
Concept explainers
Locate the isoprene units in each compound.
a.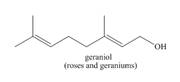 c.
c. 
b. 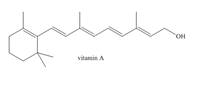 d.
d. 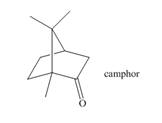
(a)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: An isoprene unit possesses following properties.
➢ It may either consist of
➢ It is always connected by one or more
➢ Each isoprene unit is consist of
➢ While locating an isoprene unit, the presence of heteroatoms is ignored.
Answer to Problem 30.11P
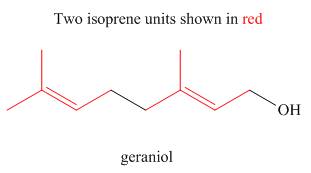
The isoprene units in the given terpene (geraniol) are located as,
Explanation of Solution
Terpenes are composed of repeating five-carbon units. These units are called isoprene units.
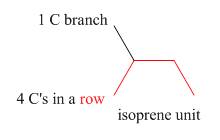
Figure 1
An isoprene unit possesses following properties.
➢ It involves one branched carbon and
➢ It may either consist of
➢ It is always connected by one or more
➢ Each isoprene unit is consist of
➢ While locating an isoprene unit, the presence of heteroatoms is ignored.
The isoprene units in the given terpene (geraniol) are located as,

Figure 2
The isoprene units in the given terpene (geraniol) are located in Figure 2.
(b)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: An isoprene unit possesses following properties.
➢ It may either consist of
➢ It is always connected by one or more
➢ Each isoprene unit is consist of
➢ While locating an isoprene unit, the presence of heteroatoms is ignored.
Answer to Problem 30.11P
The isoprene units in the given terpene (vitamin A) are located as,
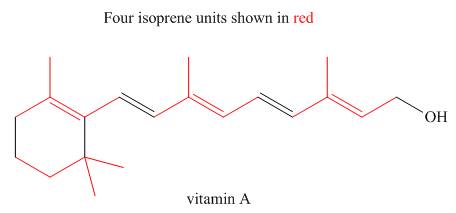
Explanation of Solution
Terpenes are composed of repeating five-carbon units. These units are called isoprene units.

Figure 1
An isoprene unit possesses following properties.
➢ It involves one branched carbon and
➢ It may either consist of
➢ It is always connected by one or more
➢ Each isoprene unit is consist of
➢ While locating an isoprene unit, the presence of heteroatoms is ignored.
The isoprene units in the given terpene (vitamin A) are located as,

Figure 3
The isoprene units in the given terpene (vitamin A) are located in Figure 3.
(c)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: An isoprene unit possesses following properties.
➢ It may either consist of
➢ It is always connected by one or more
➢ Each isoprene unit is consist of
➢ While locating an isoprene unit, the presence of heteroatoms is ignored.
Answer to Problem 30.11P
The isoprene units in the given terpene (grandisol) are located as,
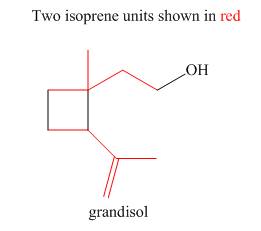
Explanation of Solution
Terpenes are composed of repeating five-carbon units. These units are called isoprene units.

Figure 1
An isoprene unit possesses following properties.
➢ It involves one branched carbon and
➢ It may either consist of
➢ It is always connected by one or more
➢ Each isoprene unit is consist of
➢ While locating an isoprene unit, the presence of heteroatoms is ignored.
The isoprene units in the given terpene (grandisol) are located as,

Figure 4
The isoprene units in the given terpene (grandisol) are located in Figure 4.
(d)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: An isoprene unit possesses following properties.
➢ It may either consist of
➢ It is always connected by one or more
➢ Each isoprene unit is consist of
➢ While locating an isoprene unit, the presence of heteroatoms is ignored.
Answer to Problem 30.11P
The isoprene units in the given terpene (camphor) are located as,
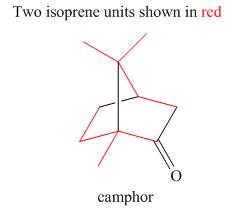
Explanation of Solution
Terpenes are composed of repeating five-carbon units. These units are called isoprene units.

Figure 1
An isoprene unit possesses following properties.
➢ It involves one branched carbon and
➢ It may either consist of
➢ It is always connected by one or more
➢ Each isoprene unit is consist of
➢ While locating an isoprene unit, the presence of heteroatoms is ignored.
The isoprene units in the given terpene (camphor) are located as,

Figure 5
The isoprene units in the given terpene (camphor) are located in Figure 5.
Want to see more full solutions like this?
Chapter 30 Solutions
Organic Chemistry-Package(Custom)
- 9. Write Me product as well as the reaction Mechanism For each of the Following Vanctions +H₂504 4.50+ T C. +212 Fellz 237 b. Praw the potential energy Diagrams For each OF Mese Rauctions and account For any differences that appear in the two potential Puergy Diagrams which of here two reactions 19 Found to be Reversable, Rationalice your answer based upon the venation mechanisms and the potential energy diagrams.arrow_forward9. Write Me product as well as the reaction Mechanism For each of the Following Veritious +H2504 4.50+ + 1/₂ Felly ◎+ 7 b. Praw he potential energy Diagrams For each OF Mese Ronctions and account for any differences that appeak in the two potential Puergy Diagramsarrow_forwardDraw the major product of this reaction. Ignore inorganic byproducts. Incorrect, 3 attempts remaining 1. excess Br2, NaOH 2. neutralizing workup Qarrow_forward
- Given the electrode Pt | Ag | Ag+ (aq), describe it.arrow_forwardAt 25°C, the reaction Zn2+ + 2e ⇄ Zn has a normal equilibrium potential versus the saturated calomel electrode of -1.0048 V. Determine the normal equilibrium potential of Zn versus the hydrogen electrode.Data: The calomel electrode potential is E° = 0.2420 V versus the normal hydrogen electrode.arrow_forwardElectrochemistry. State the difference between E and E0.arrow_forward
- In an electrolytic cell, the positive pole is always assumed to be on the right side of the battery notation. Is that correct?arrow_forwardIn an electrolytic cell, the positive pole is always assumed to be on the right side of the battery. Is that correct?arrow_forwardCalculate the free energy of formation of 1 mol of Cu in cells where the electrolyte is 1 mol dm-3 Cu2+ in sulfate solution, pH 0. E° for the Cu2+/Cu pair in this medium is +142 mV versus ENH.Assume the anodic reaction is oxygen evolution.Data: EH2 = -0.059 pH (V) and EO2 = 1.230 - 0.059 pH (V); 2.3RT/F = 0.059 Varrow_forward
- If the normal potential for the Fe(III)/Fe(II) pair in acid at zero pH is 524 mV Hg/Hg2Cl2 . The potential of the saturated calomel reference electrode is +246 mV versus the NHE. Calculate E0 vs NHE.arrow_forwardGiven the galvanic cell whose scheme is: (-) Zn/Zn2+ ⋮⋮ Ag+/Ag (+). If we know the normal potentials E°(Zn2+/Zn) = -0.76V and E°(Ag+/Ag) = 0.799 V. Indicate the electrodes that are the anode and the cathode and calculate the E0battery.arrow_forwardIndicate the functions that salt bridges have in batteries.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

