
A dilute aqueous solution of H2SO4 (Solution A) is to be mixed with a solution containing 90.0 wt% H2SO4 (Solution B) to produce a 75.0 wt% solution (Solution C).
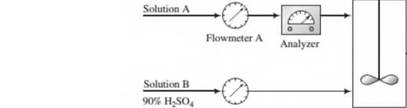
The flow rate and concentration of Solution A change periodically, so that it is necessary to adjust the flow rate of Solution B to keep the product H2SO4 concentration constant.
Flowmeters A and B have linear calibration plots of mass flow rate (m) versus meter reading (/?),
| which pass through the following points: | ||
| Flowmeter A: | mA = 150 lbn,/h, wa = 500 lbm/h. | «a = 25 «a = 70 |
| Flowmeter B: | riiB = 200 lbm/h, mB= 800 lbm/h. | Rb = 20 /?B = 60 |
The analyzer calibration is a straight line on a semilog plot of %H2SO4(x) on a logarithmic scale versus meter reading (/?,) on a linear scale. The line passes through the points (x = 20%, Rx= 4.0) and (x = 100%./?, = 10.0).
- Calculate the flow' rate of Solution B needed to process 300 lbm/h of 55% H2SO4 (Solution A), and the resulting flow rate of Solution C. (The calibration data are not needed for this part.)
- Derive the calibration equations for /ha(#a). «ib(^b), andx(/?v). Calculate the values of R\, Ru, and Rxcorresponding to the flow rates and concentrations of Part (a).
- The process technician's job is to read Flowmeter A and the analyzer periodically, and then to adjust the flow rate of Solution B to its required value. Derive a formula that the technician can use for Rbin terms of RAand Rx, and then check it by substituting the values of Part (a).
Learn your wayIncludes step-by-step video

Chapter 4 Solutions
ELEM.PRIN.OF CHEM.PROCESS-ACCESS
Additional Engineering Textbook Solutions
Introduction To Programming Using Visual Basic (11th Edition)
Starting Out with Java: From Control Structures through Data Structures (4th Edition) (What's New in Computer Science)
Database Concepts (8th Edition)
Starting Out with Python (4th Edition)
Concepts Of Programming Languages
Thinking Like an Engineer: An Active Learning Approach (4th Edition)
- Transmitance 3. Which one of the following compounds corresponds to this IR spectrum? Point out the absorption band(s) that helped you decide. OH H3C OH H₂C CH3 H3C CH3 H3C INFRARED SPECTRUM 0.8- 0.6 0.4- 0.2 3000 2000 1000 Wavenumber (cm-1) 4. Consider this compound: H3C On the structure above, label the different types of H's as A, B, C, etc. In table form, list the labeled signals, and for each one state the number of hydrogens, their shifts, and the splitting you would observe for these hydrogens in the ¹H NMR spectrum. Label # of hydrogens splitting Shift (2)arrow_forwardNonearrow_forwardDraw the Lewis structure of C2H4Oarrow_forward
- a) 5. Circle all acidic (and anticoplanar to the Leaving group) protons in the following molecules, Solve these elimination reactions, and identify the major and minor products where appropriate: 20 points + NaOCH3 Br (2 productarrow_forwardNonearrow_forwardDr. Mendel asked his BIOL 260 class what their height was and what their parent's heights were. He plotted that data in the graph below to determine if height was a heritable trait. A. Is height a heritable trait? If yes, what is the heritability value? (2 pts) B. If the phenotypic variation is 30, what is the variation due to additive alleles? (2 pts) Offspring Height (Inches) 75 67.5 60 52.5 y = 0.9264x + 4.8519 55 60 65 MidParent Height (Inches) 70 75 12pt v V Paragraph B IUA > AT2 v Varrow_forward
- Experiment: Each team will be provided with 5g of a mixture of acetanilide and salicylic acid. You will divide it into three 1.5 g portions in separate 125 mL Erlenmeyer flasks savıng some for melting point analysis. Dissolve the mixture in each flask in ~60mL of DI water by heating to boiling on a hotplate. Take the flasks off the hotplate once you have a clear solution and let them stand on the bench top for 5 mins and then allow them to cool as described below. Sample A-Let the first sample cool slowly to room temperature by letting it stand on your lab bench, with occasional stirring to promote crystallization. Sample B-Cool the second sample 1n a tap-water bath to 10-15 °C Sample C-Cool the third sample in an ice-bath to 0-2 °C Results: weight after recrystalization and melting point temp. A=0.624g,102-115° B=0.765g, 80-105° C=1.135g, 77-108 What is the percent yield of A,B, and C.arrow_forwardRel. Intensity Q 1. Which one of the following is true of the compound whose mass spectrum is shown here? Explain how you decided. 100 a) It contains chlorine. b) It contains bromine. c) It contains neither chlorine nor bromine. 80- 60- 40- 20- 0.0 0.0 TT 40 80 120 160 m/z 2. Using the Table of IR Absorptions how could you distinguish between these two compounds in the IR? What absorbance would one compound have that the other compound does not? HO CIarrow_forwardIllustrate reaction mechanisms of alkenes with water in the presence of H2SO4, detailing each step of the process. Please show steps of processing. Please do both, I will thumb up for sure #1 #3arrow_forward
