
Concept explainers
What is the geometry around the central atom in the following molecular models? (There are no “hidden” atoms: all atoms in each model are visible.)
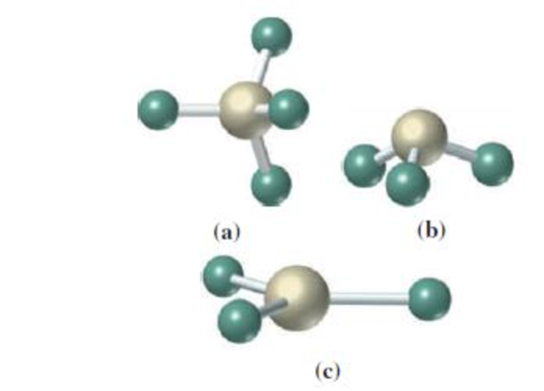
(a)
Interpretation:
The geometry around the central atom in the given molecular model is to be determined.
Concept introduction:
Molecular shape can be predicted from the Lewis structure by using the valence-shell Electron-pair repulsion (VSEPR) model.
- Count the number of valence electron pairs (bond pairs and lone pairs).
- Assume that the valence electron pairs form a structure that allows them to be as far away from each other as possible.
- If there are only two bond pair electrons, the molecule is linear.
- If there are three bond pair electrons, the molecule is shaped like a trigonal planar.
- If there are four bond pair electrons, the molecule is shaped as a regular tetrahedral.
- Repulsion between lone pair-bond pair of electrons effect the geometry of molecules.
Bond angle is the angle between two bonds of a molecule and it is determined based on the geometry.
[Bond angles:
Answer to Problem 4.25UKC
The geometry around the central atom in the given molecular model is tetrahedral.
Explanation of Solution
In the given molecular model,
Four atoms are bonded to the central atom. The angle around the central atom is around
Therefore,
The geometry of the given molecular model is tetrahedral.
(b)
Interpretation:
The geometry around the central atom in the given molecular model is to be determined.
Concept introduction:
Molecular shape can be predicted from the Lewis structure by using the valence-shell Electron-pair repulsion (VSEPR) model.
- Count the number of valence electron pairs (bond pairs and lone pairs).
- Assume that the valence electron pairs form a structure that allows them to be as far away from each other as possible.
- If there are only two bond pair electrons, the molecule is linear.
- If there are three bond pair electrons, the molecule is shaped like a trigonal planar.
- If there are four bond pair electrons, the molecule is shaped as a regular tetrahedral.
- Repulsion between lone pair-bond pair of electrons effect the geometry of molecules.
Bond angle is the angle between two bonds of a molecule and it is determined based on the geometry.
[Bond angles:
Answer to Problem 4.25UKC
The geometry around the central atom in the given molecular model is pyramidal.
Explanation of Solution
In the given molecular model,
Three atoms are bonded to the central atom. Three atoms are bonded to the central atom. The angle around the central atom is around
Therefore,
The geometry of the given molecular model is pyramidal.
(c)
Interpretation:
The geometry around the central atom in the given molecular model is to be determined.
Concept introduction:
Molecular shape can be predicted from the Lewis structure by using the valence-shell Electron-pair repulsion (VSEPR) model.
- Count the number of valence electron pairs (bond pairs and lone pairs).
- Assume that the valence electron pairs form a structure that allows them to be as far away from each other as possible.
- If there are only two bond pair electrons, the molecule is linear.
- If there are three bond pair electrons, the molecule is shaped like trigonal planar.
- If there are four bond pair electrons, the molecule is shaped as a regular tetrahedral.
- Repulsion between lone pair-bond pair of electrons effect the geometry of molecules.
Bond angle is the angle between two bonds of a molecule and it is determined based on the geometry.
[Bond angles:
Answer to Problem 4.25UKC
The geometry around the central atom in the given molecular model is trigonal planar.
Explanation of Solution
In the given molecular model,
Three atoms are bonded to the central atom. The angle around the central atom is around
Therefore,
The geometry of the given molecular model is trigonal planar.
Want to see more full solutions like this?
Chapter 4 Solutions
Pearson eText Fundamentals of General, Organic, and Biological Chemistry -- Instant Access (Pearson+)
- What would be the toxicological endpoints for neurotoxicity?arrow_forwardWhat are "endpoints" in toxicology exactly? Please give an intuitive easy explanationarrow_forwardFura-2 Fluorescence (Arbitrary Unit) 4500 4000 3500 3000 2500 2000 1500 1000 500 [Ca2+]=2970nM, 25°C [Ca2+] 2970nM, 4°C [Ca2+]=0.9nM, 25°C [Ca2+] = 0.9nM, 4°C 0 260 280 300 340 360 380 400 420 440 Wavelength (nm) ← < The figure on the LHS shows the excitation spectra of Fura-2 (Em = 510 nm) in 2 solutions with two different Ca2+ ion concentration as indicated. Except for temperature, the setting for excitation & signal acquisition was identical.< ப a) The unit in Y-axis is arbitrary (unspecified). Why? < < b) Compare & contrast the excitation wavelength of the Isosbestic Point of Fura-2 at 25 °C & 4 °C. Give a possible reason for the discrepancy. < c) The fluorescence intensity at 25 °C & 4 °C are different. Explain why with the concept of electronic configuration. <arrow_forward
- draw in the structure of each amino acid (as L-amino acids) using the Fischer projection style. an example has been included. Draw the structure for glycine, alanine, valine, isoleucine, methionine, proline, phenylalanine, tryptophan, serine, threonine, asparagine, glutamine, lysine, arginine, aspartic acid, glutamic acid, histidine, tyrosine, cysteinearrow_forwarddraw in the structure of each amino acid (as L-amino acids) using the Fischer projection style. an example has been includedarrow_forwarddraw in the structure of each amino acid (as L-amino acids) using the Fischer projection style. an example has been includedarrow_forward
- Draw out the following peptide H-R-K-E-D at physiological pH (~7.4). Make sure toreference table 3.1 for pKa values.arrow_forwardThe table provides the standard reduction potential, E', for relevant half-cell reactions. Half-reaction E'° (V) Oxaloacetate² + 2H+ + 2e malate²- -0.166 Pyruvate + 2H+ + 2e → lactate -0.185 Acetaldehyde + 2H+ + 2e¯ →→→ ethanol -0.197 NAD+ + H+ + 2e--> NADH -0.320 NADP+ + H+ + 2e →→ NADPH Acetoacetate + 2H+ + 2e¯ - -0.324 B-hydroxybutyrate -0.346 Which of the reactions listed would proceed in the direction shown, under standard conditions, in the presence of the appropriate enzymes? Malate + NAD+ oxaloacetate + NADH + H+ Malate + pyruvate oxaloacetate + lactate Pyruvate + NADH + H+ lactate + NAD+ Pyruvate + p-hydroxybutyrate lactate + acetoacetate Acetaldehyde + succinate ethanol + fumerate Acetoacetate + NADH + H+ → B-hydroxybutyrate + NAD+arrow_forwardArrange the four structures in order from most reduced to most oxidized. Most reduced R-CH2-CH3 R-CH2-CH₂-OH R-CH,-CHO R-CH₂-COO Most oxidizedarrow_forward
- for each pair of biomolecules, identify the type of reaction (oxidation-reduction, hydrolysis, isomerization, group transfer, or nternal rearrangement) required to convert the first molecule to the second. In each case, indicate the general type of enzyme and cofactor(s) c reactants required, and any other products that would result. R-CH-CH-CH-C-S-COA A(n) A(n) A(n) A(n) Palmitoyl-CoA R-CH-CH=CH-C-S-CoA ° trans-A-Enoyl-CoA reaction converts palmitoyl-CoA to trans-A2-enoyl-CoA. This reaction requires and also produces Coo HN-C-H CH₂ CH₂ CH CH CH, CH, L-Leucine CH, CH, D-Leucine 8/6881 COO HÌNH: reaction converts L-leucine to D-leucine. This reaction is catalyzed by a(n) H-C-OH H-C-OH C=0 HO-C-H HO-C-H H-C-OH H-C-OH H-C-OH CH,OH Glucose H-C-OH CH,OH Fructose OH OH OH CH-C-CH₂ reaction converts glucose to fructose. This reaction is catalyzed by a(n) OH OH OPO I CH-C-CH H Glycerol Glycerol 3-phosphate H reaction converts glycerol to glycerol 3-phosphate. This reaction requires H,N- H,N H…arrow_forwardAfter adding a small amount of ATP labeled with radioactive phosphorus in the terminal position, [7-32P]ATP, to a yeast extract, a researcher finds about half of the 32P activity in P; within a few minutes, but the concentration of ATP remains unchanged. She then carries out the same experiment using ATP labeled with 32P in the central position, [ẞ-³2P]ATP, but the 32P does not appear in P; within such a short time. Which statements explain these results? Yeast cells reincorporate P; released from [ß-³2P]ATP into ATP more quickly than P¡ released from [y-³2P]ATP. Only the terminal (y) phosphorous atom acts as an electrophilic target for nucleophilic attack. The terminal (y) phosphoryl group undergoes a more rapid turnover than the central (B) phosphate group. Yeast cells maintain ATP levels by regulating the synthesis and breakdown of ATP. Correct Answerarrow_forwardCompare the structure of the nucleoside triphosphate CTP with the structure of ATP. NH₂ 0- 0- 0- ·P—O—P—O—P—O—CH₂ H H H H OH OH Cytidine triphosphate (CTP) Consider the reaction: ATP + CDP ADP + CTP NH 0- 0- 0- ¯0— P—O— P—O—P-O-CH₂ H Η о H H OH OH Adenosine triphosphate (ATP) NH₂ Now predict the approximate K'eq for this reaction. Now predict the approximate AG for this reaction. Narrow_forward
 Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning
Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College
Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning





