
Concept explainers
Interpretation: Different structures for formula C5H10with one ring should be determined.
Concept introduction: C5H10have two hydrogen atoms less than the molecular formula of C5H12, i.e. it contains either a double bond or a cyclic group. So in
Answer to Problem 21P
- Cyclopentane
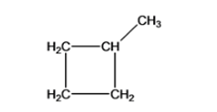
- Methylcyclobutane
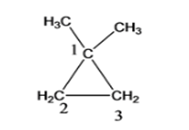
- 1,1-dimethyl cyclopropane
- 1,2- dimethyl cyclopropane
- Ethylcyclopropane
Explanation of Solution
Considering the structural formula C5H10 with a ring, the maximum number of carbon in that ring can be equal to or less than 5.
So the structures will be:
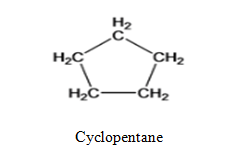
Cyclopentane
Other structures are:
Cyclobutane with one methyl substituent which is named as Methylcyclobutane
(According to IUPAC)
Cyclopropane with 2 methyl functional group (1, 1-dimethyl cyclopropane )
Cyclopropane with 2 methyl functional group, where functional group is attached to the adjacent C atoms of the cyclic groups (1,2- dimethylcyclopropane)
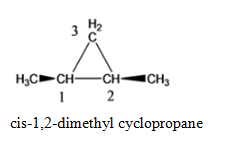
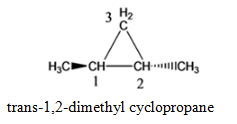
cis-1,2-dimethyl cyclopropane trans-1,2-dimethyl cyclopropane
Cyclopropane is another structure with 1 ethyl (CH2CH3) functional group is named as Ethylcyclopropane
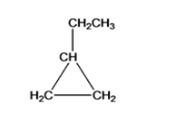
The five different structures can be written for the structural equation C5H10 and they are Cyclopentane, Methylcyclobutane, 1,1-dimethylcyclopropane, 1,2-dimethyl cyclopropane, ethylcyclopropane.
Want to see more full solutions like this?
Chapter 4 Solutions
EBK STUDY GUIDE/SOLUTIONS MANUAL FOR OR
- 20,0 Complete the electron pushing mechanism to y drawing the necomery unicaciones and carved on for Step 1: Add curved arms for the tint step, traiment with NalilĻ. The Nation 458 Step 2: Added for the second step, inalment with), how the "counterion bar Step 3: Daw the products of the last simplom organic and one incoganic spacient, including all nonbondingarrow_forwardplease provide the structure for this problem, thank you!arrow_forwardDraw the Fischer projection from the skeletal structure shown below. HO OH OH OH OH H Q Drawing Atoms, Bonds and Rings Charges I ☐ T HO H H OH HO I CH2OH H OH Drag H OH -CH2OH CHO -COOH Undo Reset Remove Donearrow_forward
- please provide the structure for this problem, thank youarrow_forwardpresented by Morallen Lig Intermine the hand product for the given mution by adding atoms, bonds, nonhonding diarion panda скуль Step 3: Comp the draw the product Step 2: Agama workup Compithe 429 ملولةarrow_forwardReaction A 0,0arrow_forward
- presented by Morillon Leaning Predict the organic product for the min кусур HSC Adithane carved arnown to come than that to the condon slchroruis in acid in in aquishri with ноюarrow_forward6.15PM Sun Mar 30 K Draw the major product of this reaction. Include any relevant stereochemistry. Ignore inorganic byproducts. Problem 1 of O H [PhзPCH2CH3]*C|¯ NaH Drawing > Q Atoms, Bonds and Draw or tap a nearrow_forward8:17 PM Sun Mar 30 Draw the major product of this reaction. Ignore inorganic byproducts. HSCH2CH2CH2SH, BF3 Probler Drawing Ato Bonds Clarrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning




