:Cengage Learning

Engineering Mechanics: Statics
Mechanical Engineering
ISBN:9781118807330
Author:James L. Meriam, L. G. Kraige, J. N. Bolton
Publisher:WILEY
First Law of Thermodynamics, Basic Introduction - Internal Energy, Heat and Work - Chemistry; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=NyOYW07-L5g;License: Standard youtube license