
Write a net ionic equation for any precipitation reaction that occurs when 1 M solutions of the following are mixed.
(a) copper(II) sulfate and sodium chloride
(b) manganese(II) nitrate and ammonium hydroxide
(c) silver nitrate and hydrochloric acid
(d) nickel(II) sulfate and potassium hydroxide
(e) ammonium carbonate and sodium nitrate
(a)
Interpretation:
The net ionic equation should be written when copper(II) sulfate and sodium chloride are mixed.
Concept introduction:
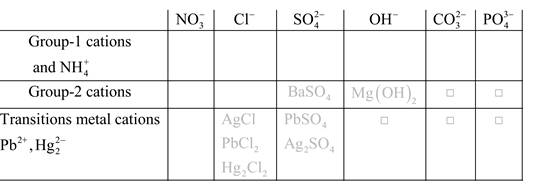
Solubility of any compound is predicted by above solubility chart.
Blank boxes indicate no precipitate formation occurs which means soluble in dilute solution.
Boxes with grey small box will form precipitate from dilute solutions and boxes where formula is written this is a cation-anion combination that will form precipitate.
Precipitation reactions: It is a type of chemical reactions where two soluble salts react with each other and formed different products, out of which one product must be insoluble in solution which is known as precipitate.
A chemical equation which shows only the species that are participated in the reaction is said to be net ionic equation.
Answer to Problem 9QAP
No precipitation occurs.
Explanation of Solution
Copper(II) sulfate:
Sodium chloride:
Reaction for the solution of copper(II) sulfate and sodium chloride is written as:
Reactants:
Ions in solution:
Ions in solution:
Products:
Ions in solution:
Ions in solution:
Now,
(b)
Interpretation:
The net ionic equation should be written when manganese(II) nitrate and ammonium hydroxide are mixed.
Concept introduction:
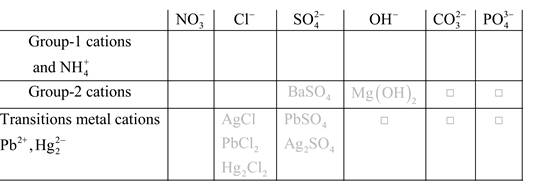
Solubility of any compound is predicted by above solubility chart.
Blank boxes indicate no precipitate formation occurs which means soluble in dilute solution.
Boxes with grey small box will form precipitate from dilute solutions and boxes where formula is written this is a cation-anion combination that will form precipitate.
Precipitation reactions: It is a type of chemical reactions where two soluble salts react with each other and formed different products, out of which one product must be insoluble in solution which is known as precipitate.
A chemical equation which shows only the species that are participated in the reaction is said to be net ionic equation.
Answer to Problem 9QAP
Precipitation occurs
The net ionic equation is:
Explanation of Solution
Manganese(II) nitrate:
Ammonium hydroxide:
Reaction for the solution of manganese(II) nitrate and ammonium hydroxide is written as:
Reactants:
Ions in solution:
Ions in solution:
Products:
Ions in solution:
Ions in solution:
Now,
So, the equation will be:
Now, cancelling out the ions which appear on both sides of the equation (
(c)
Interpretation:
The net ionic equation should be written when silver nitrate and hydrochloric acid are mixed.
Concept introduction:
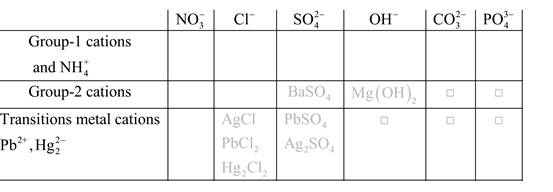
Solubility of any compound is predicted by above solubility chart.
Blank boxes indicate no precipitate formation occurs which means soluble in dilute solution.
Boxes with grey small box will form precipitate from dilute solutions and boxes where formula is written this is a cation-anion combination that will form precipitate.
Precipitation reactions: It is a type of chemical reactions where two soluble salts react with each other and formed different products, out of which one product must be insoluble in solution which is known as precipitate.
A chemical equation which shows only the species that are participated in the reaction is said to be net ionic equation.
Answer to Problem 9QAP
Precipitation occurs
The net ionic equation is:
Explanation of Solution
Silver nitrate:
Hydrochloric acid:
Reaction for the solution of silver nitrate and hydrochloric acid is written as:
Reactants:
Ions in solution:
Ions in solution:
Products:
Ions in solution:
Ions in solution:
Now,
So, the equation will be:
Now, cancelling out the ions which appear on both sides of the equation (
(d)
Interpretation:
The net ionic equation should be written when nickel(II) sulfate and potassium hydroxide are mixed.
Concept introduction:
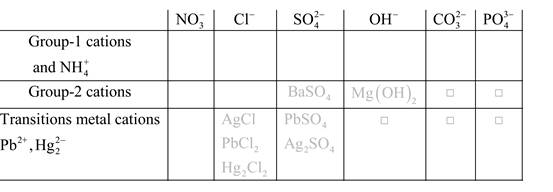
Solubility of any compound is predicted by above solubility chart.
Blank boxes indicate no precipitate formation occurs which means soluble in dilute solution.
Boxes with grey small box will form precipitate from dilute solutions and boxes where formula is written this is a cation-anion combination that will form precipitate.
Precipitation reactions: It is a type of chemical reactions where two soluble salts react with each other and formed different products, out of which one product must be insoluble in solution which is known as precipitate.
A chemical equation which shows only the species that are participated in the reaction is said to be net ionic equation.
Answer to Problem 9QAP
Precipitation occurs
The net ionic equation is:
Explanation of Solution
Nickel(II) sulfate:
Potassium hydroxide:
Reaction for the solution of silver nitrate and hydrochloric acid is written as:
Reactants:
Ions in solution:
Ions in solution:
Products:
Ions in solution:
Ions in solution:
Now,
So, the equation will be:
Now, cancelling out the ions which appear on both sides of the equation (
(e)
Interpretation:
The net ionic equation should be written when ammonium carbonate and sodium nitrate are mixed.
Concept introduction:
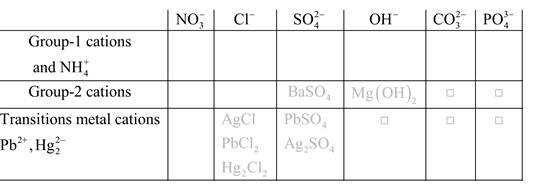
Solubility of any compound is predicted by above solubility chart.
Blank boxes indicate no precipitate formation occurs which means soluble in dilute solution.
Boxes with grey small box will form precipitate from dilute solutions and boxes where formula is written this is a cation-anion combination that will form precipitate.
Precipitation reactions: It is a type of chemical reactions where two soluble salts react with each other and formed different products, out of which one product must be insoluble in solution which is known as precipitate.
A chemical equation which shows only the species that are participated in the reaction is said to be net ionic equation.
Answer to Problem 9QAP
No precipitation occurs.
Explanation of Solution
Ammonium carbonate:
Sodium nitrate:
Reaction for the solution of silver nitrate and hydrochloric acid is written as:
Reactants:
Ions in solution:
Ions in solution:
Products:
Ions in solution:
Ions in solution:
Now,
Want to see more full solutions like this?
Chapter 4 Solutions
EBK CHEMISTRY: PRINCIPLES AND REACTIONS
- AE>AE₁ (Y/N) AE=AE₁ (Y/N) AEarrow_forwardTreatment of 2-phenylpropan-2-amine with methyl 2,4-dibromobutanoate in the presence of a nonnucleophilic base, R3N, involves two successive SN2 reactions and gives compound A. ? NH2 Br Br Propose a structural formula for compound A. You do not have to explicitly draw H atoms. You do not have to consider stereochemistry. In cases where there is more than one answer, just draw one. R3N C14H19NO2 + 2 R3NH*Br Aarrow_forwardCorrectly name this compound using the IUPAC naming system by sorting the components into the correct order. Br IN Ν Harrow_forwardHow is the radical intermediate for this structure formed? Can you please draw arrows from the first radical to the resonance form that would result in this product? I'm lost.arrow_forwardPart VI. (a) calculate the λ max of the compound using woodward - Fieser rules. (b) what types of electronic transitions are present in the compound? (c) what are the prominent peaks in the IR spectrum of the compound?arrow_forwardDon't used Ai solutionarrow_forwardPlease correct answer and don't used hand raitingarrow_forward↑ 0 Quiz List - RCC430M_RU05 X Aktiv Learning App × Qdraw resonance structure ×Q draw resonance structure xb My Questions | bartleby ×+ https://app.aktiv.com Draw a resonance structure of pyrrole that has the same number of pi bonds as the original structure. Include all lone pairs in your structure. + N H a 5 19°F Cloudy Q Search Problem 12 of 15 Atoms, Bonds and Rings Charges and Lone Pairs myhp हजु Undo Reset Remove Done Submit Drag To Pan 2:15 PM 1/25/2025arrow_forwardDon't used hand raitingarrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning





