![OWLv2 with Student Solutions Manual eBook for Masterton/Hurley's Chemistry: Principles and Reactions, 8th Edition, [Instant Access], 4 terms (24 months)](https://www.bartleby.com/isbn_cover_images/9781305863170/9781305863170_largeCoverImage.jpg)
OWLv2 with Student Solutions Manual eBook for Masterton/Hurley's Chemistry: Principles and Reactions, 8th Edition, [Instant Access], 4 terms (24 months)
8th Edition
ISBN: 9781305863170
Author: William L. Masterton; Cecile N. Hurley
Publisher: Cengage Learning US
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4, Problem 68QAP
The following figures represent species before and after they are dissolved in water. Classify each species as weak electrolyte, strong electrolyte, or nonelectrolyte. You may assume that species that dissociate during solution break up as ions.
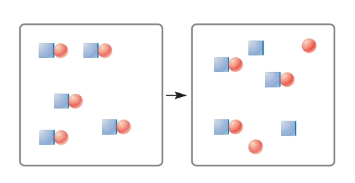
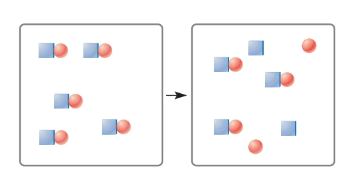
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Don't used hand raiting
Don't used hand raiting
Don't used hand raiting
Chapter 4 Solutions
OWLv2 with Student Solutions Manual eBook for Masterton/Hurley's Chemistry: Principles and Reactions, 8th Edition, [Instant Access], 4 terms (24 months)
Ch. 4 - Write the formulas of the following compounds and...Ch. 4 - Prob. 2QAPCh. 4 - Describe how you would precipitate (a) zinc...Ch. 4 - Name the reagent, if any, that you would add to a...Ch. 4 - Write net ionic equations for the formation of (a)...Ch. 4 - Write net ionic equations to explain the formation...Ch. 4 - Decide whether a precipitate will form when the...Ch. 4 - Follow the directions of Question 7 for solutions...Ch. 4 - Write a net ionic equation for any precipitation...Ch. 4 - Follow the directions for Question 9 for the...
Ch. 4 - What volume of 0.2500 M cobalt(III) sulfate is...Ch. 4 - Prob. 12QAPCh. 4 - A 50.00-mL sample of 0.0250 M silver nitrate is...Ch. 4 - Aluminum ions react with carbonate ions to form an...Ch. 4 - When Na3PO4 and Ca(NO3)2 are combined, the...Ch. 4 - When solutions of aluminum sulfate and sodium...Ch. 4 - Prob. 17QAPCh. 4 - Prob. 18QAPCh. 4 - Prob. 19QAPCh. 4 - Follow the directions of Question 19 for the...Ch. 4 - For an acid-base reaction, what is the reacting...Ch. 4 - Follow the directions of Question 21 for the...Ch. 4 - Prob. 23QAPCh. 4 - Prob. 24QAPCh. 4 - Consider the following generic equation: H+(aq)+...Ch. 4 - Consider the following generic equation...Ch. 4 - Prob. 27QAPCh. 4 - Prob. 28QAPCh. 4 - Prob. 29QAPCh. 4 - Consider several 25.00-mL solutions of perchloric...Ch. 4 - Prob. 31QAPCh. 4 - A student tries to determine experimentally the...Ch. 4 - Prob. 33QAPCh. 4 - For a product to be called vinegar, it must...Ch. 4 - The percentage of sodium hydrogen carbonate,...Ch. 4 - Prob. 36QAPCh. 4 - An artificial fruit beverage contains 12.0 g of...Ch. 4 - Lactic acid, C3H6O3 is the acid present in sour...Ch. 4 - Prob. 39QAPCh. 4 - Prob. 40QAPCh. 4 - Assign oxidation numbers to each element in (a)...Ch. 4 - Assign oxidation numbers to each element in (a)...Ch. 4 - Classify each of the following half-reactions as...Ch. 4 - Classify each of the following half-reactions as...Ch. 4 - Classify each of the following half-reactions as...Ch. 4 - Prob. 46QAPCh. 4 - For each unbalanced equation given below • write...Ch. 4 - Prob. 48QAPCh. 4 - A solution of potassium permanganate reacts with...Ch. 4 - Prob. 50QAPCh. 4 - Hydrogen gas is bubbled into a solution of barium...Ch. 4 - Consider the reaction tween silver and nitric acid...Ch. 4 - The molarity of iodine in solution can be...Ch. 4 - A wire weighing 0.250 g and containing 92.50% Fe...Ch. 4 - Glycerol (C3H8O3) is commonly used as an additive...Ch. 4 - Laws passed in some states define a drunk driver...Ch. 4 - A sample of limestone weighing 1.005 g is...Ch. 4 - The iron content of hemoglobin is determined by...Ch. 4 - In order to determine the purity of ammonium...Ch. 4 - Gold metal will dissolve only in aqua regia, a...Ch. 4 - Prob. 61QAPCh. 4 - Ten mL of concentrated H3PO4 (91.7% by mass,...Ch. 4 - Consider the following balanced redox reaction in...Ch. 4 - Prob. 64QAPCh. 4 - Using circles to represent cations and squares to...Ch. 4 - Assuming that circles represent cations and...Ch. 4 - Consider four beakers. Beaker A has an aqueous...Ch. 4 - The following figures represent species before and...Ch. 4 - A student is asked to identify the metal nitrate...Ch. 4 - Three students titrate different samples of the...Ch. 4 - Calcium in blood or urine can be determined by...Ch. 4 - Copper metal can reduce silver ions to metallic...Ch. 4 - Copper metal can reduce silver ions to metallic...Ch. 4 - A solution contains both iron(II) and iron(III)...Ch. 4 - A student is given 0.930 g of an unknown acid,...Ch. 4 - Solid iron(III) hydroxide is added to 625 mL of...Ch. 4 - A 300.0-g sample of a solid is made up of a...Ch. 4 - When 85.0 mL of 0.250 M Ba(OH)2 solution is added...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- If a high molecular weight linear polyethylene is chlorinated by inducing the substitution of chlorine atoms by hydrogen, if 5% of all hydrogen atoms are replaced, what approximate percentage of chlorine by weight would the product have?arrow_forwardO Macmillan Learning Chemistry: Fundamentals and Principles Davidson presented by Macmillan Learning Poly(ethylene terephthalate), known as PET or industrially as Dacron, is a polyester synthesized through a condensation reaction between two bifunctional monomers. The monomers, ethylene glycol and terepthalic acid, are given. Add bonds and remove atoms as necessary to show the structure of a two repeat unit portion of a longer polymer chain of PET. You may need to zoom out to see the complete structure of all four monomer units. Select Draw / || | C H 0 3 © Templates More ° ° ° || C CC - OH HO OH HOC - C Erase CC OH HO C C 〃 C H₂ Q2Qarrow_forwardc) + H₂Oarrow_forward
- 으 b) + BF. 3 H2Oarrow_forwardQ4: Draw the product of each Lewis acid-bas reaction. Label the electrophile and nucleophile. b) S + AICI 3 + BF 3arrow_forwardQ1 - What type(s) of bonding would be expected for each of the following materials: solid xenon, calcium fluoride (CaF2), bronze, cadmium telluride (CdTe), rubber, and tungsten? Material solid xenon CaF2 bronze CdTe rubber tungsten Type(s) of bonding Q2- If the atomic radius of lead is 0.175 nm, calculate the volume of its unit cell in cubic meters.arrow_forward
- Determine the atomic packing factor of quartz, knowing that the number of Si atoms per cm3 is 2.66·1022 and that the atomic radii of silicon and oxygen are 0.038 and 0.117 nm.arrow_forwardUse the following data for an unknown gas at 300 K to determine the molecular mass of the gas.arrow_forward2. Provide a complete retrosynthetic analysis and a complete forward synthetic scheme to make the following target molecule from the given starting material. You may use any other reagents necessary. Brarrow_forward
- 146. Use the following data for NH3(g) at 273 K to determine B2p (T) at 273 K. P (bar) 0.10 0.20 0.30 0.40 0.50 0.60 (Z -1)/10-4 1.519 3.038 4.557 6.071 7.583 9.002 0.70 10.551arrow_forward110. Compare the pressures given by (a) the ideal gas law, (b) the van der Waals equation, and (c) the Redlic-Kwong equation for propane at 400 K and p = 10.62 mol dm³. The van der Waals parameters for propane are a = 9.3919 dm6 bar mol-2 and b = 0.090494 dm³ mol−1. The Redlich-Kwong parameters are A = 183.02 dm bar mol-2 and B = 0.062723 dm³ mol-1. The experimental value is 400 bar.arrow_forwardResearch in surface science is carried out using stainless steel ultra-high vacuum chambers with pressures as low as 10-12 torr. How many molecules are there in a 1.00 cm3 volume at this pressure and at a temperature of 300 K? For comparison, calculate the number of molecules in a 1.00 cm3 volume at atmospheric pressure and room temperature. In outer space the pressure is approximately 1.3 x 10-11 Pa and the temperature is approximately 2.7 K (determined using the blackbody radiation of the universe). How many molecules would you expect find in 1.00 cm3 of outer space?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning
Solutions: Crash Course Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=9h2f1Bjr0p4;License: Standard YouTube License, CC-BY